Advanced Decitabine Manufacturing: Leveraging Supercritical Fluid Technology for High-Purity API Production
The pharmaceutical landscape for oncology treatments continues to evolve, with nucleoside analogues playing a pivotal role in managing myelodysplastic syndromes and acute myeloid leukemia. A significant technological advancement in this domain is detailed in patent CN112142804B, which outlines a sophisticated preparation method for decitabine, also known as 5-aza-2'-deoxycytidine. This patent addresses the longstanding challenges associated with the stereochemical purity of decitabine, specifically the separation of the pharmacologically active beta-configuration from its inactive alpha-isomer. Traditional synthesis routes often struggle with low separation efficiency and excessive solvent consumption, creating bottlenecks for reliable pharmaceutical intermediates supplier networks seeking to optimize their supply chains. By integrating supercritical fluid technology with conventional organic synthesis, this method offers a pathway to achieve high-purity endpoints suitable for stringent regulatory requirements. The innovation lies not merely in the chemical transformation but in the downstream processing, where physical separation techniques are leveraged to enhance overall yield and reduce the environmental footprint of the manufacturing process. For R&D directors and procurement specialists, understanding the nuances of this patent provides critical insight into the future of cost-effective and high-quality API production.
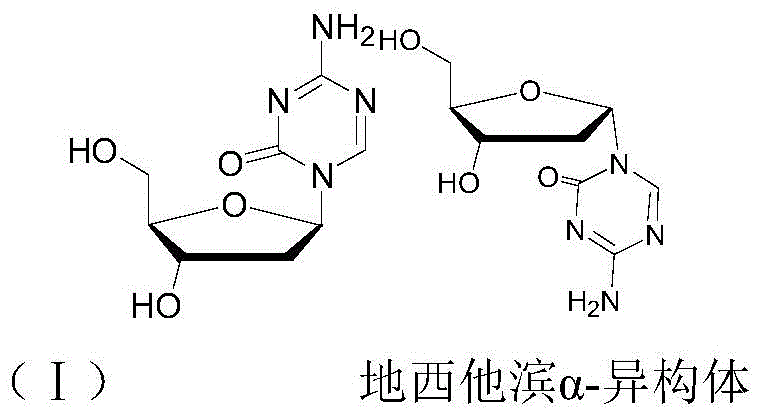
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of decitabine has been plagued by the formation of racemic mixtures containing both alpha and beta configurations, necessitating complex and costly purification strategies. As illustrated in the reaction scheme below, conventional methods typically involve reacting a 2-deoxy-D-ribose derivative with an activated 5-azacytosine product, resulting in a mixture of protected intermediates. Subsequent deprotection yields a mixture of decitabine and its alpha-isomer, which must then be separated. Prior art, such as US patent 3817980 and various academic disclosures, relies heavily on repeated recrystallization or chromatographic purification using silica gel columns. These traditional approaches are inherently inefficient; multiple recrystallization steps lead to substantial product loss, drastically reducing the overall yield and driving up the cost of goods sold. Furthermore, the extensive use of organic solvents like ethanol and ethyl acetate in these purification stages poses significant environmental and safety challenges, complicating waste management and increasing the operational burden on manufacturing facilities. The reliance on tin tetrachloride as a catalyst in some older routes also introduces the risk of heavy metal contamination, requiring additional purification steps to meet pharmaceutical grade standards.
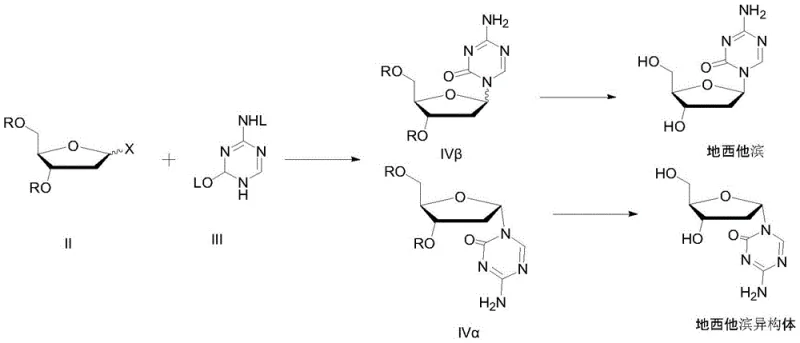
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a streamlined two-step process that combines chemical deprotection with physical separation via supercritical fluids. The process begins with the deprotection of the intermediate using an organic base system, specifically triethylamine in pyridine, which operates under mild conditions to preserve the integrity of the sensitive nucleoside structure. Following this, the crude mixture is subjected to supercritical fluid carbon dioxide treatment in a high-pressure vessel. This technique exploits the unique solvency properties of supercritical CO2, which can be finely tuned by adjusting temperature and pressure to selectively precipitate the desired beta-isomer while leaving impurities and the alpha-isomer in solution. This method effectively bypasses the need for labor-intensive column chromatography and reduces the number of recrystallization cycles required. The result is a significantly simplified workflow that enhances separation efficiency and minimizes solvent usage, aligning perfectly with modern green chemistry principles and the economic goals of a cost reduction in pharmaceutical intermediates manufacturing strategy.
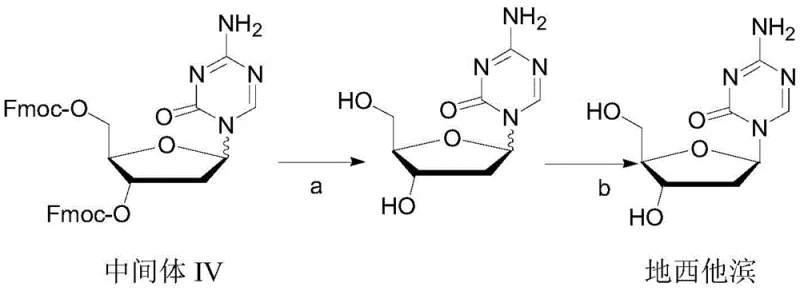
Mechanistic Insights into Supercritical Fluid Separation
The core of this technological breakthrough lies in the application of supercritical fluid technology for the resolution of stereoisomers, a mechanism that offers profound advantages over traditional crystallization. In the supercritical state, carbon dioxide exhibits properties of both a gas and a liquid, possessing high diffusivity like a gas and solvency power like a liquid. By maintaining the system at a temperature of 50-55°C and a pressure range of 50-70 MPa, the solvent power of the CO2-alcohol mixture is optimized to dissolve the impurity profile while allowing the high-purity decitabine to remain stable or precipitate selectively upon depressurization. The patent data indicates that a rapid temperature quench followed by gradual cooling promotes the formation of high-quality crystals with minimal inclusion of the alpha-isomer. This physical separation mechanism is driven by the subtle differences in solubility and crystal lattice energy between the two isomers under supercritical conditions. For technical teams, this implies a highly controllable process where purity can be managed through precise engineering parameters rather than relying solely on chemical selectivity, thereby reducing the variability often seen in batch crystallization processes.
Furthermore, the integration of activated carbon during the reflux extraction phase prior to supercritical treatment plays a crucial role in impurity control. The patent specifies the use of anhydrous methanol for reflux extraction with activated carbon at a specific mass ratio, which serves to adsorb colored impurities and residual organic byproducts generated during the deprotection step. This pre-purification step is vital for protecting the supercritical equipment from fouling and ensuring that the final separation stage operates with maximum efficiency. By removing bulk impurities early in the process, the load on the supercritical separation unit is reduced, allowing for sharper resolution of the target isomer. This multi-stage purification strategy ensures that the final product meets the rigorous purity specifications required for oncology drugs, with HPLC data from the patent examples showing purity levels reaching 99.91% and alpha-isomer content as low as 0.05%. Such high fidelity in impurity profiling is essential for ensuring patient safety and regulatory compliance in global markets.
How to Synthesize Decitabine Efficiently
The synthesis of high-purity decitabine via this patented route involves a carefully orchestrated sequence of chemical and physical operations designed to maximize yield and stereochemical purity. The process initiates with the dissolution of the protected intermediate in a pyridine solvent system, followed by the controlled addition of triethylamine to effect deprotection at ambient temperatures. Once the protecting groups are removed, the reaction mixture undergoes a workup involving reflux extraction with anhydrous methanol and activated carbon to remove color bodies and side products. The resulting solid, a mixture of decitabine and its alpha-isomer, is then transferred to a high-pressure vessel for the critical supercritical fluid separation step. Detailed standardized synthesis steps see the guide below.
- Dissolve decitabine intermediate IV in pyridine and treat with triethylamine at room temperature to remove protecting groups, followed by reflux extraction with anhydrous methanol and activated carbon.
- Dissolve the resulting mixture of decitabine and alpha-isomer in an alcohol solvent within a high-pressure vessel.
- Introduce liquid carbon dioxide to reach a supercritical state at 50-55°C and 50-70 MPa, then reduce temperature and pressure to precipitate high-purity decitabine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this supercritical fluid-based synthesis route presents compelling economic and operational advantages that extend beyond simple yield improvements. The primary benefit lies in the drastic simplification of the downstream processing workflow, which directly translates to reduced operational expenditures. By eliminating the need for multiple rounds of recrystallization and expensive chromatographic resins, the process significantly lowers the consumption of consumables and reduces the labor hours required for purification. Additionally, the use of supercritical carbon dioxide as a separation medium offers a sustainable alternative to volatile organic compounds; CO2 is non-toxic, non-flammable, and can be easily recycled within the system, thereby minimizing waste disposal costs and environmental compliance burdens. This alignment with green manufacturing standards not only reduces direct costs but also mitigates regulatory risks associated with solvent emissions and hazardous waste generation.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts like tin tetrachloride and the reduction in solvent volume contribute to a leaner cost structure. Without the need for expensive heavy metal scavengers or extensive solvent recovery distillation trains, the overall cost of goods sold is optimized. The qualitative improvement in process efficiency means that less raw material is wasted during purification, leading to better mass balance and higher effective throughput per batch. This logical deduction of cost savings is derived from the mechanistic simplification of the route, making it a robust choice for long-term commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the supercritical fluid process enhances supply chain continuity by reducing the dependency on specialized chromatography media which can face supply constraints. The equipment required for supercritical extraction is durable and capable of continuous operation, reducing downtime associated with column packing and regeneration. Furthermore, the milder reaction conditions and reduced use of hazardous reagents improve workplace safety, minimizing the risk of production stoppages due to safety incidents. This reliability ensures a steady flow of high-purity pharmaceutical intermediates to downstream formulation partners, securing the production schedule for critical oncology medications.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is facilitated by the inherent nature of supercritical fluid systems, which are easily adaptable to larger vessel sizes without losing separation efficiency. The closed-loop nature of the CO2 system ensures that emissions are contained, helping manufacturers meet increasingly stringent environmental regulations globally. The ability to recycle the alcohol solvent used in conjunction with CO2 further enhances the sustainability profile of the manufacturing process. This scalability ensures that the technology can support reducing lead time for high-purity pharmaceutical intermediates as market demand grows, without compromising on quality or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this decitabine synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and quality implications for potential manufacturing partners. Understanding these details is crucial for evaluating the technology's fit within existing production frameworks.
Q: How does supercritical fluid technology improve decitabine purity compared to traditional recrystallization?
A: Supercritical fluid technology utilizes the tunable solvency of carbon dioxide under high pressure to selectively separate the beta-isomer from the alpha-isomer based on subtle solubility differences, achieving purity levels exceeding 99.9% without the extensive solvent loss associated with multiple recrystallizations.
Q: What are the critical process parameters for the deprotection step in this synthesis?
A: The deprotection step requires precise control of the organic base ratio, specifically using triethylamine and pyridine at a mass-volume ratio of approximately 1:2 to 1:3 relative to the intermediate, maintained at room temperature (20-30°C) to prevent degradation while ensuring complete removal of protecting groups.
Q: Is this manufacturing process scalable for industrial production of oncology intermediates?
A: Yes, the process is designed for industrial scalability as it eliminates complex chromatographic columns and reduces reliance on large volumes of organic solvents, utilizing a closed-loop supercritical system that enhances safety and environmental compliance for large-scale API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Decitabine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of advanced synthesis technologies in delivering high-quality oncology therapeutics to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the supercritical fluid method described in CN112142804B can be successfully translated from the lab to the plant. We maintain stringent purity specifications and operate rigorous QC labs equipped to handle the complex analytical requirements of nucleoside analogues, guaranteeing that every batch meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of stereochemical separation and impurity control with precision.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your development and commercialization goals for decitabine and related pharmaceutical intermediates.
