Advanced Synthesis of Chiral BIDIME Selenium Sulfur Ligands for Commercial Scale Production
Introduction to High-Efficiency Ligand Synthesis
The landscape of asymmetric catalysis is constantly evolving, driven by the demand for more robust and efficient chiral ligands that can withstand the rigors of industrial production. Patent CN115490730B introduces a groundbreaking methodology for the synthesis of selenium and sulfur ligands based on the chiral BIDIME (benzoxepin bisaryl monophosphine) backbone. This technology represents a significant leap forward in the field of organophosphorus chemistry, addressing critical pain points related to synthesis complexity and product purity. By utilizing a direct oxidation strategy involving elemental selenium or sulfur powder, the process eliminates the need for complex multi-step functionalization sequences that have traditionally plagued ligand manufacturing. The resulting chiral BIDIME selenium/sulfur ligands exhibit exceptional catalytic universality and efficiency, making them ideal candidates for high-value pharmaceutical intermediate production where stereochemical control is paramount.
Furthermore, the patent highlights a novel purification technique employing chemically modified diatomite, which serves as a game-changer for achieving high-purity specifications required by regulatory bodies. This innovation not only simplifies the downstream processing but also ensures that the final ligand products possess the structural integrity necessary for sensitive catalytic cycles. For research and development teams seeking to optimize their catalytic systems, this patent offers a reliable pathway to access advanced ligand architectures that were previously difficult to synthesize at scale. The integration of simple inorganic reagents with sophisticated organic backbones demonstrates a clever approach to balancing cost-effectiveness with performance, setting a new standard for ligand development in the fine chemical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral phosphine ligands containing heavier chalcogens like selenium and sulfur has been fraught with challenges that hinder their widespread adoption in commercial settings. Traditional routes often rely on harsh reaction conditions, expensive transition metal catalysts, or moisture-sensitive reagents that require stringent exclusion of air and water throughout the entire process. These conventional methods frequently suffer from low atom economy and generate substantial amounts of hazardous waste, creating significant burdens for environmental compliance and waste management teams. Moreover, the purification of these ligands is notoriously difficult due to their tendency to oxidize further or decompose during standard chromatographic procedures, leading to inconsistent batch-to-batch quality. The reliance on specialized precursors that are not readily available on the bulk market further exacerbates supply chain vulnerabilities, causing delays and price volatility for manufacturers dependent on these critical catalytic components.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in patent CN115490730B utilizes a remarkably straightforward oxidation protocol that transforms chiral BIDIME derivatives into their corresponding selenium or sulfur analogues with exceptional efficiency. The core innovation lies in the direct reaction of the phosphine precursor with elemental selenium or sulfur powder in a mild solvent system, typically dichloromethane, at room temperature. This approach drastically reduces the energy consumption associated with heating or cooling reactors and minimizes the risk of thermal degradation of the sensitive chiral backbone. The simplicity of the reagent profile means that procurement teams can source materials easily without relying on exotic supply chains, thereby enhancing the overall resilience of the manufacturing process. Additionally, the reaction proceeds with high conversion rates, minimizing the formation of side products and simplifying the isolation of the target molecule.
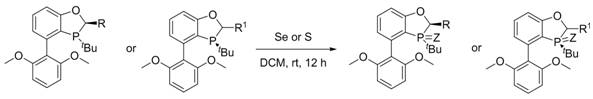
The visual representation of this synthetic route underscores its elegance, showing the direct transformation of the phosphorus center from the trivalent to the pentavalent state without altering the surrounding chiral environment. This preservation of stereochemistry is crucial for maintaining the enantioselectivity of the ligand in subsequent catalytic applications. By avoiding complex protecting group strategies or multi-step sequences, this novel approach significantly shortens the production timeline, allowing for faster turnaround from laboratory discovery to pilot plant validation. The robustness of this method suggests that it can be scaled up with minimal engineering modifications, providing a clear path for industrial implementation that addresses both technical and economic constraints faced by modern chemical manufacturers.
Mechanistic Insights into Chalcogen Oxidation of Phosphine Backbones
The mechanistic foundation of this synthesis relies on the nucleophilic attack of the trivalent phosphorus atom within the BIDIME backbone on the electrophilic selenium or sulfur species. In the presence of a non-polar or moderately polar solvent like dichloromethane, the lone pair on the phosphorus center attacks the elemental chalcogen, leading to the formation of a stable phosphorus-chalcogen double bond (P=Se or P=S). This oxidation step is thermodynamically favorable and kinetically accessible at ambient temperatures, which explains the high yields observed across various substrate derivatives. The rigid biaryl monophosphine skeleton of the BIDIME ligand plays a critical role in stabilizing the transition state, preventing unwanted rotation around the phosphorus-aryl bond that could lead to racemization. This structural rigidity ensures that the chiral information encoded in the backbone is faithfully transmitted to the catalytic active site, a feature that is essential for achieving high enantiomeric excess in asymmetric transformations.
Beyond the primary oxidation event, the patent introduces a sophisticated purification mechanism involving surface-modified diatomite that warrants detailed attention from a process chemistry perspective. The diatomite is treated with dimethylcarbamoyl chloride and p-methoxycinnamoyl chloride to alter its surface properties, specifically reducing the density of hydroxyl groups and increasing hydrophobicity through the introduction of aromatic rings. This modification prevents the strong adsorption of the organic ligand onto the silica surface, which is a common cause of yield loss in traditional filtration methods. Instead, the modified diatomite acts as a highly efficient scavenger for polar impurities and unreacted inorganic salts while allowing the target ligand to pass through with minimal retention. This selective filtration capability results in a crude product of significantly higher purity before it even reaches the final column chromatography stage, thereby reducing the load on downstream purification units and extending the life of chromatographic media.
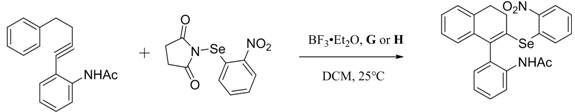
The utility of these ligands extends beyond their synthesis, as demonstrated by their application in the construction of complex axial chiral architectures. The catalytic cycle involves the coordination of the selenium or sulfur-modified phosphine to a metal center, likely palladium or similar transition metals, facilitating the coupling of benzyne derivatives with selenylaryl reagents. The steric bulk provided by the tert-butyl groups and the electronic tuning from the chalcogen atom create a unique pocket that discriminates between prochiral faces of the substrate. This level of precision is vital for the synthesis of bioactive molecules where a single enantiomer is required for therapeutic efficacy. The ability of these ligands to tolerate a wide range of functional groups further expands their applicability, allowing medicinal chemists to explore diverse chemical spaces without the need for extensive protecting group manipulation. This versatility makes the chiral BIDIME selenium/sulfur ligand platform a powerful tool in the arsenal of modern drug discovery and process development.
How to Synthesize Chiral BIDIME Selenium Sulfur Ligand Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to anhydrous and anaerobic protocols to prevent premature oxidation or hydrolysis of the sensitive phosphine intermediates. The process begins with the dissolution of the chiral BIDIME derivative in ultra-dry dichloromethane, ensuring that the solvent is free from any trace moisture that could interfere with the reaction kinetics. Once the substrate is fully solubilized, a stoichiometric excess of elemental selenium or sulfur powder is introduced to the reaction vessel under an inert atmosphere, typically argon or nitrogen. The mixture is then stirred at room temperature for a period ranging from 10 to 15 hours, allowing sufficient time for the oxidation to reach completion as monitored by thin-layer chromatography. Following the reaction, the mixture undergoes a critical filtration step using the aforementioned modified diatomite to remove insoluble residues and polar byproducts.
- Dissolve the chiral BIDIME derivative in ultra-dry dichloromethane under anhydrous and anaerobic conditions.
- Add inorganic simple substances such as selenium powder or sulfur powder at room temperature and stir for 10 to 15 hours.
- Filter the reaction mixture using modified diatomite, concentrate the filtrate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this synthesis method offers profound advantages that directly impact the bottom line and operational stability of chemical manufacturing operations. The primary driver for cost optimization lies in the substitution of expensive, specialized oxidizing agents with commodity-grade elemental selenium or sulfur powder. These inorganic reagents are abundant, globally sourced, and priced at a fraction of the cost of traditional organometallic oxidants, leading to a drastic reduction in raw material expenditure per kilogram of finished ligand. Furthermore, the elimination of transition metal catalysts from the synthesis step itself removes the necessity for costly and time-consuming heavy metal scavenging processes, which are often required to meet stringent residual metal specifications in pharmaceutical grades. This simplification of the workflow not only lowers direct material costs but also reduces the consumption of auxiliary chemicals and solvents associated with additional purification stages.
- Cost Reduction in Manufacturing: The streamlined nature of this process significantly lowers the operational expenditure associated with ligand production by minimizing unit operations and energy consumption. Since the reaction proceeds efficiently at room temperature, there is no requirement for energy-intensive heating or cryogenic cooling systems, resulting in substantial savings on utility costs over the lifecycle of the product. The high reaction yield, consistently reported above 90 percent, ensures that raw material utilization is maximized, reducing the waste disposal costs associated with low-efficiency processes. Additionally, the robustness of the modified diatomite purification method allows for the reuse or regeneration of filtration media in some configurations, further driving down the recurring costs of consumables. These cumulative efficiencies translate into a more competitive pricing structure for the final ligand product, enabling downstream users to achieve better margins in their own manufacturing value chains.
- Enhanced Supply Chain Reliability: The reliance on universally available inorganic reagents such as sulfur and selenium powder mitigates the risk of supply disruptions that are common with proprietary or niche organic reagents. This commoditization of key inputs ensures that production schedules can be maintained even during periods of global supply chain volatility, providing a secure source of critical catalytic materials for long-term projects. The simplicity of the synthesis also means that the technology can be easily transferred between different manufacturing sites or contract manufacturing organizations without the need for highly specialized equipment or expertise. This flexibility allows supply chain managers to diversify their supplier base and establish redundant production capabilities, safeguarding against single-point failures. Moreover, the stability of the intermediates and the final product under standard storage conditions simplifies logistics and inventory management, reducing the need for cold chain shipping or specialized containment.
- Scalability and Environmental Compliance: The benign nature of the reagents and the absence of toxic byproducts make this process inherently safer and more environmentally friendly than conventional alternatives. The use of dichloromethane, while requiring proper handling, is a well-understood solvent in industrial settings with established recovery and recycling protocols, minimizing the environmental footprint of the operation. The high selectivity of the reaction reduces the generation of hazardous waste streams, easing the burden on waste treatment facilities and ensuring compliance with increasingly strict environmental regulations. From a scalability standpoint, the exothermic profile of the reaction is manageable, allowing for safe scale-up from gram to ton quantities without significant engineering hurdles. This ease of scale-up ensures that the supply of these high-performance ligands can grow in tandem with the demand from the pharmaceutical and agrochemical sectors, supporting the commercialization of new drugs and materials without bottlenecking at the catalyst supply stage.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide accurate guidance for potential adopters. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production workflows or research pipelines. The clarity provided here aims to resolve uncertainties regarding purity standards, yield expectations, and specific application scopes.
Q: What is the primary advantage of using modified diatomite in this synthesis?
A: The use of modified diatomite, treated with dimethylcarbamoyl chloride and p-methoxycinnamoyl chloride, significantly enhances the purity of the final ligand by improving dispersion in organic solvents and reducing agglomeration during filtration.
Q: What yields can be expected from this oxidation method?
A: The patent data indicates that the product yield for the chiral BIDIME selenium/sulfur ligand is consistently greater than or equal to 90 percent, with preferred embodiments achieving yields of 96 percent or higher.
Q: What are the catalytic applications of these ligands?
A: These ligands are specifically designed for organic chiral catalytic reactions, including the preparation of axial chiral selenium/sulfur diaryl derivatives via coupling reactions with benzyne derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral BIDIME Ligand Supplier
As the global demand for high-performance chiral catalysts continues to surge, NINGBO INNO PHARMCHEM stands ready to support your development goals with our advanced manufacturing capabilities and deep technical expertise. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory bench to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and analytical instruments capable of meeting stringent purity specifications, guaranteeing that every batch of chiral BIDIME ligand we deliver performs consistently in your critical catalytic processes. We understand the critical importance of supply continuity in the pharmaceutical industry and have built a resilient supply chain that prioritizes reliability and quality above all else.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how switching to this method can optimize your overall production economics. We encourage you to reach out for specific COA data and route feasibility assessments to validate the compatibility of our ligands with your existing processes. Our commitment to partnership extends beyond mere supply; we act as a strategic ally in your journey to bring novel therapeutics and advanced materials to market faster and more cost-effectively.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
