Revolutionizing Azaphenanthrone Production: A High-Efficiency Pd-Catalyzed Route for Commercial Scale-Up
Revolutionizing Azaphenanthrone Production: A High-Efficiency Pd-Catalyzed Route for Commercial Scale-Up
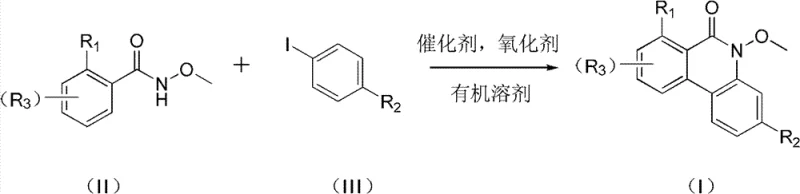
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for more efficient, cost-effective, and scalable methodologies. A pivotal advancement in this domain is detailed in patent CN102127014B, which discloses a novel class of azaphenanthrone compounds and a highly effective preparation method thereof. These nitrogen-containing heterocyclic structures are not merely academic curiosities; they serve as critical scaffolds in the development of bioactive molecules with potential applications in oncology and other therapeutic areas. The patent outlines a robust synthetic strategy that bypasses the limitations of classical approaches, utilizing a palladium-catalyzed oxidative coupling between hydroxamic acid derivatives and iodoarenes. This technical breakthrough offers a compelling value proposition for R&D directors seeking high-purity intermediates and procurement managers aiming for supply chain optimization. By leveraging this direct cyclization strategy, manufacturers can achieve superior regioselectivity and yield profiles compared to legacy methods.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the azaphenanthrone core has been a formidable challenge in organic synthesis, often plagued by multi-step sequences and harsh reaction conditions. Traditional routes frequently rely on Suzuki coupling followed by nitrile hydrolysis and intramolecular cyclization, or the reduction of nitro compounds and carboxylates followed by condensation. These conventional pathways suffer from significant drawbacks, including the requirement for difficult-to-prepare starting materials that are not readily available on the bulk chemical market. Furthermore, methods involving intramolecular Heck reactions often necessitate expensive ligands and stringent anhydrous conditions, which complicates process safety and increases operational expenditures. The cumulative effect of these inefficiencies is a prolonged lead time for drug development projects and inflated costs for the final active pharmaceutical ingredient, creating a bottleneck for commercial viability.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the methodology described in CN102127014B introduces a streamlined, one-pot oxidative cyclization that dramatically simplifies the synthetic landscape. This novel approach utilizes readily accessible iodoarenes and hydroxamic acids as building blocks, reacting them in the presence of a divalent palladium catalyst and a mild oxidant. The reaction proceeds smoothly in common organic solvents such as toluene, DMF, or acetic acid at temperatures ranging from 90°C to 130°C. This shift represents a paradigm change in how these complex heterocycles are accessed, eliminating the need for pre-functionalized precursors like cyano-groups or nitro-compounds. The result is a process that is not only chemically elegant but also industrially pragmatic, offering a direct path from simple commodity chemicals to high-value intermediates with minimal purification burden.
Mechanistic Insights into Pd-Catalyzed Oxidative Cyclization
The core of this technological advancement lies in the sophisticated yet efficient palladium catalytic cycle that drives the formation of the carbon-nitrogen and carbon-carbon bonds simultaneously. The mechanism likely initiates with the oxidative addition of the aryl iodide to the Pd(II) species, generating an organopalladium intermediate. This is followed by the coordination and insertion of the hydroxamic acid moiety, facilitated by the electron-rich nature of the nitrogen atom. The subsequent reductive elimination step closes the ring to form the azaphenanthrone skeleton, regenerating the active catalyst species in the presence of the oxidant, such as silver acetate or oxone. This oxidative regeneration is crucial, as it allows the use of catalytic amounts of palladium (0.05 to 0.2 mmol relative to substrate), thereby minimizing heavy metal contamination in the final product—a critical parameter for pharmaceutical compliance.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-temperature thermal cyclizations. The directed nature of the palladium coordination ensures high regioselectivity, preventing the formation of unwanted isomers that are notoriously difficult to separate via chromatography. The patent data indicates that varying the substituents R1, R2, and R3 (methyl, halogen, methoxy, ethoxycarbonyl) does not significantly compromise the reaction efficiency, with yields consistently remaining above 70%. This robustness suggests that the electronic and steric properties of the substrates are well-tolerated by the catalytic system. For R&D teams, this implies a versatile platform technology capable of generating diverse libraries of analogs for structure-activity relationship (SAR) studies without needing to re-optimize reaction conditions for every new derivative.
How to Synthesize Azaphenanthrone Efficiently
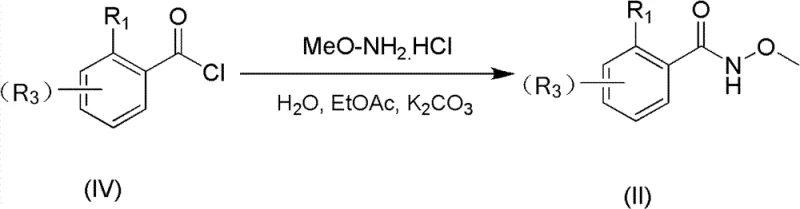
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the preparation of the hydroxamic acid precursor, which serves as the nucleophilic partner in the coupling reaction. As detailed in the patent, this precursor is easily synthesized by reacting the corresponding aryl acid chloride with methoxyamine hydrochloride in a biphasic system of water and ethyl acetate, using potassium carbonate as a base. This preliminary step is high-yielding (often exceeding 90%) and produces the necessary hydroxamic acid in high purity. Once the precursor is secured, the main cyclization can be executed by simply mixing the reagents in a Schlenk tube or reactor, heating to the specified temperature, and stirring until conversion is complete. The detailed standardized synthesis steps for this process are outlined below.
- Prepare the hydroxamic acid precursor by reacting the corresponding aryl acid chloride with methoxyamine hydrochloride in the presence of a base like potassium carbonate.
- Combine the hydroxamic acid, iodoarene substrate, palladium catalyst (e.g., PdCl2 or Pd(OAc)2), and a silver-based oxidant in an organic solvent such as toluene or DMF.
- Heat the reaction mixture to 90-130°C for 8-20 hours, then filter and purify the crude product via column chromatography to isolate the target azaphenanthrone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits that extend far beyond the laboratory bench. The primary advantage lies in the drastic simplification of the raw material portfolio. By relying on commodity chemicals like iodoarenes and acid chlorides, companies can mitigate the risks associated with sourcing exotic or custom-synthesized starting materials that often have long lead times and volatile pricing. This shift towards widely available feedstocks enhances supply chain resilience, ensuring that production schedules are not disrupted by single-source supplier issues. Furthermore, the reduction in synthetic steps directly correlates to a reduction in overall processing time and labor costs, allowing for faster turnaround on custom synthesis orders.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the elimination of expensive reagents and the minimization of waste generation. Traditional methods often require stoichiometric amounts of organometallic reagents or harsh reducing agents, which are costly to purchase and dispose of. In contrast, this catalytic method uses sub-stoichiometric amounts of palladium and benign oxidants, significantly lowering the cost of goods sold (COGS). Additionally, the high reaction yields reported (up to 92%) mean that less raw material is wasted, maximizing the output per batch and improving the overall atom economy of the process.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable starting materials such as iodoarenes and hydroxamic acids ensures a consistent supply flow. Unlike sensitive organometallic reagents that require cold storage and inert atmosphere handling, the reagents for this process are robust and easy to transport. This stability reduces the logistical complexity and cost associated with raw material storage and handling. For global supply chains, this means that production can be decentralized or scaled up in different geographic regions without compromising on quality or consistency, providing a strategic buffer against regional disruptions.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns well with green chemistry principles. The use of solvents like toluene and acetic acid, while requiring proper management, is standard in the industry and easier to recover and recycle compared to more exotic solvents. The simplified workup procedure, involving filtration and standard column chromatography, reduces the volume of aqueous waste generated. This ease of scalability makes the process ideal for transitioning from gram-scale discovery to kilogram or ton-scale commercial production, ensuring that environmental permits and waste treatment facilities are not overwhelmed by complex effluent streams.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this azaphenanthrone synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a realistic overview of what partners can expect when adopting this methodology. Understanding these nuances is essential for making informed decisions about process integration and resource allocation.
Q: What are the primary advantages of this Pd-catalyzed method over traditional Suzuki couplings?
A: Unlike traditional methods requiring complex pre-functionalized starting materials like cyano-groups or nitro-compounds, this method utilizes readily available iodoarenes and hydroxamic acids, significantly reducing raw material costs and synthesis steps.
Q: What is the typical yield range for these azaphenanthrone derivatives?
A: According to the patent data, the reaction consistently achieves high yields ranging from 72% to 92% across various substituted derivatives, demonstrating robust process reliability.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the use of common organic solvents like toluene and acetic acid, combined with standard filtration and chromatography workups, makes this protocol highly adaptable for kilogram-to-ton scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azaphenanthrone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of azaphenanthrone intermediate meets the highest international standards. Our commitment to technical excellence means we can rapidly adapt this patented methodology to meet your specific customization needs, whether you require milligram quantities for screening or metric tons for clinical supply.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make data-driven decisions that optimize both your timeline and budget. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing.
