Revolutionizing Azaphenanthrone Production: A Scalable Pd-Catalyzed Route for Pharmaceutical Intermediates
Revolutionizing Azaphenanthrone Production: A Scalable Pd-Catalyzed Route for Pharmaceutical Intermediates
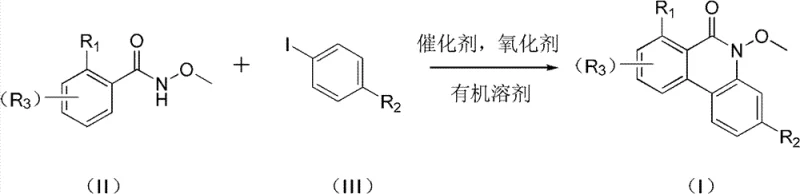
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for more efficient, cost-effective, and scalable methodologies. A significant breakthrough in this domain is documented in patent CN102127014B, which discloses a novel class of azaphenanthrone compounds and their preparation methods. These compounds, characterized by the general structure shown in Formula (I), serve as critical building blocks for a wide array of bioactive molecules and drug candidates. The patent highlights a robust synthetic strategy that leverages palladium-catalyzed oxidative coupling, offering a distinct advantage over legacy synthetic routes. For R&D directors and procurement specialists alike, understanding the nuances of this technology is paramount for securing a reliable supply chain of high-purity pharmaceutical intermediates. The structural versatility allowed by varying substituents R1, R2, and R3 enables the fine-tuning of physicochemical properties essential for downstream drug development.

The core innovation lies in the direct construction of the azaphenanthrone scaffold through a convergent approach. By utilizing readily accessible starting materials, specifically hydroxamic acid derivatives and iodoarenes, the process circumvents the need for laborious multi-step sequences often associated with heterocyclic synthesis. This method not only streamlines the production workflow but also enhances the overall atom economy of the transformation. As the pharmaceutical industry increasingly demands greener and more sustainable manufacturing practices, this catalytic approach stands out as a viable solution for the commercial scale-up of complex pharmaceutical intermediates. The ability to introduce diverse functional groups at multiple positions on the aromatic rings further underscores the utility of this platform for generating focused libraries of analogs during the lead optimization phase.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of azaphenanthrone derivatives has been plagued by significant inefficiencies and reliance on harsh reaction conditions. Traditional pathways often involve Suzuki coupling reactions followed by hydrolysis of cyano groups to amides and subsequent intramolecular cyclization, a sequence that is both time-consuming and prone to low overall yields. Another conventional route involves the reduction of carboxylates and nitro compounds using zinc reagents and acetic acid, which generates substantial amounts of metal waste and requires rigorous purification protocols to remove heavy metal residues. Furthermore, intramolecular Heck reactions, while effective for ring closure, typically demand expensive ligands and strictly anhydrous conditions, driving up the cost of goods sold (COGS). These legacy methods often suffer from poor regioselectivity and limited substrate scope, restricting the chemical diversity accessible to medicinal chemists. Consequently, the search for a more direct and operationally simple methodology has been a priority for process research teams aiming to reduce lead time for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these cumbersome traditional techniques, the method disclosed in CN102127014B introduces a streamlined palladium-catalyzed oxidative coupling strategy. This novel approach directly couples a hydroxamic acid compound (II) with an iodoarene compound (III) in the presence of a divalent palladium catalyst and an oxidant. The reaction proceeds smoothly in common organic solvents such as toluene, DMF, or acetic acid at moderate temperatures ranging from 90 to 130°C. This eliminates the need for pre-functionalized precursors like organoboron species or sensitive organometallic reagents, thereby simplifying the raw material sourcing process. The use of silver salts or oxone as oxidants facilitates the regeneration of the active palladium species, ensuring catalytic turnover without the need for stoichiometric amounts of expensive metals. This direct C-H activation and cyclization cascade significantly reduces the number of unit operations, translating to substantial cost savings in pharmaceutical intermediate manufacturing. The robustness of this method is evidenced by its tolerance to various substituents, allowing for the efficient synthesis of a broad library of azaphenanthrone derivatives with high purity.
Mechanistic Insights into Pd-Catalyzed Oxidative Cyclization
The mechanistic underpinning of this transformation involves a sophisticated interplay between the palladium catalyst, the oxidant, and the organic substrates. Initially, the divalent palladium species, such as PdCl2 or Pd(OAc)2, coordinates with the hydroxamic acid moiety, likely facilitating the activation of the ortho-C-H bond on the aromatic ring. This step is crucial for setting the stage for the subsequent C-C bond formation. The presence of the iodoarene suggests an oxidative addition step where the palladium center inserts into the carbon-iodine bond, forming an aryl-palladium intermediate. The oxidant plays a dual role: it not only re-oxidizes the palladium(0) species back to palladium(II) to close the catalytic cycle but may also assist in the final reductive elimination step that forms the new carbon-carbon bond, closing the phenanthrone ring system. Understanding this catalytic cycle is vital for R&D teams aiming to optimize reaction parameters such as temperature and catalyst loading to maximize yield and minimize side products. The choice of solvent, whether polar aprotic like DMF or non-polar like toluene, can significantly influence the solubility of the ionic intermediates and the stability of the catalyst, thereby affecting the overall reaction kinetics.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-temperature thermal cyclizations. The directed nature of the palladium coordination ensures high regioselectivity, minimizing the formation of isomeric byproducts that are notoriously difficult to separate. The mild reaction conditions (90-130°C) prevent the degradation of sensitive functional groups such as esters or methoxy groups, which might otherwise decompose under the harsh conditions required for traditional Friedel-Crafts type cyclizations. Furthermore, the use of well-defined divalent palladium catalysts allows for precise control over the metal loading, typically in the range of 0.05 to 0.2 mmol relative to the substrate. This precision helps in keeping residual palladium levels in the final product within acceptable limits for pharmaceutical applications, reducing the burden on downstream purification processes. The ability to tune the electronic properties of the catalyst ligands, although not explicitly detailed in the basic examples, offers a potential avenue for further process intensification and yield improvement in future iterations of this chemistry.
How to Synthesize Azaphenanthrone Compounds Efficiently
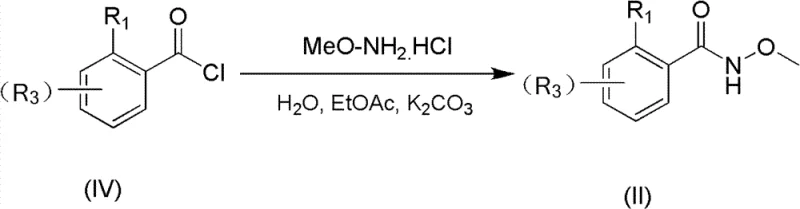
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the preparation of the hydroxamic acid precursor and the optimization of the coupling conditions. The process begins with the synthesis of the hydroxamic acid (II), which is achieved by reacting the corresponding aryl acid chloride (IV) with methoxyamine hydrochloride in a biphasic system of water and ethyl acetate, using potassium carbonate as a base. This step is highly efficient, yielding the precursor in excellent purity, which is critical for the success of the subsequent coupling reaction. Once the precursor is secured, the main cyclization is performed by combining all reagents in a Schlenk tube or reactor under inert atmosphere. The reaction mixture is heated to the specified temperature, typically around 110-125°C, and stirred for 8 to 20 hours depending on the specific substrate reactivity. Post-reaction workup is straightforward, involving filtration to remove insoluble salts followed by silica gel chromatography. For a detailed standard operating procedure regarding specific molar ratios and purification techniques, please refer to the guide below.
- Prepare the hydroxamic acid precursor (II) by reacting the corresponding aryl acid chloride with methoxyamine hydrochloride in the presence of a base like potassium carbonate.
- Combine the hydroxamic acid (II), iodoarene compound (III), a divalent palladium catalyst (e.g., PdCl2), and an oxidant (e.g., silver acetate) in an organic solvent such as toluene or DMF.
- Heat the reaction mixture to 90-130°C for 8-20 hours, then filter and purify the crude product via column chromatography to obtain the target azaphenanthrone compound (I).
To ensure the highest quality of the starting hydroxamic acid, the acylation step should be monitored closely to prevent over-reaction or hydrolysis of the acid chloride. The use of ethyl acetate as a co-solvent facilitates the extraction of the organic product while maintaining a homogeneous reaction environment for the base-mediated neutralization of the HCl byproduct. Following the isolation of compound (II), it can be directly subjected to the palladium-catalyzed step without extensive drying, provided the water content is controlled to prevent catalyst deactivation. This two-step sequence from commercially available acid chlorides represents a significant improvement in process efficiency.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits. The primary advantage stems from the utilization of commodity chemicals as starting materials. Aryl acid chlorides and iodoarenes are widely available from global chemical suppliers, ensuring a stable and competitive supply base. Unlike specialized organometallic reagents that require cold chain logistics and have short shelf lives, these precursors are stable solids or liquids that can be stocked in bulk, mitigating the risk of supply disruptions. The simplicity of the reaction setup, which does not require exotic ligands or ultra-low temperature equipment, further lowers the barrier to entry for contract manufacturing organizations (CMOs). This accessibility translates directly into cost reduction in pharmaceutical intermediate manufacturing, as the capital expenditure for specialized reactor setups is minimized. Additionally, the high yields reported in the patent examples, often exceeding 80%, mean that less raw material is wasted, improving the overall material throughput and reducing the environmental footprint associated with waste disposal.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal ligands and the use of catalytic rather than stoichiometric amounts of palladium significantly lower the direct material costs. Furthermore, the simplified workup procedure, which avoids complex aqueous extractions or distillations in favor of simple filtration and chromatography, reduces energy consumption and labor hours. By removing the need for multi-step functional group manipulations required in older syntheses, the overall process mass intensity (PMI) is drastically improved. This lean manufacturing approach ensures that the final cost per kilogram of the azaphenanthrone intermediate is highly competitive, allowing pharmaceutical companies to allocate more budget towards clinical development and marketing. The robustness of the chemistry also means fewer batch failures, which is a hidden but significant cost driver in chemical production.
- Enhanced Supply Chain Reliability: Sourcing reliability is paramount for maintaining continuous drug production. Since the key building blocks—acid chlorides and iodoarenes—are produced on a massive industrial scale for various sectors, the risk of single-source dependency is minimized. The reaction conditions are tolerant to minor variations in reagent quality, providing a buffer against supply chain fluctuations. Moreover, the relatively short reaction times (8-20 hours) allow for faster turnaround times compared to multi-day traditional syntheses. This agility enables manufacturers to respond quickly to changes in demand, ensuring that inventory levels can be optimized without the fear of long lead times. The ability to scale this reaction from gram to kilogram scale without significant re-optimization further strengthens the supply chain resilience, making it an ideal candidate for long-term commercial partnerships.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability increase, this process offers a greener alternative to legacy methods. The avoidance of toxic tin or zinc reagents reduces the generation of hazardous heavy metal waste, simplifying compliance with environmental regulations. The solvents used, such as toluene and acetic acid, are well-understood and can be effectively recovered and recycled in a closed-loop system, minimizing volatile organic compound (VOC) emissions. The high atom economy of the oxidative coupling means that a larger proportion of the reactant mass ends up in the final product, reducing the volume of waste streams that require treatment. This alignment with green chemistry principles not only enhances the corporate social responsibility profile of the manufacturer but also future-proofs the supply chain against tightening environmental legislation, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these azaphenanthrone compounds. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a clear picture of the technology's capabilities. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their drug discovery pipelines. The answers reflect the consensus on best practices for handling and synthesizing these valuable chemical entities.
Q: What are the key advantages of this Pd-catalyzed method over traditional Suzuki couplings?
A: Unlike traditional methods requiring complex pre-functionalized starting materials like cyano-groups or nitro-compounds, this method utilizes readily available iodoarenes and hydroxamic acids, significantly reducing raw material costs and synthesis steps.
Q: What represents the optimal temperature range for this oxidative cyclization?
A: The patent data indicates an optimal temperature range of 90-130°C. Lower temperatures may result in incomplete conversion, while higher temperatures could degrade sensitive functional groups, making this range ideal for balancing yield and purity.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process uses common organic solvents like toluene and acetic acid and avoids extremely hazardous reagents. The simple workup involving filtration and column chromatography suggests high feasibility for scale-up in industrial settings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azaphenanthrone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in accelerating drug development timelines. Our team of expert chemists has extensively evaluated the synthetic route described in CN102127014B and has successfully adapted it for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of azaphenanthrone intermediate delivered meets the highest industry standards. We understand that in the fast-paced world of pharmaceutical R&D, reliability is just as important as quality, and we are committed to being a partner you can trust.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Whether you need small quantities for preclinical studies or metric tons for commercial launch, we have the capacity and expertise to support you. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing processes can add value to your supply chain.
