Scalable Synthesis of Dialkoxyamine Intermediates for Next-Generation Antiviral Production
The pharmaceutical industry's demand for robust and scalable synthesis routes for antiviral agents has never been more critical, particularly for cap-dependent endonuclease inhibitors like Baloxavir Marvoxil (Xofluza). Patent CN111170875B introduces a groundbreaking preparation method for dialkoxyamine compounds, which serve as vital intermediates in the production of this innovative influenza treatment. Unlike traditional pathways that struggle with moisture sensitivity and hazardous reagents, this novel approach leverages a streamlined four-step sequence starting from ethylene glycol. The methodology addresses key pain points in fine chemical manufacturing, offering a pathway that significantly enhances product purity and operational safety. By replacing unstable bases and dangerous deprotection agents with more manageable alternatives, this technology provides a reliable foundation for the commercial scale-up of complex pharmaceutical intermediates. For R&D teams and supply chain leaders, understanding this shift is essential for securing a stable supply of high-quality antiviral precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in WO2017221869, have historically plagued manufacturers with significant technical and safety hurdles that impede efficient production. A primary defect in these conventional routes is the reliance on sodium tert-butoxide as a base in critical synthesis steps; this reagent is notoriously hygroscopic, meaning it readily absorbs moisture from the atmosphere, leading to inconsistent stoichiometry and poor reaction reproducibility. Furthermore, the deprotection stages in older methodologies often necessitate the use of hydrazine hydrate, a reagent classified as highly dangerous due to its toxicity and potential explosiveness, thereby imposing severe safety protocols and waste disposal costs on the facility. Another major bottleneck is the physicochemical nature of the intermediates; specifically, the products in the third step of conventional methods exhibit high water solubility, making effective separation via aqueous workup nearly impossible and resulting in significant yield losses and purity issues for downstream processing. These cumulative inefficiencies create a fragile supply chain vulnerable to batch failures and regulatory scrutiny regarding worker safety and environmental compliance.
The Novel Approach
The methodology outlined in CN111170875B represents a paradigm shift by systematically eliminating the instability and hazards inherent in previous generations of synthesis. This new route initiates with a robust alkylation of ethylene glycol under controlled thermal conditions, bypassing the need for moisture-sensitive strong bases that compromise batch consistency. Crucially, the strategy employs a protective group strategy using sulfonyl chlorides, which allows for precise functional group manipulation without resorting to hazardous hydrazine derivatives for removal. The process is engineered to produce intermediates with optimized solubility profiles, ensuring that the final amine product can be isolated with high efficiency and minimal loss during workup. By integrating catalytic hydrogenation for the final reduction step, the method aligns with green chemistry principles, reducing the environmental footprint while simultaneously enhancing the safety profile of the manufacturing plant. This holistic redesign of the synthetic pathway ensures that the production of dialkoxyamine compounds is not only chemically superior but also commercially viable for large-scale operations.
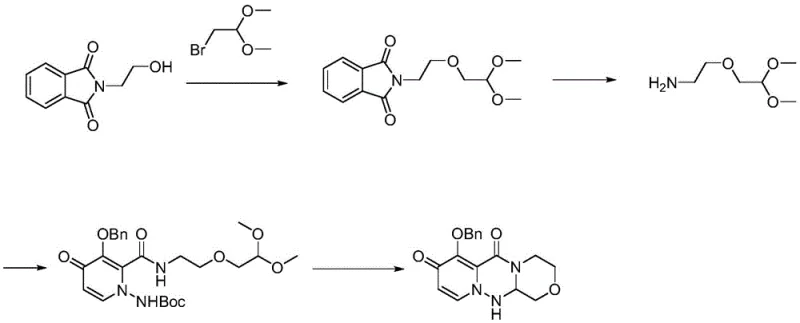
Mechanistic Insights into the Four-Step Dialkoxyamine Synthesis
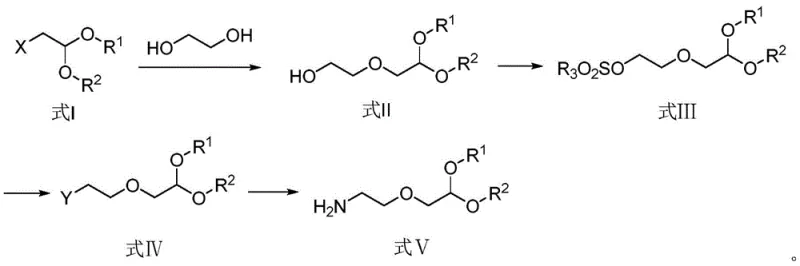
The core of this technological advancement lies in a meticulously orchestrated four-step reaction sequence that transforms simple starting materials into the complex Formula V intermediate. The process begins with the nucleophilic attack of ethylene glycol on a halo-acetal derivative in the presence of an alkali metal hydroxide, forming the hydroxyl-substituted ether linkage found in Formula II. This is followed by a protection step where the newly formed hydroxyl group is converted into a sulfonate ester (Formula III) using reagents like methanesulfonyl chloride, effectively turning a poor leaving group into an excellent one for subsequent substitution. The third stage involves a nucleophilic displacement reaction where an amine source, such as sodium azide, attacks the sulfonate carbon to install the nitrogen functionality, yielding the azide intermediate (Formula IV). Finally, the sequence concludes with a catalytic hydrogenation over Pd/C, which cleanly reduces the azide group to the primary amine while simultaneously removing any benzyl-type protecting groups if present, delivering the target dialkoxyamine with exceptional fidelity.
Impurity control is inherently built into this mechanism through the selection of reagents that minimize side reactions and facilitate easy purification. By avoiding the use of hydrazine, the process eliminates the formation of hydrazine-related byproducts that are notoriously difficult to purge from the final API. The use of distinct organic solvents for each step, such as dichloromethane for protection and ethyl acetate for reduction, allows for straightforward phase separations that wash away inorganic salts and unreacted starting materials. The high yields observed in the experimental data, reaching up to 98.1% in the final hydrogenation step, suggest that the reaction kinetics are highly favorable and that competing degradation pathways are effectively suppressed. This level of control over the reaction trajectory ensures that the impurity profile of the intermediate remains well within the stringent specifications required for pharmaceutical grade materials, reducing the burden on downstream purification processes.
How to Synthesize Dialkoxyamine Compound Efficiently
The synthesis of this critical intermediate requires precise control over reaction parameters to maximize the benefits of the new protocol. Operators must maintain strict temperature gradients, particularly during the initial alkylation where temperatures between 40°C and 120°C are utilized to drive the reaction to completion without degrading the sensitive acetal moiety. The choice of base in the first step is flexible, allowing for the use of sodium hydroxide or potassium hydroxide, which are far more stable and cost-effective than the tert-butoxides used in legacy methods. Detailed standardized operating procedures for each transformation, including specific molar ratios and solvent volumes, are essential to replicate the high purity and yield demonstrated in the patent examples. For a comprehensive breakdown of the exact experimental conditions and workup procedures, please refer to the technical guide below.
- React ethylene glycol with alkali and a halo-acetal derivative at 40-120°C to form the hydroxyl-substituted intermediate (Formula II).
- Protect the hydroxyl group of Formula II using a sulfonyl chloride (e.g., methanesulfonyl chloride) and an organic base to yield Formula III.
- Perform nucleophilic substitution on Formula III using an amine source such as sodium azide in a polar solvent at 40-110°C to generate the azide intermediate (Formula IV).
- Reduce the azide group in Formula IV to the primary amine (Formula V) using hydrogen gas and a Pd/C catalyst in an autoclave.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits that extend beyond mere chemical elegance. The elimination of hazardous reagents like hydrazine hydrate drastically simplifies the regulatory compliance landscape, reducing the costs associated with specialized storage, handling, and waste disposal of toxic materials. Furthermore, the robustness of the reaction conditions means that production schedules are less likely to be disrupted by batch failures caused by reagent instability or moisture ingress, ensuring a more predictable and reliable flow of materials. The simplified workup procedures, driven by the improved solubility characteristics of the intermediates, translate directly into reduced processing time and lower utility consumption per kilogram of product. These factors combine to create a manufacturing process that is not only safer and cleaner but also significantly more cost-efficient, providing a competitive edge in the sourcing of high-value antiviral intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive and sensitive reagents with commodity chemicals like ethylene glycol and sodium hydroxide drives down the raw material cost base significantly. Additionally, the high yields achieved in the final reduction step minimize the loss of valuable intermediates, ensuring that the overall material throughput is maximized. The avoidance of complex purification steps required to remove hydrazine residues further reduces the operational expenditure related to chromatography or extensive recrystallization. Consequently, the total cost of goods sold (COGS) for this intermediate is structurally lower compared to routes relying on legacy chemistry.
- Enhanced Supply Chain Reliability: By utilizing reagents that are widely available and stable under ambient conditions, the risk of supply disruption due to specialized chemical shortages is markedly reduced. The process tolerance to minor variations in reaction conditions ensures consistent output quality, which is critical for maintaining long-term contracts with pharmaceutical partners. This reliability allows supply chain planners to optimize inventory levels with greater confidence, knowing that the production lead times are stable and reproducible. Ultimately, this stability fosters stronger partnerships between chemical suppliers and drug manufacturers by guaranteeing the continuity of supply for critical medication pipelines.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations such as filtration and distillation that are standard in multi-ton manufacturing facilities. The removal of toxic hydrazine from the workflow significantly lowers the environmental impact, facilitating easier permitting and reducing the liability associated with hazardous waste generation. This alignment with green chemistry principles positions the manufacturer favorably in an increasingly regulated global market, where sustainability metrics are becoming key decision criteria for procurement. The ability to scale this process from laboratory grams to commercial tons without fundamental changes to the chemistry ensures a smooth transition from development to full-scale production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific manufacturing needs. The responses highlight the practical advantages in terms of safety, yield, and operational simplicity that distinguish this method from prior art.
Q: How does this new method improve upon the prior art WO2017221869?
A: The new method eliminates the use of hygroscopic sodium tert-butoxide, which caused reproducibility issues in previous methods. It also replaces hazardous hydrazine hydrate with safer catalytic hydrogenation and improves product isolation by avoiding highly water-soluble intermediates that were difficult to separate.
Q: What are the critical safety advantages of this synthesis route?
A: This route avoids the use of hydrazine hydrate, a highly toxic and potentially explosive reagent required in older methods for deprotection. Instead, it utilizes catalytic hydrogenation, which is a standard, controllable, and safer industrial process for reducing azides to amines.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It uses common solvents like ethylene glycol, dichloromethane, and ethyl acetate, and avoids complex purification steps. The high yields reported in the examples (up to 98.1% in the final step) indicate strong potential for cost-effective mass production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dialkoxyamine Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation antivirals depends on the availability of high-quality, reliably sourced intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of dialkoxyamine compound adheres to the highest industry standards. We are committed to supporting your R&D and commercial goals by providing a seamless bridge between innovative patent chemistry and industrial reality.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this safer, more efficient method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a robust and cost-effective supply of this critical pharmaceutical intermediate.
