Advanced Synthesis of Entecavir Intermediates via Naphthyl Silane Protection Strategy
Advanced Synthesis of Entecavir Intermediates via Naphthyl Silane Protection Strategy
The global demand for effective antiviral therapies continues to drive innovation in the synthesis of nucleoside analogs, particularly for the treatment of chronic hepatitis B. Patent CN101891741A discloses a groundbreaking methodology for the preparation of entecavir, a potent deoxyguanine nucleoside analog. This technical insight report analyzes the novel synthetic route which utilizes dimethylnaphthylchlorosilane as a strategic protecting group, offering a robust alternative to conventional processes that rely on expensive chiral pools. By leveraging sodium cyclopentadienide as a foundational building block, this approach addresses critical bottlenecks in cost, scalability, and operational complexity that have historically plagued the manufacturing of this high-value active pharmaceutical ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for entecavir, such as those disclosed in earlier patents, often depend on chiral starting materials like [1S-(1α,2α,3β,5α)]-2-[(benzyloxy)methyl]-6-oxabicyclo[3.1.0]hexan-3-ol. These precursors are not only prohibitively expensive but also suffer from limited commercial availability, creating significant supply chain vulnerabilities for manufacturers. Furthermore, legacy processes frequently necessitate the use of palladium-carbon catalysts for reduction steps and specialized methylating reagents like the Nysted reagent for methylenation. These reagents increase the overall cost of goods sold (COGS) and introduce complex purification challenges, often requiring column chromatography which is notoriously difficult to scale beyond pilot plant operations. The cumulative effect of these factors is a process with low overall yield, high waste generation, and substantial technical barriers to entry for generic manufacturers.
The Novel Approach
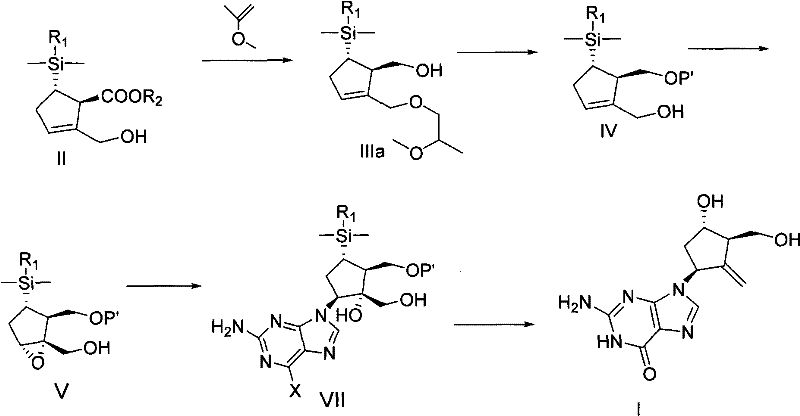
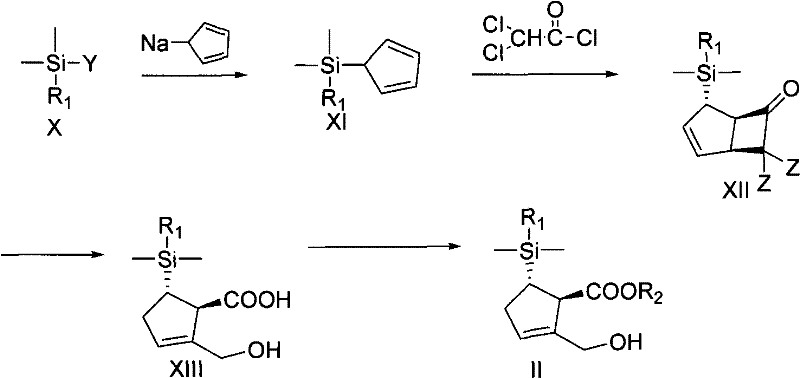
The methodology outlined in CN101891741A represents a paradigm shift by utilizing inexpensive and readily available raw materials to construct the carbocyclic core. The process initiates with the reaction of sodium cyclopentadienide with a silylating agent to form a silyl-cyclopentadiene derivative, which then undergoes a [2+2] cycloaddition with a ketene generated from dichloroacetyl chloride. This sequence efficiently builds the requisite carbon skeleton without relying on scarce chiral pools. A key innovation is the introduction of the dimethylnaphthylsilyl group, which serves a dual purpose: it acts as a masked hydroxyl group and facilitates chiral resolution through the formation of crystalline diastereomeric salts. This strategic modification eliminates the need for expensive chiral HPLC or extensive column chromatography, streamlining the path to high-purity intermediates suitable for downstream coupling.
Mechanistic Insights into Diastereoselective Epoxidation and Silyl Protection
The stereochemical integrity of the entecavir molecule is established through a highly controlled diastereoselective epoxidation step. In this critical transformation, the allylic alcohol intermediate is subjected to Sharpless-type epoxidation conditions using titanium isopropoxide, diethyl tartrate (DIPT), and tert-butyl hydroperoxide. The presence of the bulky dimethylnaphthylsilyl group exerts a profound steric influence, directing the approach of the oxidant to ensure the formation of the desired epoxide diastereomer with high fidelity. This level of stereocontrol is essential for the subsequent nucleophilic ring-opening by the purine base, as incorrect stereochemistry would lead to inactive isomers that are difficult to separate. The robustness of this epoxidation protocol ensures consistent optical purity throughout the synthesis, minimizing the burden on downstream purification units.
Furthermore, the choice of the naphthyl-substituted silane protecting group offers distinct mechanistic advantages over traditional phenyl analogues. The extended pi-system of the naphthyl ring enhances the stability of the silyl ether during basic and neutral conditions while remaining susceptible to cleavage under specific acidic or fluoride-mediated conditions. More importantly, the naphthyl group imparts solid-state properties to the intermediates that are conducive to crystallization. For instance, the racemic carboxylic acid precursor can be resolved using chiral amines to form diastereomeric salts that precipitate selectively from solution. This physical property allows for the enrichment of the desired enantiomer to high levels of optical purity simply through recrystallization, a unit operation that is vastly superior to chromatographic separation in terms of throughput and solvent consumption.
How to Synthesize Entecavir Efficiently
The synthesis of entecavir via this novel route involves a sequence of well-defined chemical transformations designed for industrial robustness. The process begins with the construction of the chiral cyclopentene core, followed by functional group manipulation to install the necessary hydroxyl and methylene moieties. The coupling of the carbocyclic intermediate with the purine base is performed under polar aprotic conditions to ensure high conversion rates. Subsequent steps involve the conversion of the vicinal diol into the exocyclic methylene group and the final deprotection of the silyl and benzyl groups to reveal the active drug substance. Detailed standard operating procedures for each reaction step, including specific reagent stoichiometry, temperature profiles, and workup protocols, are provided in the technical guide below.
- Prepare the key chiral ester intermediate by reacting sodium cyclopentadienide with a silylating agent, followed by cycloaddition and chiral resolution.
- Protect the hydroxyl group, reduce the ester to an alcohol, and perform diastereoselective epoxidation to establish the core stereochemistry.
- Couple the epoxide with a purine base, followed by condensation, desilylation, and oxidation to yield the final entecavir product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and operational benefits. By shifting away from proprietary chiral starting materials to commodity chemicals like cyclopentadiene and dichloroacetyl chloride, manufacturers can drastically reduce their exposure to volatile raw material pricing and single-source supplier risks. The elimination of precious metal catalysts such as palladium further contributes to cost reduction in entecavir manufacturing, removing the need for expensive metal scavenging steps and reducing the environmental footprint associated with heavy metal waste disposal. These structural changes to the bill of materials translate directly into improved margin potential and greater flexibility in sourcing strategies.
- Cost Reduction in Manufacturing: The replacement of expensive chiral pools and specialized reagents with bulk commodities significantly lowers the direct material costs. Additionally, the ability to purify intermediates via crystallization rather than column chromatography reduces solvent consumption and labor hours, leading to substantial cost savings in the production budget. The streamlined process flow also minimizes the number of unit operations required, thereby decreasing utility costs and equipment occupancy time.
- Enhanced Supply Chain Reliability: Reliance on widely available feedstocks ensures a stable and continuous supply of raw materials, mitigating the risk of production stoppages due to material shortages. The robustness of the chemistry allows for flexible manufacturing across multiple sites, enhancing the resilience of the supply network against regional disruptions. This reliability is critical for meeting the consistent demand patterns of global pharmaceutical markets.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, avoiding techniques that are difficult to transfer from the laboratory to the plant floor. The reduction in hazardous waste generation, particularly from the avoidance of column chromatography and heavy metal catalysts, simplifies regulatory compliance and waste management logistics. This alignment with green chemistry principles supports long-term sustainability goals and reduces the regulatory burden associated with environmental permits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of technology transfer and the potential return on investment for adopting this new manufacturing pathway.
Q: What are the advantages of using dimethylnaphthylchlorosilane over phenyl analogues?
A: The naphthyl group enhances the crystallinity of intermediates, facilitating easier chiral separation via diastereomeric salt formation. Additionally, the naphthyl moiety is more active than phenyl, allowing for milder deprotection conditions and higher product purity without requiring harsh reagents.
Q: Does this synthesis route require column chromatography for purification?
A: No, a significant advantage of this patented process is the elimination of column chromatography. The intermediates, particularly the chiral ammonium salts and final products, can be purified through crystallization, which is far more suitable for large-scale industrial production.
Q: Is the starting material sodium cyclopentadienide readily available?
A: Yes, sodium cyclopentadienide is derived from cyclopentadiene, which is a commodity chemical. This contrasts with prior art methods that relied on expensive, hard-to-obtain chiral starting materials like oxabicyclohexanes, significantly reducing raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Entecavir Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient and scalable synthesis routes for high-value antiviral agents. Our team of process chemists has extensively evaluated the methodology described in CN101891741A and possesses the technical expertise to optimize these reactions for commercial production. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of entecavir intermediate meets the highest international standards for safety and efficacy.
We invite pharmaceutical partners to collaborate with us on optimizing this cost-effective synthesis route. By leveraging our process development capabilities, we can help you achieve a Customized Cost-Saving Analysis tailored to your specific production volume and quality requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this advanced technology into your supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
