Optimizing Emtricitabine Production Through Novel Chiral Pool Synthesis And Commercial Scale-Up Strategies
The pharmaceutical landscape for antiretroviral therapies continues to evolve, with Emtricitabine standing as a cornerstone nucleoside reverse transcriptase inhibitor for treating HIV and HBV infections. Recent advancements documented in patent CN110467608B introduce a transformative synthetic methodology that addresses longstanding inefficiencies in producing this critical active pharmaceutical ingredient. This novel approach leverages dihaloacetic acid and L-menthol as foundational building blocks, offering a streamlined pathway that enhances both stereochemical control and economic viability. By shifting away from complex resolution techniques towards a chiral pool strategy, manufacturers can achieve superior optical purity without the prohibitive costs associated with traditional methods. 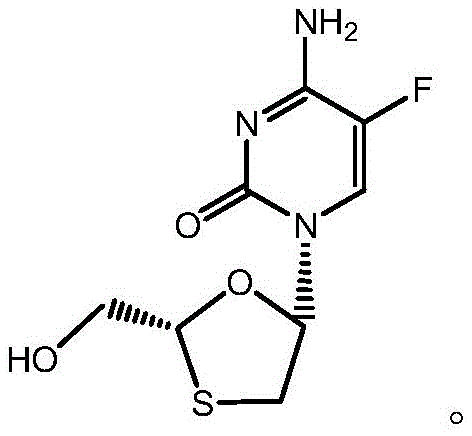 For R&D directors and procurement specialists, understanding this mechanistic shift is vital for securing a reliable emtricitabine supplier capable of meeting stringent global quality standards while optimizing cost reduction in API manufacturing.
For R&D directors and procurement specialists, understanding this mechanistic shift is vital for securing a reliable emtricitabine supplier capable of meeting stringent global quality standards while optimizing cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Emtricitabine has been plagued by significant technical and economic bottlenecks that hinder efficient commercial scale-up of complex nucleoside analogs. Traditional routes often rely on expensive chiral starting materials or require cumbersome resolution steps to separate enantiomers, leading to substantial material loss and inflated production costs. Furthermore, many legacy processes involve harsh reaction conditions, such as extreme temperatures or the use of toxic heavy metal catalysts, which complicate waste management and pose safety risks in large-scale facilities. The cumulative effect of these inefficiencies is a supply chain vulnerable to disruptions and a final product with variable purity profiles that may fail rigorous pharmacopeial specifications. These challenges necessitate a reevaluation of synthetic strategies to ensure reducing lead time for high-purity pharmaceutical ingredients remains achievable in a competitive market.
The Novel Approach
The methodology outlined in the referenced patent presents a robust alternative by utilizing L-menthol, a readily available chiral auxiliary, to dictate stereochemistry throughout the synthesis. This route begins with the esterification of dihaloacetic acid, a cheap and abundant raw material, which is then hydrolyzed to form menthyl glyoxylate. This intermediate undergoes condensation with 2,5-dihydroxy-1,4-dithiane to construct the critical oxathiolane ring system with high diastereoselectivity. Subsequent halogenation and coupling with silylated 5-fluorocytosine proceed under mild conditions, avoiding the degradation issues common in older protocols. 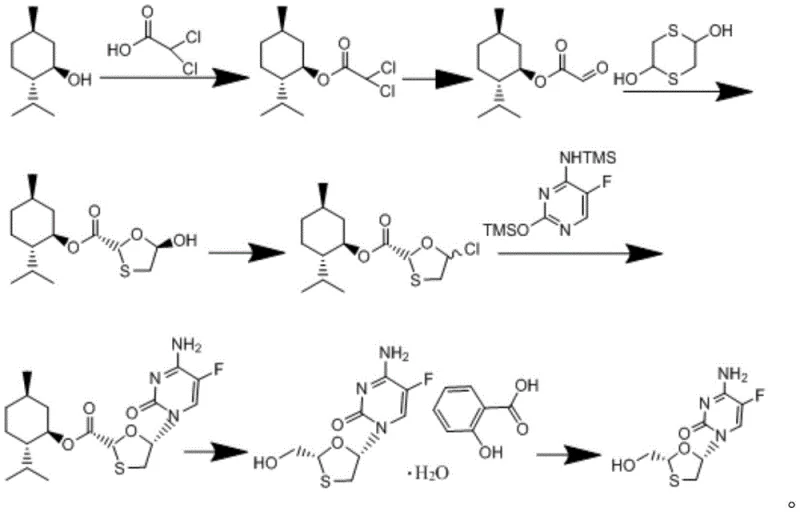 The final steps involve reduction and salification with salicylic acid, which serves as an effective purification handle to isolate the optically pure drug substance. This integrated approach not only simplifies the operational workflow but also aligns with modern green chemistry principles, making it an ideal candidate for cost reduction in electronic chemical manufacturing and broader pharmaceutical applications.
The final steps involve reduction and salification with salicylic acid, which serves as an effective purification handle to isolate the optically pure drug substance. This integrated approach not only simplifies the operational workflow but also aligns with modern green chemistry principles, making it an ideal candidate for cost reduction in electronic chemical manufacturing and broader pharmaceutical applications.
Mechanistic Insights into Menthyl Ester Mediated Cyclization
The core innovation of this synthetic pathway lies in the strategic use of the menthyl ester group to control the stereochemical outcome of the oxathiolane ring formation. During the condensation of menthyl glyoxylate with 2,5-dihydroxy-1,4-dithiane, the bulky menthyl moiety exerts significant steric influence, favoring the formation of the trans-isomer which is essential for biological activity. This intrinsic stereocontrol eliminates the need for external chiral catalysts or downstream resolution, thereby streamlining the process flow. The reaction mechanism proceeds through a hemithioacetal intermediate, which cyclizes under acidic conditions to form the five-membered ring. Careful control of temperature, typically maintained between 50°C and 100°C, ensures that the thermodynamic product is favored while minimizing side reactions such as polymerization or decomposition of the sensitive dithiane moiety.
Impurity control is further enhanced during the coupling and reduction phases, where the choice of reagents plays a pivotal role in maintaining product integrity. The coupling of the halogenated oxathiolane intermediate with silylated 5-fluorocytosine is facilitated by Lewis acidic conditions that promote nucleophilic substitution without affecting the fluorine atom on the pyrimidine ring. Following coupling, the reduction of the ester group is performed using mild hydride sources like potassium borohydride at controlled temperatures ranging from 0°C to 40°C. This gentle reduction prevents over-reduction or epimerization at the anomeric center. Finally, the formation of the salicylate salt acts as a crystallization-induced diastereomeric purification, effectively scrubbing minor impurities and ensuring the final free base meets the high-purity OLED material standards often demanded in adjacent high-tech sectors, demonstrating the versatility of this purification logic.
How to Synthesize Emtricitabine Efficiently
Implementing this synthesis requires precise adherence to the reaction parameters defined in the patent to maximize yield and purity. The process involves a sequence of seven distinct chemical transformations, starting from the preparation of menthyl dihaloacetate and concluding with the recrystallization of the free base. Each step demands careful monitoring of stoichiometry, particularly the molar ratios of L-menthol to dihaloacetic acid which are optimized between 1:1 and 1:3 to drive the esterification to completion. Solvent selection is also critical, with non-polar solvents like n-hexane used for esterification and polar aprotic solvents like tetrahydrofuran employed during the reduction phase. Detailed standardized synthesis steps see the guide below.
- Esterify L-menthol with dihaloacetic acid using DCC and DMAP catalysts, followed by hydrolysis to form menthyl glyoxylate.
- Condense menthyl glyoxylate with 2,5-dihydroxy-1,4-dithiane to form the oxathiolane ring, followed by halogenation.
- Couple the halogenated intermediate with silylated 5-fluorocytosine, reduce the ester, and purify via salicylate salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling strategic benefits that extend beyond simple unit cost metrics. By leveraging commodity chemicals like dihaloacetic acid and L-menthol, manufacturers can decouple their production costs from the volatility associated with specialized chiral reagents. This stability in raw material sourcing translates directly into enhanced supply chain reliability, ensuring consistent delivery schedules even during periods of market fluctuation. Furthermore, the simplified purification protocol reduces the consumption of chromatographic media and solvents, contributing to substantial cost savings in waste disposal and environmental compliance. These factors collectively strengthen the resilience of the supply network, making it easier to secure a reliable agrochemical intermediate supplier or pharmaceutical partner who can guarantee continuity.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolution columns and the use of low-cost starting materials significantly lower the overall cost of goods sold. By avoiding complex separation technologies, the process reduces capital expenditure on specialized equipment and lowers operational expenses related to solvent recovery. The high atom economy of the condensation steps ensures that raw materials are utilized efficiently, minimizing waste generation. Additionally, the ability to recycle catalysts and solvents in certain steps further drives down the variable costs associated with production. This economic efficiency allows for more competitive pricing structures without compromising on quality margins.
- Enhanced Supply Chain Reliability: Sourcing L-menthol and dihaloacetic acid is far less risky than procuring proprietary chiral building blocks, as these materials are produced by multiple global vendors. This diversification of the supply base mitigates the risk of single-source failures and ensures that production lines can remain operational even if one supplier faces disruptions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the rate of batch rejections. Consequently, lead times are stabilized, and inventory planning becomes more predictable for downstream partners.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of toxic heavy metals make this process inherently safer and easier to scale from pilot plants to multi-ton reactors. The reduced generation of hazardous waste aligns with increasingly strict environmental regulations, lowering the regulatory burden on manufacturing sites. Efficient solvent usage and the potential for closed-loop recycling systems contribute to a smaller carbon footprint. This sustainability profile is increasingly important for corporate social responsibility goals and can facilitate faster regulatory approvals in environmentally conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. They are derived from the specific beneficial effects and background technical problems identified in the patent documentation. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this route for their specific production needs. The answers provide clarity on scalability, purity, and operational requirements.
Q: What are the primary advantages of using L-menthol in Emtricitabine synthesis?
A: Using L-menthol as a chiral auxiliary eliminates the need for expensive chiral chromatography resolution steps, significantly reducing production costs and simplifying the purification process while maintaining high optical purity.
Q: How does this synthetic route impact environmental compliance?
A: The process utilizes mild reaction conditions and readily available raw materials like dihaloacetic acid, resulting in reduced hazardous waste generation and lower energy consumption compared to traditional high-temperature or high-pressure methods.
Q: Is this method suitable for large-scale commercial manufacturing?
A: Yes, the route is designed for scalability with robust reaction parameters, high yields at each step, and the use of common industrial solvents, making it highly viable for metric-ton scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Emtricitabine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic pathways in delivering high-quality antiviral agents to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our commitment to technical excellence means that we can adapt the novel menthyl ester route to meet your specific volume requirements while maintaining the highest standards of quality and consistency.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this methodology. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your project timelines. Our goal is to serve as a strategic partner, providing not just chemicals but comprehensive solutions that enhance your operational efficiency and market competitiveness.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
