Revolutionizing Nitrogen-Fused Ring Synthesis: A Metal-Free NHC Catalytic Strategy for Commercial Scale-Up
Revolutionizing Nitrogen-Fused Ring Synthesis: A Metal-Free NHC Catalytic Strategy for Commercial Scale-Up
The landscape of organic synthesis for complex heterocyclic scaffolds is undergoing a paradigm shift, driven by the urgent need for greener, more efficient, and economically viable manufacturing processes. Patent CN115260192B introduces a groundbreaking methodology for the preparation of nitrogen-containing fused ring compounds, specifically targeting the challenging synthesis of pyrido[4,3,2-gh]phenanthridinone derivatives. This innovation leverages N-heterocyclic carbene (NHC) organocatalysis to facilitate a radical tandem-cyclization reaction, effectively bypassing the traditional reliance on precious transition metals. For R&D directors and process chemists, this represents a critical advancement, as it addresses the persistent issues of metal residue contamination and harsh reaction conditions that have long plagued the production of high-value pharmaceutical intermediates. The ability to construct these intricate polycyclic architectures under mild conditions opens new avenues for accessing bioactive molecules with anticancer and antiviral properties.
The significance of this technology extends beyond mere academic interest; it directly impacts the commercial feasibility of producing drug candidates that rely on these nitrogen-fused cores. By utilizing alpha-bromo N-aryl acrylamides as readily available starting materials, the process simplifies the supply chain and reduces the dependency on exotic reagents. The patent details a robust protocol that employs cesium carbonate as a base and 1,4-dioxane as a solvent, creating a reaction environment that is both controllable and scalable. This approach not only enhances the overall yield and selectivity but also aligns with the stringent environmental and safety standards required by modern regulatory bodies. As we delve deeper into the technical specifics, it becomes evident that this NHC-catalyzed route offers a superior alternative for the reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of nitrogen-containing fused heterocycles has predominantly relied on transition metal-catalyzed radical tandem-cyclization reactions involving metals such as iridium, ruthenium, or silver. While effective in laboratory settings, these conventional methods present substantial hurdles for industrial application, particularly concerning cost and purity. The use of precious metals inherently drives up the raw material costs, and more critically, the difficulty in completely removing trace metal residues from the final product poses a severe risk for pharmaceutical applications. Regulatory agencies impose strict limits on heavy metal content in Active Pharmaceutical Ingredients (APIs), necessitating additional, expensive purification steps such as scavenging or recrystallization, which inevitably lower the overall process yield. Furthermore, many oxidative coupling protocols require stoichiometric amounts of strong oxidants, generating significant chemical waste and complicating post-reaction treatment, thereby increasing the environmental footprint and disposal costs for manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN115260192B utilizes an organic small molecule, specifically an N-heterocyclic carbene (NHC), as the catalyst, fundamentally altering the economic and operational dynamics of the synthesis. This metal-free approach operates under remarkably mild conditions, often at room temperature, which drastically reduces energy consumption compared to high-temperature thermal processes. The elimination of transition metals means that the resulting products are free from heavy metal contamination, significantly simplifying the downstream purification process and ensuring compliance with rigorous quality specifications without the need for specialized metal removal resins. Moreover, the reaction demonstrates excellent chemical and regioselectivity, minimizing the formation of by-products and isomers that would otherwise complicate isolation. This novel strategy provides a green, efficient, and highly selective pathway that is ideally suited for the commercial scale-up of complex pharmaceutical intermediates.
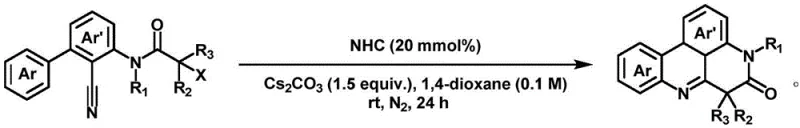
Mechanistic Insights into NHC-Catalyzed Radical Tandem Cyclization
The core of this technological breakthrough lies in the unique mechanistic pathway facilitated by the N-heterocyclic carbene catalyst. Unlike traditional transition metal cycles that involve oxidative addition and reductive elimination steps, the NHC catalyst activates the alpha-bromo N-aryl acrylamide substrate through a single-electron transfer (SET) or radical generation mechanism. This activation initiates a cascade of intramolecular cyclization events, where the generated radical species attacks the proximal aromatic or olefinic moiety to form the new carbon-carbon bonds required for the fused ring system. The precise electronic tuning of the NHC ligand allows for fine control over the reactivity of the intermediate radicals, ensuring that the cyclization proceeds with high fidelity. This mechanistic understanding is crucial for process optimization, as it explains why the reaction tolerates a wide range of functional groups, including electron-donating methoxy groups and electron-withdrawing cyano or chloro substituents, without compromising the integrity of the final scaffold.
From an impurity control perspective, the high selectivity of the NHC catalytic cycle is a major advantage. In conventional radical reactions, uncontrolled radical propagation often leads to polymerization or non-specific coupling, resulting in complex impurity profiles that are difficult to characterize and remove. However, the confined catalytic environment provided by the NHC species directs the radical intermediate specifically towards the desired intramolecular cyclization pathway. This inherent selectivity minimizes the generation of side products, leading to cleaner reaction mixtures and higher crude purity. For quality control teams, this translates to more consistent batch-to-batch reproducibility and reduced analytical burden. The ability to predictably manage the reaction trajectory ensures that the final nitrogen-containing fused ring compounds meet the stringent purity specifications required for subsequent biological testing and clinical development, thereby accelerating the overall drug discovery timeline.
How to Synthesize Nitrogen-Containing Fused Ring Compounds Efficiently
The practical implementation of this synthesis involves a straightforward procedure that can be easily adapted for both laboratory screening and pilot-scale production. The process begins with the careful selection of the alpha-bromo N-aryl acrylamide precursor, which dictates the substitution pattern of the final fused ring system. The reaction is typically conducted in a sealed vessel under an inert nitrogen atmosphere to prevent the quenching of radical intermediates by oxygen. The standard protocol involves mixing the substrate with cesium carbonate and the NHC catalyst in 1,4-dioxane, followed by stirring at ambient temperature or slightly elevated temperatures depending on the substrate's reactivity. Detailed standardized synthetic steps for optimizing yield and purity are provided in the guide below, ensuring that technical teams can replicate the results with high precision.
- Charge a reaction vessel with alpha-bromo N-aryl acrylamide substrate, cesium carbonate base, and NHC catalyst under inert atmosphere.
- Add 1,4-dioxane solvent to achieve the specified concentration and stir the mixture at room temperature or mild heating.
- Monitor reaction progress via TLC, then purify the crude product through column chromatography to isolate the target fused ring compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this NHC-catalyzed technology offers transformative benefits that extend well beyond the laboratory bench. The primary value proposition lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and enhanced supply chain resilience. By removing the dependency on volatile and expensive transition metal markets, manufacturers can stabilize their raw material costs and mitigate the risks associated with supply disruptions of critical catalysts. Furthermore, the mild reaction conditions reduce the energy load on production facilities, contributing to lower utility costs and a smaller carbon footprint, which is increasingly important for meeting corporate sustainability goals. The streamlined purification process also means faster turnaround times from synthesis to final product release, allowing companies to respond more agilely to market demands.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts such as iridium or ruthenium removes a significant cost driver from the bill of materials, while simultaneously erasing the expense associated with specialized metal scavenging resins and extensive purification protocols. This dual saving mechanism results in substantial cost optimization for the production of high-purity pharmaceutical intermediates, making the final API more competitive in the global market. Additionally, the use of common solvents like 1,4-dioxane and inexpensive bases like cesium carbonate further lowers the input costs, ensuring that the process remains economically viable even at large scales.
- Enhanced Supply Chain Reliability: Relying on organic small molecule catalysts rather than scarce transition metals diversifies the supply base and reduces vulnerability to geopolitical fluctuations affecting metal mining and refining. The robustness of the reaction conditions, which tolerate a wide variety of substrates and functional groups, ensures consistent production output even when slight variations in raw material quality occur. This reliability is critical for maintaining continuous supply lines for key drug substances, preventing costly production stoppages and ensuring that downstream customers receive their orders on time without compromise.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, having been demonstrated to work efficiently from milligram to gram scales with the potential for tonnage production. The absence of heavy metals and strong oxidants simplifies waste treatment procedures, reducing the environmental burden and ensuring compliance with increasingly strict environmental regulations. This green chemistry profile not only facilitates easier regulatory approval but also enhances the brand reputation of the manufacturer as a responsible and sustainable partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the NHC-catalyzed synthesis of nitrogen-containing fused ring compounds. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, providing clarity for stakeholders evaluating this technology for integration into their existing pipelines. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation.
Q: What are the key advantages of using NHC catalysis over transition metals for this synthesis?
A: The NHC catalytic method eliminates the need for expensive and toxic transition metals like iridium or ruthenium, thereby removing the requirement for costly heavy metal removal steps and ensuring higher purity suitable for pharmaceutical applications.
Q: What are the typical reaction conditions described in patent CN115260192B?
A: The reaction typically proceeds in 1,4-dioxane solvent using cesium carbonate as a base, often at room temperature or mild heating (e.g., 50°C), under a nitrogen atmosphere for approximately 24 hours.
Q: Is this method scalable for industrial production of API intermediates?
A: Yes, the patent explicitly states the method is capable of being amplified to gram scale and beyond, featuring simple operation and mild conditions that facilitate commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrogen-Containing Fused Ring Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the NHC-catalyzed synthesis described in patent CN115260192B and are fully equipped to leverage this technology for our global partners. As a premier CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless and efficient. Our state-of-the-art facilities are staffed by seasoned chemists who specialize in complex organic synthesis, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of nitrogen-containing fused ring compounds meets the highest industry standards. We are committed to delivering excellence in every aspect of our service, from initial route scouting to final commercial manufacturing.
We invite you to collaborate with us to unlock the full commercial potential of this innovative synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating exactly how this metal-free approach can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why NINGBO INNO PHARMCHEM is the preferred partner for cost reduction in pharmaceutical intermediate manufacturing and reliable supply chain solutions.
