Advanced Synthetic Route for Fondaparinux Sodium: Enhancing Purity and Scalability for Global Pharma
Advanced Synthetic Route for Fondaparinux Sodium: Enhancing Purity and Scalability for Global Pharma
The pharmaceutical landscape for anticoagulants has been revolutionized by the development of synthetic heparin pentasaccharides, specifically Fondaparinux Sodium, which offers a safer alternative to animal-derived heparins by eliminating the risk of cross-species viral contamination. Patent CN103360439A introduces a groundbreaking methodology for preparing key intermediates in the synthesis of this critical active pharmaceutical ingredient (API). This technology addresses long-standing challenges in carbohydrate chemistry, particularly regarding the stability of reducing ends and the complexity of selective sulfation. By implementing a methyl-blocking strategy at the reducing end and reordering the sequence of reduction and sulfation reactions, this invention provides a robust pathway that is highly suitable for commercial-scale manufacturing. The technical breakthroughs detailed in this patent not only enhance the chemical efficiency but also streamline the downstream processing required to meet stringent regulatory standards for parenteral medications.
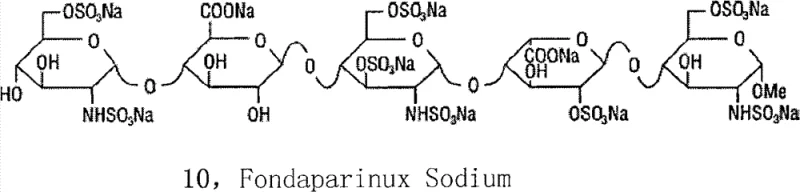
For procurement managers and supply chain directors, the implications of this synthetic route are profound. Traditional methods often suffer from low yields due to side reactions involving the reducing end aldehyde, which can polymerize into dimers or trimers, complicating purification and driving up costs. The novel approach described in CN103360439A mitigates these risks by stabilizing the molecular architecture early in the synthesis. This stability translates directly into a more predictable supply chain for high-purity fondaparinux sodium, reducing the likelihood of batch failures and ensuring consistent availability of this life-saving medication. As a reliable heparin pentasaccharide supplier, understanding these underlying process improvements is essential for evaluating the long-term viability and cost-effectiveness of sourcing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of heparin pentasaccharides has been plagued by significant chemical hurdles, particularly during the final deprotection and functionalization stages. Conventional routes, such as those pioneered by Sinay and Boeckel, typically involve the simultaneous removal of benzyl protecting groups and the reduction of azido or carbobenzoxy-protected amines. A critical flaw in these legacy processes is the instability of the reducing end; once the protecting groups are removed, the free aldehyde at the reducing end can undergo hemiacetal tautomerization. This reactive species readily engages in side reactions with other pentasaccharide molecules, leading to the formation of stable dimers or trimers. These oligomeric byproducts are structurally similar to the target molecule, making them extremely difficult to separate via standard chromatographic techniques. Consequently, the overall reaction efficiency is severely compromised, and the purification of the end product becomes a bottleneck that limits throughput and increases manufacturing costs substantially.
The Novel Approach
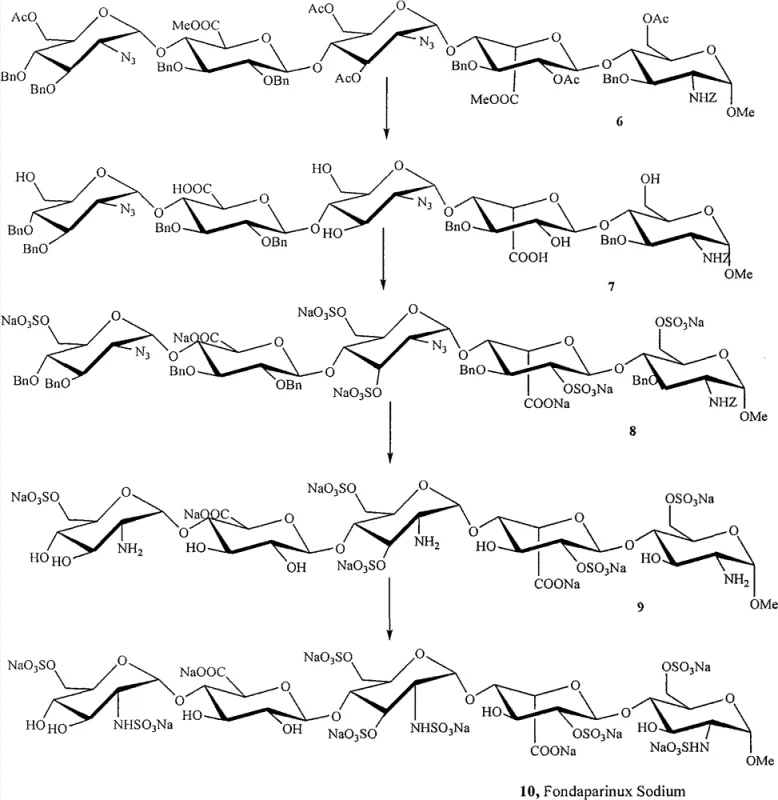
The methodology disclosed in patent CN103360439A fundamentally reengineers the synthetic sequence to bypass these thermodynamic traps. By employing a methyl-blocking strategy at the reducing end, the formation of the reactive aldehyde is prevented entirely throughout the intermediate stages. This modification ensures that the pentasaccharide backbone remains stable and inert to polymerization, allowing for high-yield transformations. Furthermore, the invention optimizes the order of operations by performing the reduction of azido groups to exposed amines prior to the final global sulfation and hydrogenation steps. This specific sequencing allows for the generation of intermediates with higher crude purity, which can often proceed to the next reaction step without rigorous intermediate purification. The result is a streamlined process that drastically simplifies the operational workflow, reduces solvent consumption, and enhances the overall mass balance of the synthesis, making it ideally suited for cost reduction in anticoagulant manufacturing.
Mechanistic Insights into Phosphine-Mediated Azido Reduction and Sulfation
The core chemical innovation in this patent lies in the efficient conversion of azido groups to amino groups using phosphine-based reducing agents within a complex oligosaccharide framework. Unlike catalytic hydrogenation which might prematurely cleave benzyl ethers or affect other sensitive functionalities, the use of reagents such as trimethylphosphine or triphenylphosphine offers exceptional chemoselectivity. In the presence of water or aqueous sodium hydroxide, these phosphines facilitate the Staudinger reduction mechanism, converting the azido functionality into an iminophosphorane intermediate which is subsequently hydrolyzed to the primary amine. This reaction proceeds rapidly and quantitatively under mild conditions, preserving the integrity of the glycosidic linkages and the ester protecting groups elsewhere on the molecule. The ability to perform this reduction on a fully protected or partially deprotected scaffold without generating significant impurities is a testament to the robustness of the chosen reagents and conditions.
Following the reduction, the process employs a strategic sulfation protocol to install the critical sulfate esters and sulfamates required for biological activity. The patent describes the use of sulfur trioxide complexes, such as sulfur trioxide-pyridine or sulfur trioxide-trimethylamine, in polar aprotic solvents like DMF or pyridine. The mechanistic advantage here is the ability to sulfate both hydroxyl and amino groups in a controlled manner. By adjusting the stoichiometry and reaction temperature, specifically utilizing a stepwise approach where bulk sulfation is followed by a finishing step with excess reagent, the process ensures complete conversion of all target sites. This comprehensive sulfation is crucial because incomplete sulfation leads to analogues with reduced anticoagulant potency. The retention of benzyl groups during this stage further aids in solubility and handling, preventing the premature precipitation of highly charged polyanionic species that can occur if deprotection happens too early.
How to Synthesize Fondaparinux Sodium Efficiently
The synthesis of Fondaparinux Sodium via this novel intermediate pathway involves a logical progression of deprotection, functionalization, and final refinement steps designed to maximize yield and purity. The process begins with the hydrolysis of ester groups on a fully protected pentasaccharide precursor, followed by the selective reduction of azido groups to amines. Subsequent sulfation introduces the necessary anionic charge density, and the final catalytic hydrogenation removes the lipophilic benzyl protecting groups to reveal the bioactive molecule. This sequence minimizes side reactions and simplifies isolation procedures at each stage. For detailed laboratory protocols and specific reaction conditions tailored to your facility's capabilities, please refer to the standardized synthesis guide below.
- Perform ester hydrolysis on the fully protected pentasaccharide compound using sodium hydroxide to expose hydroxyl groups.
- Reduce azido groups to exposed amino groups using phosphine-based reducing agents such as trimethylphosphine or triphenylphosphine.
- Conduct sulfation reactions on all exposed hydroxyl and amino groups using sulfur trioxide complexes to introduce sulfate esters.
- Execute final catalytic hydrogenation using palladium catalysts to remove benzyl protecting groups and yield the final sodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of the synthetic route described in CN103360439A offers substantial advantages for organizations managing the supply of complex pharmaceutical intermediates. The primary value driver is the significant enhancement in process reliability, which directly correlates to supply chain security. By eliminating the formation of difficult-to-remove dimeric impurities, the manufacturing process becomes more predictable, reducing the risk of batch rejections that can disrupt inventory levels. Furthermore, the ability to carry forward crude intermediates without extensive purification between steps reduces the consumption of chromatography media and solvents, leading to a leaner and more cost-effective production model. This efficiency is critical for maintaining competitive pricing in the global market for anticoagulants while adhering to strict quality standards.
- Cost Reduction in Manufacturing: The novel synthetic strategy eliminates the need for expensive and time-consuming purification steps between the reduction and sulfation stages. Because the intermediates generated possess higher crude purity and stability, manufacturers can operate with reduced solvent volumes and lower consumption of silica gel or resin materials. Additionally, the use of phosphine reducing agents avoids the need for precious metal catalysts in the early stages, further lowering the raw material costs. The overall simplification of the workflow means less labor hours are required per kilogram of output, driving down the operational expenditure significantly without compromising on the quality of the final API.
- Enhanced Supply Chain Reliability: The stability of the methyl-blocked intermediates ensures that the synthesis is less susceptible to environmental variables such as moisture or slight temperature fluctuations that often plague carbohydrate chemistry. This robustness translates to higher success rates in large-scale reactors, ensuring that production schedules are met consistently. For supply chain heads, this means a more dependable source of high-purity fondaparinux sodium, reducing the need for safety stock and minimizing the risk of shortages. The scalability of the process allows for seamless transition from pilot plant to commercial production, securing the long-term availability of this critical therapeutic agent.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing reagents and conditions that are manageable in large vessels. The reduction in solvent usage and the elimination of heavy metal catalysts in certain steps contribute to a greener manufacturing profile, aligning with modern environmental regulations and corporate sustainability goals. Waste streams are easier to manage due to the higher purity of intermediates, reducing the burden on effluent treatment plants. This environmental compliance not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible producer of specialty chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the practical aspects of producing heparin pentasaccharides. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: Why is methyl blocking employed at the reducing end in this synthesis?
A: Methyl blocking prevents the formation of unstable hemiacetal tautomers at the reducing end, which traditionally react to form unwanted dimers or trimers, thereby significantly improving reaction efficiency and yield.
Q: What reducing agents are compatible with this novel intermediate pathway?
A: The process utilizes phosphine-based reducing agents such as trimethylphosphine, triethylphosphine, tributylphosphine, or triphenylphosphine, which efficiently reduce azido groups to amino groups under mild conditions.
Q: How does this method improve purification compared to conventional routes?
A: By retaining benzyl protection until the final stages and optimizing the order of reduction and sulfation, the intermediates exhibit higher crude purity and stability, eliminating the need for complex purification steps between reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fondaparinux Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving anticoagulants. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in patent CN103360439A can be translated into reality with precision. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and potency of every batch. Our commitment to excellence ensures that the fondaparinux sodium intermediates we supply meet the exacting standards required by global regulatory bodies, providing our partners with peace of mind and a competitive edge in the marketplace.
We invite you to collaborate with us to optimize your supply chain for heparin pentasaccharides. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and production timelines. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your growth in the pharmaceutical sector.
