Optimizing Moxifloxacin Side Chain Production: A Technical Breakthrough in Chiral Resolution
The pharmaceutical industry continuously seeks robust methodologies for synthesizing high-purity chiral intermediates, particularly for fourth-generation fluoroquinolone antibiotics like Moxifloxacin Hydrochloride. Patent CN112707901A, published in April 2021, introduces a transformative preparation method for Compound A, chemically identified as (S, R)-8-benzyl-7, 9-dioxo-2, 8-diazabicyclo [4.3.0] nonane. This compound serves as a pivotal chiral precursor in the synthesis of the Moxifloxacin side chain, (S, S)-2, 8-diazabicyclo [4.3.0] nonane. The disclosed innovation addresses long-standing inefficiencies in chiral resolution by employing gulonic acid as a novel resolving agent, achieving a single-resolution yield exceeding 40% and an enantiomeric excess (ee) value greater than 98%. This technical advancement represents a significant leap forward for manufacturers aiming to optimize the production of broad-spectrum antibacterial agents while maintaining stringent quality standards.
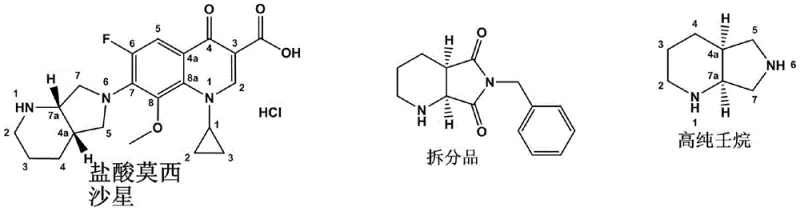
The synthesis of Moxifloxacin relies heavily on the availability of high-purity chiral building blocks. The target molecule, (S, R)-8-benzyl-7, 9-dioxo-2, 8-diazabicyclo [4.3.0] nonane, is typically derived from hydrogenation products which must be resolved to isolate the specific stereoisomer required for biological activity. Traditional pathways often struggle with low efficiency and high costs, creating bottlenecks in the supply chain for this critical antibiotic. The new method detailed in the patent simplifies this landscape by integrating a highly effective salification and crystallization protocol that maximizes both yield and optical purity in a single operational sequence.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of (S, R)-8-benzyl-7, 9-dioxo-2, 8-diazabicyclo [4.3.0] nonane has relied on resolving agents such as D(-)-tartaric acid, L(+)-tartaric acid, or N-acetyl-L-leucine. While these agents can facilitate separation, they are plagued by significant economic and technical drawbacks. For instance, N-acetyl-L-leucine is characterized by a high market price and instability under acidic conditions, necessitating careful handling and increasing raw material costs. Similarly, tartaric acid-based resolutions often suffer from low single-pass yields, typically falling below 30%, which forces manufacturers to perform multiple resolution cycles to achieve the necessary chiral purity. This multi-step approach not only consumes excessive amounts of starting materials and resolving agents but also extends production timelines and generates substantial chemical waste. Furthermore, the theoretical maximum yield for a standard resolution is 50%, and achieving even 70% of this theoretical limit (35% actual yield) is considered difficult with older methods, leading to poor atom economy and inflated production costs.
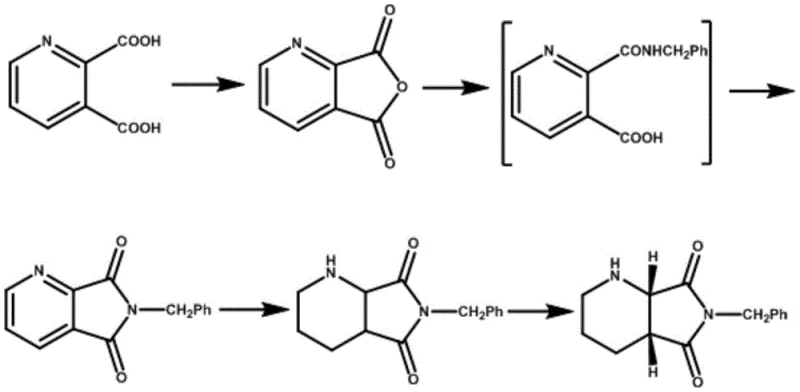
The Novel Approach
In stark contrast to these legacy processes, the method disclosed in CN112707901A utilizes gulonic acid as the resolving agent, offering a paradigm shift in process efficiency. Gulonic acid is a conventional, commercially available commodity chemical that is significantly cheaper and more stable than specialized amino acid derivatives. The novel protocol involves dissolving the hydrogenated racemic product in a specifically engineered mixed solvent system, adding gulonic acid, and controlling the temperature to precipitate the desired diastereomeric salt. This approach consistently delivers a single-resolution yield of over 40%, surpassing the typical performance of tartaric acid methods. Moreover, the resulting product exhibits an ee value exceeding 98%, eliminating the need for repetitive recrystallization steps. By streamlining the resolution into a single, high-yield event, this method drastically reduces the complexity of the manufacturing process, lowers solvent consumption, and enhances the overall throughput of the facility.
Mechanistic Insights into Gulonic Acid-Mediated Chiral Resolution
The success of this resolution strategy lies in the precise interplay between the resolving agent, the solvent system, and the thermodynamic properties of the diastereomeric salts. Gulonic acid forms a salt with the (S, R)-isomer that possesses distinct solubility characteristics compared to the (R, S)-isomer salt in the chosen solvent matrix. The patent highlights the critical importance of the solvent composition, specifically an aqueous mixed solvent containing C1-4 alcohols (such as ethanol) and ketones (such as butanone). The inclusion of water, maintained at a volume fraction of 5% to 15% (optimally 10%), plays a pivotal role in modulating solubility. Water increases the solubility of the unwanted racemic components in the mother liquor, thereby driving the equilibrium towards the crystallization of the pure target salt. If the water content is too low, the chiral purity drops due to co-crystallization of impurities; if too high, the yield suffers as the product remains dissolved. This delicate balance ensures that the crystal lattice formed is highly selective for the desired stereoisomer.
Furthermore, the thermal profile of the reaction is meticulously controlled to maximize crystal growth and purity. The process involves heating the mixture to 50-80°C to ensure complete dissolution and salt formation, followed by a controlled cooling phase to 20-30°C. This temperature gradient facilitates the nucleation and growth of large, pure crystals of the resolving salt, which can be easily filtered and washed. The subsequent alkaline treatment (pH 8-9) effectively strips the gulonic acid from the salt, releasing the free base (S, R)-8-benzyl-7, 9-dioxo-2, 8-diazabicyclo [4.3.0] nonane without racemization. This mechanistic robustness ensures that the high optical purity achieved during crystallization is preserved in the final isolated product, making the process highly reliable for industrial scale-up.
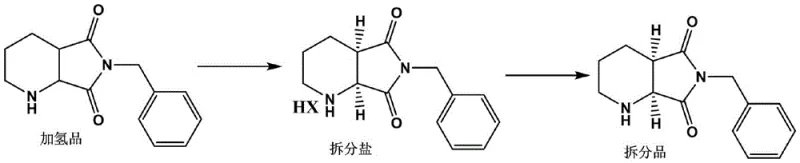
How to Synthesize (S,R)-8-benzyl-7,9-dioxo-2,8-diazabicyclo[4.3.0]nonane Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this high-efficiency resolution in a production environment. The process begins with the preparation of the resolving salt by dissolving the hydrogenated precursor in a mixed solvent of ethanol, butanone, and water, followed by the addition of gulonic acid at a molar ratio of 0.5 to 1.0 relative to the substrate. The mixture is heated to promote salt formation and then cooled to induce crystallization. After filtration and washing, the solid salt undergoes alkaline hydrolysis to release the target chiral intermediate. This straightforward two-step sequence—salification followed by stripping—minimizes unit operations and reduces the potential for yield loss. For a comprehensive understanding of the specific parameters, including exact solvent ratios and temperature ramps, please refer to the detailed standardized synthesis steps provided in the guide below.
- Dissolve the hydrogenated racemic product in a mixed solvent system (e.g., ethanol/butanone/water) and add gulonic acid as the resolving agent.
- Heat the mixture to 50-80°C for 1-2 hours, then cool to 20-30°C to induce crystallization of the diastereomeric salt.
- Filter the resolving salt, wash, and treat with alkali (pH 8-9) followed by organic extraction to isolate the pure chiral intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this gulonic acid-based resolution method offers tangible benefits that extend beyond mere technical feasibility. The primary advantage is the substantial reduction in raw material costs. By replacing expensive, specialty resolving agents like N-acetyl-L-leucine with gulonic acid—a bulk chemical with a stable and low market price—the direct material cost of the intermediate is significantly lowered. Additionally, the improved single-pass yield (>40% vs <30%) means that less starting material is required to produce the same amount of final product, further enhancing cost efficiency. This reduction in material intensity translates directly to improved margins and a more competitive pricing structure for the final API.
- Cost Reduction in Manufacturing: The elimination of multiple resolution cycles is a major driver for cost savings. Traditional methods often require recycling mother liquors or performing second resolutions to boost ee values, which consumes additional solvents, energy, and labor. The new method achieves >98% ee in a single step, effectively halving the processing time and resource consumption associated with purification. Furthermore, the use of common solvents like ethanol and butanone, rather than exotic or hazardous chemicals, simplifies solvent recovery and waste treatment protocols, leading to lower operational expenditures and reduced environmental compliance costs.
- Enhanced Supply Chain Reliability: Reliance on scarce or volatile resolving agents poses a risk to supply continuity. Gulonic acid is a widely available commodity, ensuring a stable supply chain that is less susceptible to market fluctuations or vendor shortages. The robustness of the crystallization process also means that production schedules are more predictable, with fewer batches rejected due to failure to meet chiral purity specifications. This reliability allows for tighter inventory management and shorter lead times for delivering high-purity pharmaceutical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The simplicity of the process—dissolve, heat, cool, filter, strip—makes it inherently scalable from pilot plant to commercial tonnage production. The absence of complex chromatographic separations or hazardous reagents simplifies equipment requirements and safety protocols. Moreover, the higher yield and reduced solvent usage contribute to a smaller environmental footprint, aligning with modern green chemistry principles and facilitating easier regulatory approval for the manufacturing process. This scalability ensures that the method can meet growing global demand for Moxifloxacin without requiring massive capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These answers are derived directly from the experimental data and comparative analysis presented in patent CN112707901A, providing clarity on why this method represents a superior choice for modern pharmaceutical manufacturing. Understanding these nuances is essential for R&D teams evaluating process transfers and procurement teams assessing supplier capabilities.
Q: Why is gulonic acid preferred over tartaric acid for this resolution?
A: Gulonic acid offers significant cost advantages as a conventional market commodity compared to expensive resolving agents like N-acetyl-L-leucine or specialized tartaric acid derivatives. Furthermore, it enables a single-step resolution with yields exceeding 40% and ee values >98%, whereas traditional methods often require multiple resolution cycles to achieve similar purity.
Q: What is the critical role of water in the solvent system?
A: Water content (optimized at 5-15%) is crucial for balancing solubility and chiral purity. It increases the solubility of the unwanted racemic salt in the mother liquor, thereby enhancing the chiral purity of the crystallized product, while preventing yield loss associated with overly polar solvent systems.
Q: How does this process impact the overall cost of Moxifloxacin production?
A: By utilizing a low-cost resolving agent and achieving higher single-pass yields (>40% vs <30% in prior art), the process drastically reduces raw material consumption and processing time. The elimination of complex multi-step resolution procedures further lowers operational expenditures and waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S,R)-8-benzyl-7,9-dioxo-2,8-diazabicyclo[4.3.0]nonane Supplier
The technical potential of the gulonic acid resolution method underscores the importance of partnering with a manufacturer who possesses deep expertise in chiral chemistry and process optimization. NINGBO INNO PHARMCHEM stands at the forefront of this field, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the precise solvent engineering and temperature control required for this high-purity synthesis, ensuring that every batch meets stringent purity specifications. With rigorous QC labs and a commitment to continuous improvement, we guarantee the consistent delivery of high-quality intermediates that comply with international regulatory standards.
We invite pharmaceutical partners to leverage our technical capabilities to optimize their supply chains for Moxifloxacin and related antibiotics. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples that demonstrate the superior quality of our chiral intermediates. Let us help you secure a reliable, cost-effective supply of critical building blocks for your next-generation therapeutic formulations.
