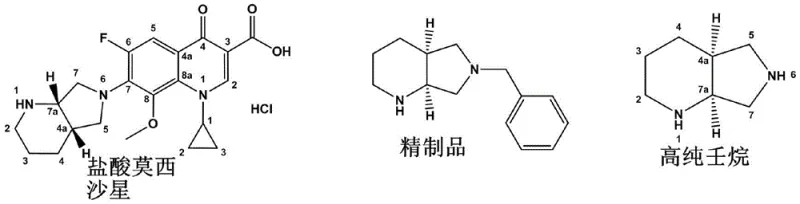
Advanced Chiral Purification Technology for Moxifloxacin Intermediates and Commercial Scale-Up
The pharmaceutical industry continuously seeks robust methodologies to enhance the purity and yield of critical antibiotic intermediates, particularly for fourth-generation fluoroquinolones like Moxifloxacin Hydrochloride. Patent CN112724129A introduces a groundbreaking chiral purification method for Compound B, specifically (S, R)-8-benzyl-7, 9-dioxo-2, 8-diazabicyclo [4.3.0] nonane, which serves as a pivotal precursor in the synthesis of this broad-spectrum antibacterial agent. The technical breakthrough lies in replacing traditional, inefficient optical resolution techniques with a streamlined hydrohalic acid salt crystallization process. This innovation addresses the chronic industry pain points of low recovery rates and excessive waste generation associated with conventional resolving agents. By leveraging specific solvent systems and precise pH control, the method ensures that the chiral integrity of the molecule is preserved while drastically improving the overall process mass intensity. For R&D directors and supply chain leaders, this represents a significant opportunity to optimize the manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
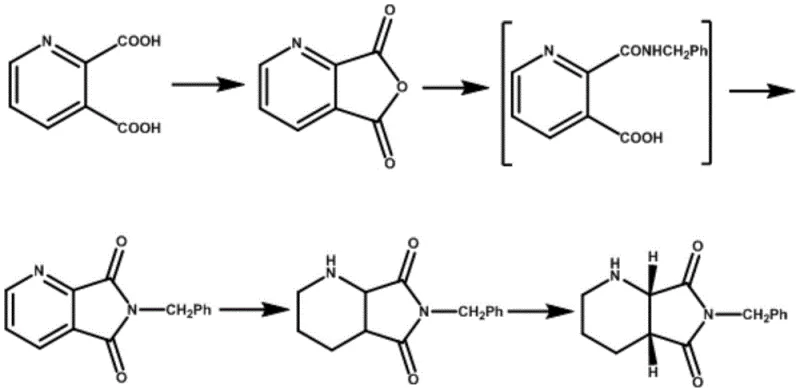
Historically, the synthesis of key chiral intermediates for quinolone antibiotics has relied heavily on resolution techniques using expensive optical resolving agents such as D(-)-tartaric acid or L(+)-tartaric acid. These conventional pathways, as documented in various literature patents including WO2009/125425 and US6235908, typically start from 2,3-pyridinedicarboxylic acid derivatives and involve multiple hydrogenation and imidization steps before reaching the resolution stage. The fundamental flaw in these legacy processes is the inherent inefficiency of the resolution step itself, which often yields less than 30% of the desired enantiomer. Furthermore, achieving the requisite chiral purity of greater than 98% ee frequently necessitates a secondary resolution, which compounds the material loss and increases the operational complexity. The reliance on specific chiral acids also introduces supply chain vulnerabilities and increases the cost of goods sold due to the high price of these resolving agents. Additionally, the generation of waste salts from the resolving agents creates significant environmental disposal challenges, complicating regulatory compliance for large-scale manufacturing facilities.
The Novel Approach
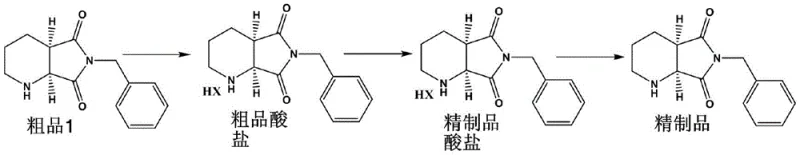
In stark contrast to the cumbersome traditional methods, the novel approach detailed in the patent utilizes a hydrohalic acid salt formation strategy that bypasses the need for chiral resolving agents entirely. This method takes a crude product with an initial ee value ranging from 90% to 98% and subjects it to a controlled salification process using hydrochloric or hydrobromic acid. The resulting acid salt is then purified through a sophisticated recrystallization process in specific alcohol or ketone solvent systems, such as ethanol, acetone, or butanone. This physical purification mechanism leverages the solubility differences between the desired enantiomer salt and impurities, allowing for the isolation of a highly refined acid salt with exceptional purity. Subsequent alkaline hydrolysis and extraction release the free base, yielding the final product with an ee value exceeding 99.5%. This shift from chemical resolution to physical purification via salt crystallization not only simplifies the operational workflow but also dramatically enhances the material throughput, making it a superior choice for commercial production environments.
Mechanistic Insights into Hydrohalic Acid Salt Crystallization
The core mechanism driving the success of this purification technology is the selective crystallization behavior of the hydrohalic acid salt of the target intermediate. When the crude base reacts with a hydrohalic acid, such as hydrochloric acid or hydrobromic acid, it forms a salt that exhibits distinct solubility characteristics compared to the impurities and the minor enantiomer. The patent specifies that controlling the reaction temperature between 55°C and 80°C during the reflux refining stage is critical to ensuring complete dissolution and subsequent uniform nucleation upon cooling. The choice of solvent system plays a pivotal role; for instance, a mixed solvent of alcohol and ketone with a specific volume fraction of alcohol (5%-15%) optimizes the solubility profile to favor the precipitation of the high-purity salt. This precise control over the thermodynamic conditions allows the crystal lattice to exclude impurities effectively, resulting in a refined acid salt that is chemically and stereochemically superior to the starting material. The mechanism avoids the formation of complex diastereomeric salts typical of tartaric acid resolution, thereby eliminating the need for intricate separation techniques.
Impurity control is further enhanced during the alkaline hydrolysis and extraction phase, which serves as a final polishing step for the product. By adjusting the pH to a mildly alkaline range of 8-9 using bases like sodium bicarbonate or sodium hydroxide, the free base is regenerated without inducing racemization or degradation of the sensitive bicyclic structure. The use of water-immiscible solvents like toluene for extraction ensures that water-soluble inorganic salts and residual acid are effectively partitioned away from the organic phase containing the product. This liquid-liquid extraction step acts as a secondary purification barrier, removing any remaining trace impurities that might have co-crystallized during the salt formation stage. The result is a final product that not only meets the stringent chiral purity specifications of greater than 99.5% ee but also possesses a clean impurity profile suitable for downstream pharmaceutical synthesis. This dual-stage purification mechanism ensures robust quality control and consistency across different production batches.
How to Synthesize (S, R)-8-benzyl-7, 9-dioxo-2, 8-diazabicyclo[4.3.0]nonane Efficiently
The implementation of this synthesis route requires careful attention to solvent selection and temperature control to maximize the benefits of the patent technology. The process begins with the conversion of the crude intermediate into its hydrohalic acid salt form, followed by a thermal refining step in a selected organic solvent. Detailed operational parameters, including specific solvent ratios and cooling rates, are essential to replicate the high yields and purity reported in the patent examples. Operators must ensure that the pH is strictly maintained during the salification endpoint to prevent the formation of excessive waste salts or incomplete reaction. The subsequent stripping of the alkaloid requires precise base addition to avoid local over-basicity which could compromise the product integrity. For a comprehensive guide on the exact procedural steps and safety considerations, please refer to the standardized protocol below.
- Preparation of Acid Salt: Dissolve crude compound in solvent A, add hydrohalic acid to form crude salt, evaporate solvent, then reflux in solvent B for purification.
- Crystallization: Cool the refined acid salt solution to room temperature to induce crystallization of the high-purity hydrohalic acid salt.
- Stripping Alkaloid: Perform alkaline hydrolysis on the refined salt, extract with organic solvent C, and evaporate to obtain the final high-purity chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this chiral purification method offers substantial strategic advantages for procurement managers and supply chain heads responsible for sourcing high-purity pharmaceutical intermediates. The elimination of expensive optical resolving agents like tartaric acid directly translates to a significant reduction in raw material costs, as the process relies on commodity chemicals such as hydrochloric acid and common organic solvents. This shift simplifies the procurement portfolio, reducing the risk associated with sourcing specialized chiral reagents that may have limited supplier availability. Furthermore, the drastic improvement in refining yield, which exceeds 90% compared to the sub-30% yields of traditional methods, means that less starting material is required to produce the same amount of final product. This efficiency gain lowers the overall cost of goods sold and reduces the volume of chemical waste that needs to be treated and disposed of, aligning with modern environmental sustainability goals and reducing regulatory compliance costs.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of the secondary resolution step and the associated expensive resolving agents. By achieving high purity through crystallization of hydrohalic acid salts, manufacturers avoid the material losses inherent in diastereomeric salt resolution. This leads to a more efficient use of raw materials and a reduction in the overall processing time, as fewer unit operations are required to achieve the target specification. The use of recoverable solvents like toluene and ethanol further enhances the economic viability of the process, allowing for solvent recycling and minimizing waste disposal expenses. Consequently, the total manufacturing cost per kilogram of the intermediate is significantly lowered, providing a competitive edge in the global market for moxifloxacin precursors.
- Enhanced Supply Chain Reliability: Supply chain stability is greatly improved by relying on widely available commodity chemicals rather than specialized chiral resolving agents. Hydrochloric acid, hydrobromic acid, and solvents like acetone and ethanol are standard industrial chemicals with robust global supply networks, minimizing the risk of supply disruptions. The simplicity of the process also reduces the dependency on highly specialized technical expertise for operation, making it easier to transfer technology between manufacturing sites or qualify alternative contract manufacturing organizations. This flexibility ensures a continuous and reliable supply of the critical intermediate, which is essential for maintaining the production schedules of the final antibiotic drug. The robustness of the process against minor variations in raw material quality further contributes to supply chain resilience.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scale-up, with parameters that are easily controllable in large-scale reactors. The avoidance of complex chromatographic separations or sensitive enzymatic reactions makes the technology highly scalable from pilot plant to commercial production volumes. From an environmental standpoint, the reduction in chemical waste, particularly the absence of large quantities of tartaric acid salts, simplifies wastewater treatment and solid waste management. This aligns with increasingly stringent environmental regulations in the pharmaceutical industry, reducing the risk of compliance violations and associated fines. The greener profile of the manufacturing process also enhances the corporate social responsibility standing of the supply chain, appealing to end-users who prioritize sustainable sourcing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral purification technology. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing manufacturing workflows. The information provided here aims to clarify the operational advantages and quality outcomes associated with the hydrohalic acid salt purification method.
Q: How does this purification method improve upon traditional tartaric acid resolution?
A: Traditional methods using optical resolving agents like tartaric acid often suffer from low yields (typically under 30%) and require secondary resolution steps. This novel hydrohalic acid salt method achieves refining yields exceeding 90% and eliminates the need for expensive resolving agents, significantly simplifying the workflow.
Q: What chiral purity levels can be achieved with this process?
A: The process is designed to upgrade crude material with an ee value of 90%-98% to a refined product with an ee value greater than 99.5%. This high level of stereochemical purity is critical for meeting the stringent quality specifications required for fourth-generation quinolone antibiotics.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly states the process is easy to amplify and suitable for industrial production. It utilizes common solvents like ethanol, acetone, and toluene, and avoids complex chromatographic separations, making it highly robust for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S, R)-8-benzyl-7, 9-dioxo-2, 8-diazabicyclo[4.3.0]nonane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the production of life-saving antibiotics like Moxifloxacin. Our technical team has extensively analyzed the potential of the purification route described in CN112724129A and is fully equipped to implement this advanced technology at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. Our facilities are supported by rigorous QC labs and stringent purity specifications, guaranteeing that every batch of (S, R)-8-benzyl-7, 9-dioxo-2, 8-diazabicyclo[4.3.0]nonane meets the exacting standards required by global pharmaceutical regulators. We are committed to delivering not just a chemical product, but a reliable supply solution that enhances your production efficiency.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific production needs. By leveraging this optimized purification technology, we can help you achieve substantial reductions in manufacturing costs while maintaining the highest levels of chiral purity. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. Let us partner with you to secure a stable, cost-effective, and high-quality supply chain for your critical pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
