Advanced Chemical Synthesis of Prednisolone Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for corticosteroids, particularly for high-value anti-inflammatory agents like prednisolone. Patent CN103641879A introduces a groundbreaking preparation method for prednisolone intermediates and their analogues, addressing critical bottlenecks in traditional steroid manufacturing. This technology leverages a sophisticated sequence of elimination, cyano group substitution, siloxy protection, intramolecular nucleophilic substitution, and esterification reactions. By starting from readily available plant sterol derivatives, specifically 11α-hydroxy-1,4-pregnadiene-3,20-dione, the process circumvents the high costs and toxicity associated with conventional iodine-based side-chain modifications. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and economically viable production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for prednisolone and its analogues have long been plagued by economic and environmental inefficiencies. Historically, the industry has relied heavily on a combination of biological fermentation and chemical synthesis, often utilizing cortisone acetate or mold oxide as starting materials. A significant drawback in existing chemical routes, such as those described in prior art documents like CN201210070704.7, is the dependence on iodine for side-chain iodization. Iodine is not only an expensive reagent with fluctuating market prices but also poses significant toxicity challenges, requiring rigorous waste treatment and specialized handling equipment. Furthermore, biological catalysis methods, while stereoselective, often suffer from lower yields and longer cycle times, creating supply chain vulnerabilities for manufacturers needing consistent, large-volume outputs.
The Novel Approach
The methodology disclosed in CN103641879A offers a transformative alternative by replacing expensive halogenation agents with a cost-effective cyano group substitution strategy. Instead of relying on toxic iodine, this novel approach utilizes acetone cyanohydrin or alkali metal cyanides to introduce the necessary functional groups at the C-17 position. This strategic substitution drastically reduces raw material costs and simplifies the downstream purification process by eliminating the need for heavy metal removal steps. The route is designed to be linear and high-yielding, transforming cheap plant sterol derivatives into complex steroid intermediates through a series of well-controlled chemical transformations that are inherently safer and more scalable for industrial applications.
Mechanistic Insights into Siloxy Protection and Intramolecular Substitution
The core innovation of this synthesis lies in the precise manipulation of the steroid side chain through a protected intermediate. After the initial elimination and cyano substitution steps generate Compound III, the process employs a siloxy protective reaction using reagents like chloromethyl dimethyl chlorosilane (CMDMCS). This protection is crucial as it masks the 17-position hydroxyl group, preventing unwanted side reactions during the subsequent aggressive nucleophilic attack. The protected Compound IV then undergoes a critical intramolecular nucleophilic substitution at low temperatures, typically between -40°C and -30°C, using strong bases like n-Butyl Lithium or potassium tert-butoxide. This low-temperature environment is essential for controlling the stereochemistry and preventing the decomposition of the sensitive steroid backbone while successfully constructing the chloromethyl ketone side chain precursor.
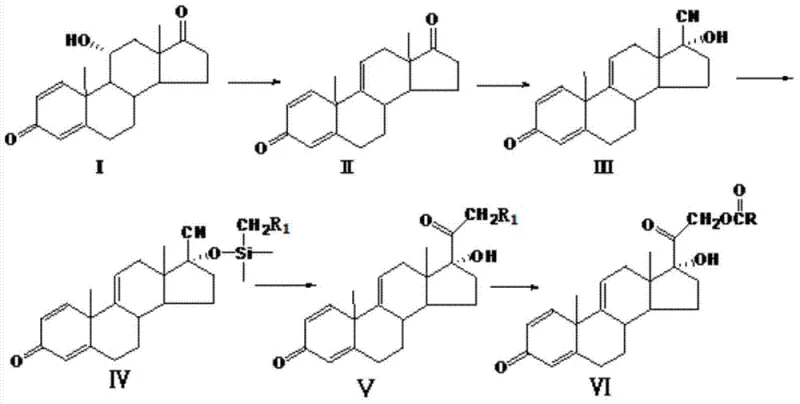
Following the construction of the side chain, the final esterification step converts the intermediate into the target acetate structure. The mechanistic elegance of this route ensures that impurities are minimized at each stage. For instance, the use of specific organic solvents like tetrahydrofuran and controlled addition rates of dehydrating agents ensures that the elimination reaction proceeds cleanly to form the 1,2-double bond without degrading the 11-hydroxyl group. This level of control results in intermediates with HPLC purity consistently exceeding 96%, providing a robust foundation for the final API synthesis. The ability to achieve such high purity without chromatographic purification at every step is a testament to the selectivity of the catalytic and stoichiometric reagents chosen for this pathway.
How to Synthesize Prednisolone Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing prednisolone intermediates with high efficiency and reproducibility. The process begins with the dehydration of the starting steroid using phosphorus pentachloride or tosyl chloride, followed by the introduction of the nitrile group. Detailed operational parameters, including solvent ratios and temperature controls, are critical for maximizing yield. For a comprehensive understanding of the specific reaction conditions, reagent equivalents, and workup procedures required to replicate this high-purity synthesis, please refer to the standardized guide below.
- Perform an elimination reaction on Compound I using a dehydrating agent like phosphorus pentachloride in an organic solvent to obtain Compound II.
- Execute a cyano group substitution on Compound II using acetone cyanohydrin or sodium cyanide to introduce the nitrile group, yielding Compound III.
- Protect the hydroxyl group of Compound III with a siloxy reagent such as CMDMCS, followed by intramolecular nucleophilic substitution at low temperatures to form the side chain.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage is the substantial reduction in raw material expenditure by eliminating the need for costly iodine reagents and expensive fermentation-derived starting materials like cortisone acetate. By shifting to plant sterol derivatives, which are abundant and commoditized, manufacturers can insulate their production costs from the volatility of specialty reagent markets. Additionally, the removal of toxic heavy metals from the process flow significantly lowers waste disposal costs and regulatory compliance burdens, contributing to a leaner and more sustainable manufacturing operation.
- Cost Reduction in Manufacturing: The replacement of iodine-based iodization with cyano substitution fundamentally alters the cost structure of steroid production. Iodine is a high-cost input that also necessitates expensive recovery or disposal systems due to its toxicity. By utilizing inexpensive cyanating reagents and common organic solvents, the overall variable cost per kilogram of the intermediate is significantly lowered. Furthermore, the high yields reported in the patent examples indicate less material loss during processing, which directly translates to improved material efficiency and reduced cost of goods sold (COGS) without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on plant sterols as the foundational feedstock ensures a stable and continuous supply chain, as these materials are produced on a massive global scale for various industries. Unlike specialized biological enzymes or fermentation broths which can suffer from batch-to-batch variability and supply interruptions, chemical reagents like phosphorus pentachloride and acetone cyanohydrin are readily available from multiple global suppliers. This diversification of the supply base reduces the risk of production stoppages and allows for more accurate forecasting and inventory management for long-term contracts.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as filtration, drying, and crystallization that are easily transferable from pilot plant to commercial scale. The avoidance of biological fermentation steps for the side-chain modification simplifies the facility requirements, removing the need for sterile environments and complex bioreactors. Moreover, the reduced toxicity profile of the reagents aligns with increasingly stringent environmental regulations, facilitating easier permitting and reducing the carbon footprint associated with hazardous waste treatment, thereby future-proofing the manufacturing asset.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this prednisolone intermediate synthesis. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy for decision-makers evaluating this technology for potential licensing or procurement partnerships.
Q: What are the primary advantages of this synthesis route over conventional biological methods?
A: This chemical synthesis route eliminates the reliance on expensive iodine reagents and complex biological fermentation steps for side-chain modification, resulting in lower raw material costs and simplified purification processes.
Q: How does the process ensure high purity for the final intermediate?
A: The process utilizes specific protective group strategies (siloxy protection) and controlled low-temperature nucleophilic substitutions which minimize side reactions and impurity formation, allowing for high HPLC purity greater than 96%.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method uses cheap and easily available plant sterol derivatives as starting materials and avoids toxic heavy metals, making it highly scalable and environmentally compliant for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prednisolone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the competitive landscape of corticosteroid manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in steroid chemistry allows us to optimize processes like the one described in CN103641879A to maximize yield and minimize impurities for our global partners.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced technology for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your product portfolio and drive down your overall production costs.
