Advanced Catalytic Hydrogen Transfer for Commercial Scale Menthone Production
Advanced Catalytic Hydrogen Transfer for Commercial Scale Menthone Production
The global demand for high-purity terpenes and their derivatives continues to surge, driven by the pharmaceutical, flavor, and fragrance industries. A pivotal advancement in this sector is detailed in patent CN107602357B, which discloses a novel method for preparing menthone from isopulegol. Unlike conventional routes that rely on hazardous stoichiometric oxidants or expensive heterogeneous hydrogenation setups, this invention utilizes a sophisticated homogeneous catalytic system. By employing transition metal complexes coupled with specific auxiliary agents, the process facilitates an efficient intramolecular hydrogen transfer. This technological leap not only achieves conversion rates exceeding 99% but also delivers exceptional catalyst turnover numbers (TON) reaching up to 100,000. For industry leaders seeking a reliable menthone supplier, understanding the mechanistic depth and commercial viability of this solvent-free approach is essential for securing a competitive edge in the supply chain.
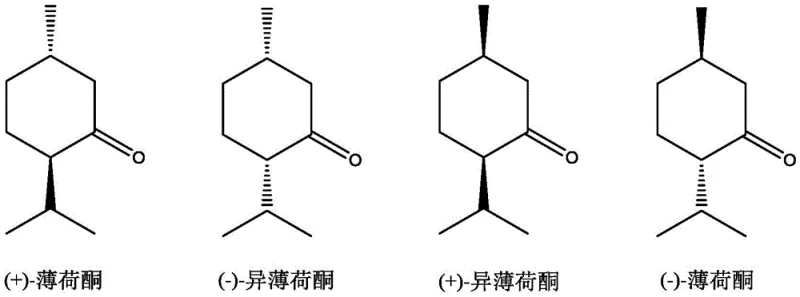
Menthone exists in various stereoisomeric forms, as illustrated in the structural diagrams above, each possessing distinct organoleptic and biological properties. The ability to selectively synthesize these isomers from isopulegol precursors is a hallmark of advanced fine chemical manufacturing. The patent highlights that the term 'menthone' in this context encompasses any possible stereoisomer, acknowledging the complexity of the chiral landscape involved in terpene synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of menthone has been plagued by significant environmental and operational challenges. Traditional pathways predominantly rely on the oxidation of menthol using stoichiometric oxidants such as chromates, permanganates, or hypochlorites. While these methods are chemically mature, they are inherently unsustainable due to the generation of massive quantities of toxic heavy metal waste and byproduct salts. Furthermore, alternative routes involving the hydrogenation of thymol often suffer from raw material availability issues, as thymol is not always easily sourced in the required purity and volume. Other catalytic attempts, such as those using activated copper oxide in the gas phase, necessitate rigorous pre-activation steps involving hydrogen and alcohol at elevated temperatures, adding layers of operational complexity and safety risks. Additionally, prior art involving phosphine-ligand metal complexes often fails to achieve high turnover numbers, resulting in short catalyst lifespans and prohibitive costs that render them unsuitable for large-scale flavor and fragrance intermediates manufacturing.
The Novel Approach
The methodology described in patent CN107602357B represents a paradigm shift by eliminating the need for external hydrogen donors or toxic oxidants. Instead, it leverages a hydrogen transfer mechanism where the isopulegol substrate undergoes rearrangement catalyzed by a transition metal complex. This approach operates under mild conditions without the addition of organic solvents, effectively creating a 'neat' reaction system. The integration of specific auxiliary agents, such as norbornadiene or cyclopentadiene, stabilizes the active catalytic species, preventing decomposition and extending the catalyst's functional life. This results in a process that is not only greener, with significantly reduced three-waste discharge, but also economically superior due to the drastic reduction in catalyst loading requirements. The system's ability to function without solvent simplifies the post-reaction purification process, directly translating to lower energy consumption and faster production cycles for commercial scale-up of complex terpenes.
Mechanistic Insights into Ru-Catalyzed Hydrogen Transfer
At the heart of this innovation lies a robust homogeneous catalytic cycle driven by Group VIII transition metals, with Ruthenium (Ru) being the preferred candidate. The catalyst, typically a ruthenium acetylacetonate complex, interacts with the isopulegol substrate to facilitate the migration of hydrogen atoms within the molecule. The presence of the auxiliary agent is critical; it coordinates with the metal center to form a highly active intermediate that lowers the activation energy for the hydrogen transfer step. This synergy allows the reaction to proceed efficiently at temperatures ranging from 100°C to 300°C under inert gas pressure. The mechanism avoids the formation of unstable radical species that often lead to polymerization or degradation in thermal processes, ensuring a clean reaction profile. The result is a highly selective transformation where the double bond in isopulegol is reduced while the ketone functionality is preserved or generated via tautomerization, depending on the specific isomer pathway.
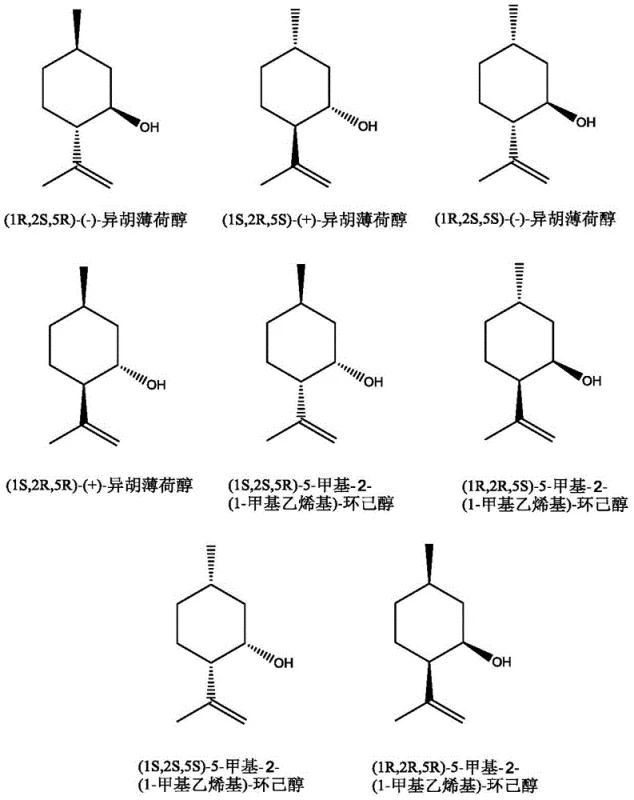
The starting material, isopulegol, also presents a complex stereochemical profile, as shown in the diagram above. The catalytic system demonstrates remarkable tolerance to these variations, capable of processing different stereoisomers of isopulegol into the desired menthone products. The reaction selectivity is maintained between 85% and 99%, indicating that the catalyst effectively discriminates against side reactions such as over-reduction or skeletal rearrangement. Impurity control is further enhanced by the absence of solvent, which minimizes the potential for solvent-substrate interactions that could lead to etherification or other solvent-derived byproducts. The final product mixture typically contains a controlled proportion of isomenthone (20-50 mol%), allowing manufacturers to tailor the output for specific applications requiring distinct cooling sensations or aromatic profiles.
How to Synthesize Menthone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a pilot or production setting. The process begins with the preparation of a homogeneous solution containing the transition metal catalyst and the auxiliary agent directly dissolved in the isopulegol feedstock. This mixture is then subjected to a strict oxygen-free and water-free environment, typically achieved by purging with high-purity nitrogen or argon. The reaction is conducted under autogenous pressure or slight pressurization (0.1-10 MPa) to maintain the integrity of the volatile components and ensure efficient mass transfer. Detailed standard operating procedures regarding specific molar ratios, heating ramps, and quenching methods are critical for reproducibility and safety.
- Prepare the reaction mixture by dissolving a homogeneous transition metal catalyst (such as ruthenium acetylacetonate) and a specific auxiliary agent (like cyclopentadiene) directly into the isopulegol substrate under an oxygen-free and water-free atmosphere.
- Introduce high-purity inert gas (nitrogen or argon) to maintain an absolute pressure between 0.1 and 10 MPa, ensuring a strictly anaerobic environment essential for catalyst stability.
- Heat the mixture to a temperature range of 100-300°C and stir for 1 to 100 hours to facilitate the intramolecular hydrogen transfer, yielding menthone with conversion rates up to 99.9%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic hydrogen transfer technology offers profound strategic benefits beyond mere chemical efficiency. The elimination of stoichiometric oxidants and organic solvents fundamentally alters the cost structure of menthone production. By removing the need for expensive waste treatment associated with heavy metal sludge and spent solvents, the overall operational expenditure is significantly reduced. Furthermore, the high turnover number (TON) of the catalyst means that a minute quantity of precious metal can produce a vast amount of product, decoupling production capacity from the volatility of noble metal markets. This efficiency ensures a more stable pricing model for high-purity menthone, shielding downstream customers from raw material price fluctuations.
- Cost Reduction in Manufacturing: The solvent-free nature of this reaction is a major driver for cost optimization. In traditional processes, the recovery and recycling of solvents constitute a significant portion of energy costs and capital investment in distillation columns. By running the reaction neat, the energy load for solvent recovery is entirely eliminated. Additionally, the simplified workup procedure—often requiring only filtration or simple distillation to remove the catalyst residue—reduces labor hours and equipment downtime. The high selectivity of the reaction minimizes the formation of hard-to-separate byproducts, thereby increasing the overall yield of saleable product per batch and maximizing asset utilization.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the catalytic system. The use of readily available transition metal salts and common diene auxiliaries reduces dependency on exotic or single-source reagents. The process operates effectively under a wide range of pressures and temperatures, providing operational flexibility that allows manufacturers to adapt to utility constraints without compromising output quality. This flexibility ensures consistent delivery schedules, a critical factor for clients managing just-in-time inventory systems for pharmaceutical intermediates and consumer goods.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental emissions tighten globally, this technology offers a future-proof solution. The drastic reduction in hazardous waste generation aligns with green chemistry principles and simplifies compliance with environmental protection regulations. The scalability of the homogeneous system is proven by the high TON values, indicating that the catalyst remains effective even as reaction volumes increase. This makes the transition from laboratory scale to multi-ton commercial production seamless, reducing the risk and time associated with process scale-up campaigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrogen transfer technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: How does this hydrogen transfer method improve upon traditional oxidation processes?
A: Traditional methods often rely on stoichiometric oxidants like chromates or permanganates, which generate toxic waste and require complex purification. This patented hydrogen transfer method eliminates the need for external oxidants or hydrogen donors, utilizing the substrate itself for hydrogen redistribution, thereby drastically reducing three-waste discharge and simplifying downstream processing.
Q: What is the significance of the auxiliary agent in this catalytic system?
A: The auxiliary agent, such as cyclopentadiene or cyclooctadiene, plays a critical role in stabilizing the transition metal center. It prevents catalyst deactivation and aggregation, allowing the system to achieve exceptionally high Turnover Numbers (TON) ranging from 1,000 to 100,000, which is vital for industrial cost-effectiveness.
Q: Can the ratio of menthone to isomenthone be controlled in this process?
A: Yes, the process allows for tunable selectivity. While the reaction naturally produces a mixture of stereoisomers, the proportion of isomenthone can be managed within a range of 20 to 50 mol% by adjusting reaction parameters such as temperature, pressure, and the specific choice of auxiliary agents, ensuring the final product meets diverse application specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Menthone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic hydrogen transfer route for menthone production. As a leading CDMO partner, we possess the technical expertise to adapt and optimize this sophisticated chemistry for your specific needs. Our facilities are equipped to handle complex homogeneous catalytic reactions with precision, ensuring that the stringent purity specifications required by the flavor and pharmaceutical industries are consistently met. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our rigorous QC labs to monitor every step of the synthesis and guarantee product integrity.
We invite you to collaborate with us to explore how this advanced manufacturing route can enhance your supply chain efficiency. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how we can become your strategic partner in delivering high-quality menthone solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
