Advanced Procatide Manufacturing: Overcoming Synthesis Barriers with Stepwise Oxidation
Introduction to Advanced Procatide Synthesis Technology
The pharmaceutical industry continuously seeks robust manufacturing routes for complex cyclic peptides, particularly those targeting gastrointestinal disorders like Procatide (a Plecanatide analogue). A significant technological breakthrough in this domain is documented in Chinese patent CN112194708A, which outlines a sophisticated preparation method designed to overcome the inherent difficulties of synthesizing 16-amino acid cyclic peptides with dual disulfide bonds. This patent addresses critical pain points such as low coupling efficiency, resin aggregation, and the formation of incorrect disulfide isomers, which have historically plagued the commercial production of uroguanylin analogues. By introducing a novel resin modification strategy using FMPB and a precise two-stage oxidation protocol, the technology offers a pathway to significantly higher purity and yield. For global procurement leaders and R&D directors, understanding this methodology is essential for securing a reliable procatide supplier capable of delivering high-purity active pharmaceutical ingredients at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional solid-phase peptide synthesis (SPPS) for Procatide often suffers from severe limitations that impact both cost and quality. Conventional methods typically involve direct coupling of amino acids onto standard resins without specific modifications to prevent secondary structure formation. As the peptide chain grows, particularly with hydrophobic sequences, the resin-bound peptides tend to aggregate through beta-sheet formation. This aggregation shields reactive amino groups, leading to incomplete couplings, deletion sequences, and a complex impurity profile that is difficult to purge. Furthermore, standard cyclization protocols often attempt to form both disulfide bonds simultaneously in the liquid phase. This non-selective approach frequently results in scrambled disulfide connectivities (e.g., Cys4-Cys7 instead of Cys4-Cys12) and intermolecular polymerization, drastically reducing the yield of the desired native structure and necessitating expensive and wasteful purification steps.
The Novel Approach
The methodology described in CN112194708A introduces a paradigm shift by modifying the solid support itself and decoupling the oxidation events. The process begins with the modification of AM or MBHA resin using 4-(4'-acetyl-3'-methoxy)phenoxybutyric acid (FMPB), followed by the attachment of the first amino acid, H-Leu-OtBu, via reductive amination. This creates a stable anchor that supports efficient chain elongation. Crucially, the synthesis incorporates Hmb (2-hydroxy-4-methoxybenzyl) protection at specific difficult-to-couple positions (R1 and R2 in the sequence), which acts as a temporary solubilizing tag to break up beta-sheets during synthesis. Following cleavage, the linear peptide undergoes a sequential oxidation strategy: first forming the Cys4-Cys12 bond under mild conditions, purifying the intermediate, diluting the solution to minimize intermolecular collisions, and finally forming the Cys7-Cys15 bond. This stepwise logic effectively eliminates the formation of scrambled isomers and ensures a much cleaner final product profile.
Mechanistic Insights into FMPB-Modified SPPS and Stepwise Oxidation
The core of this synthesis lies in the chemical engineering of the resin-peptide interface and the kinetic control of disulfide bond formation. The initial modification involves reacting the amino group on the AM or MBHA resin with FMPB in the presence of condensing agents like HOBT-DIC or HATU-NMM. This forms an amide linkage that serves as a handle for the subsequent reductive amination with H-Leu-OtBu. The use of FMPB provides a flexible spacer arm that reduces steric hindrance near the resin surface, allowing the growing peptide chain greater freedom of movement and accessibility for reagents. During the chain assembly, the strategic insertion of Hmb groups on the backbone nitrogen of specific amino acids prevents the inter-chain hydrogen bonding that leads to aggregation. This ensures that each coupling step proceeds with high efficiency, minimizing the accumulation of n-1 deletion impurities that are notoriously difficult to separate from the final API.
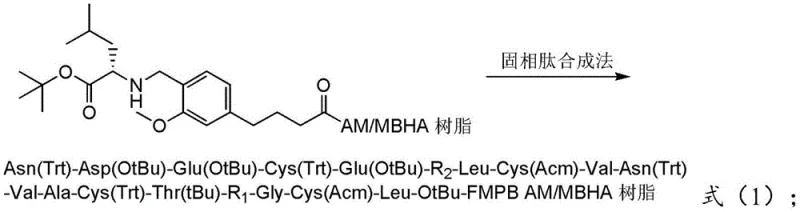
Following the assembly and cleavage of the fully protected linear peptide using a TFA-based cocktail (typically TFA:EDT:TIS:H2O at ratios such as 90:5:2.5:2.5), the critical oxidation phase begins. The first oxidation targets the Cys4 and Cys12 residues. By adjusting the pH to 7-10 and using mild oxidants like air, hydrogen peroxide, or DMSO, the thiol groups are selectively converted to a disulfide bond while the other cysteine residues (Cys7 and Cys15) remain protected with Acm (acetamidomethyl) groups. This orthogonality is vital; the Acm group is stable under these mild oxidative conditions, preventing premature or incorrect bonding. After this first cyclization, the intermediate is subjected to column chromatography to remove reagents and byproducts before the second oxidation.
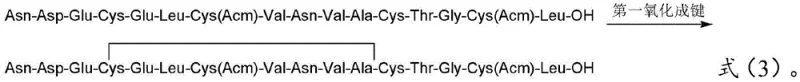
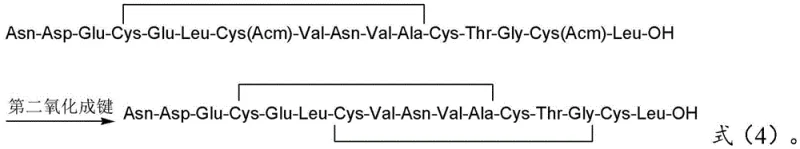
The second oxidation step is engineered to maximize yield by controlling concentration. The purified mono-cyclic intermediate is diluted significantly (dilution ratio of 2-20 times) before adding a stronger oxidant such as iodine, mercury salts, or silver salts. This high dilution principle is thermodynamic; it favors intramolecular ring closure (forming the Cys7-Cys15 bond) over intermolecular polymerization (dimerization or oligomerization). The removal of the Acm protecting groups occurs concurrently or subsequently depending on the oxidant choice (e.g., Iodine removes Acm), finalizing the bicyclic structure of Procatide. This meticulous separation of oxidation events ensures that the final product possesses the correct native topology required for biological activity as a guanylate cyclase-C receptor agonist.
How to Synthesize Procatide Efficiently
Implementing this synthesis route requires precise adherence to the reaction parameters defined in the patent to ensure reproducibility and quality. The process moves from solid-phase modification to liquid-phase cyclization, demanding strict control over stoichiometry, pH, and purification intervals. The following overview summarizes the operational flow, highlighting the critical control points where deviations could lead to impurity spikes. For detailed standard operating procedures and specific reagent grades required for GMP compliance, please refer to the technical guidelines below.
- Modify AM or MBHA resin with FMPB and couple H-Leu-OtBu via reductive amination to form the starting resin.
- Perform solid-phase peptide synthesis (SPPS) coupling amino acids sequentially, utilizing Hmb modification at difficult positions to prevent aggregation.
- Cleave the protected peptide, perform first oxidation (Cys4-Cys12), purify, dilute, and perform second oxidation (Cys7-Cys15) followed by preparative chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic advantages beyond mere technical feasibility. The primary value driver is the substantial improvement in process robustness, which directly correlates to supply continuity. By mitigating the risks of resin aggregation and incorrect disulfide pairing, the manufacturing process becomes more predictable, reducing the likelihood of batch failures that can disrupt global supply lines. Furthermore, the enhanced purity profile achieved through the intermediate purification step means that the final polishing chromatography is less burdened, leading to better recovery rates of the final API. This efficiency is crucial for maintaining cost competitiveness in the therapeutic peptide market.
- Cost Reduction in Manufacturing: The elimination of complex scrambling issues significantly lowers the cost of goods sold. In conventional synthesis, a large portion of the budget is often consumed by the loss of material during the purification of scrambled isomers. By preventing these impurities from forming in the first place through stepwise oxidation and Hmb modification, the overall material throughput is optimized. Additionally, the use of readily available oxidants like air or hydrogen peroxide for the first step avoids the immediate consumption of expensive heavy metal reagents, reserving them only for the final specific deprotection step where absolutely necessary. This logical allocation of reagents ensures that cost reduction in polypeptide manufacturing is achieved through chemical intelligence rather than simple resource cutting.
- Enhanced Supply Chain Reliability: The scalability of this method is a key asset for long-term supply agreements. The solid-phase synthesis steps utilize standard equipment found in most peptide CDMOs, meaning there is no need for exotic hardware that could create bottlenecks. The liquid-phase oxidation steps are performed under mild conditions (room temperature to 35°C), which reduces energy consumption and safety risks associated with exothermic reactions. This operational simplicity ensures that the reliable procatide supplier can ramp up production from kilogram to multi-ton scales without encountering the nonlinear difficulties often seen in complex peptide cyclizations, thereby guaranteeing consistent lead times.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, the process is designed to minimize waste generation. The intermediate purification removes impurities early, preventing them from carrying through to the final stages where they would require larger volumes of solvent to remove. The specific control over oxidant usage (e.g., titrating iodine until color fade) prevents the discharge of excess hazardous chemicals. This aligns with modern green chemistry principles, making the commercial scale-up of complex cyclic peptides more sustainable and easier to approve within strict environmental frameworks, ultimately reducing the regulatory burden on the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Procatide using this advanced methodology. These insights are derived directly from the experimental data and claims within the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders assess the feasibility of integrating this supply source into their existing development pipelines.
Q: How does the FMPB modification improve Procatide synthesis yield?
A: FMPB modification allows for the attachment of the first amino acid (Leucine) via a stable linkage that facilitates subsequent coupling. More importantly, the use of Hmb (2-hydroxy-4-methoxybenzyl) groups at specific positions disrupts beta-sheet formation during chain elongation, significantly reducing resin aggregation and improving coupling efficiency for difficult sequences.
Q: Why is a two-step oxidation process preferred for Procatide?
A: Procatide contains two disulfide bonds (Cys4-Cys12 and Cys7-Cys15). Simultaneous oxidation often leads to scrambled isomers and intermolecular polymerization. By oxidizing Cys4-Cys12 first under mild conditions (air/H2O2), purifying, and then oxidizing Cys7-Cys15 under controlled conditions (Iodine) at high dilution, the method drastically reduces intermolecular side reactions and ensures the correct native fold.
Q: What are the critical purity specifications mentioned in the patent?
A: The patent demonstrates that following this specific protocol, including column chromatography coarse purification between oxidation steps, can achieve a final purity of over 99.7% with a maximum single impurity content as low as 0.04%, meeting stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Procatide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate steps of FMPB modification and stepwise oxidation are executed with precision. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect even trace levels of disulfide scramblers, guaranteeing that every batch of Procatide meets the highest international standards for gastrointestinal therapeutics.
We invite you to collaborate with us to leverage this advanced synthesis technology for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing process can enhance your supply chain security and product quality.
