Revolutionizing Aziridine Intermediate Production Via Enzymatic Catalysis For Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to construct high-value heterocyclic scaffolds, among which the aziridine three-membered ring stands out as a critical structural motif in numerous bioactive compounds. Patent CN110982800B introduces a groundbreaking biocatalytic approach that utilizes a specific sulfonyltransferase to mediate the biosynthesis of aziridine rings, marking a significant departure from traditional chemical synthesis routes. This technology leverages the enzyme Fic28 to catalyze the transfer of a sulfonyl group from 3'-phosphoadenosine-5'-phosphosulfate (PAPS) to amino alcohol substrates, followed by a spontaneous cyclization under alkaline conditions. The establishment of this in vitro catalytic system provides a robust theoretical and practical foundation for the green manufacturing of aziridine drug intermediates, addressing long-standing challenges regarding selectivity and reaction severity. By shifting from harsh chemical reagents to precise enzymatic catalysis, this innovation opens new avenues for producing complex derivatives like Mitomycin C precursors with enhanced purity profiles and reduced environmental footprints.
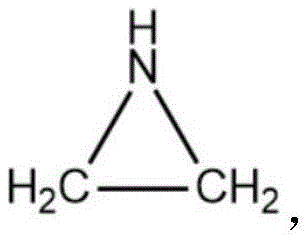
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of aziridine rings has relied heavily on homogeneous and heterogeneous chemical methods that impose severe constraints on manufacturing efficiency and safety. Traditional homogeneous methods, such as the ethanolamine sulfuric acid esterification or the 2-chloroethylamine route, necessitate the use of corrosive strong acids and bases, leading to significant equipment degradation and high maintenance costs over time. Furthermore, these processes often generate substantial quantities of acidic or alkaline wastewater, creating a heavy burden on environmental treatment facilities and complicating regulatory compliance for large-scale production sites. The heterogeneous methods, while attempting to mitigate some liquid waste issues, frequently require high-temperature gas-phase dehydration which demands excessive energy input and often suffers from catalyst deactivation and short operational lifespans. These inherent drawbacks result in a fragmented supply chain where production stability is frequently compromised by the need for frequent catalyst replacement and rigorous waste management protocols.
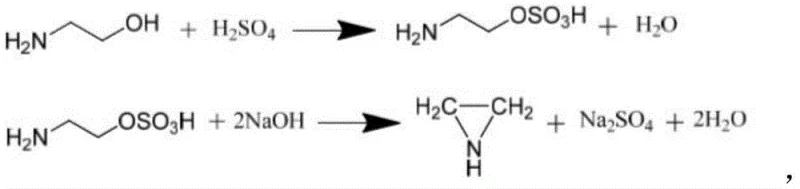
The Novel Approach
In stark contrast to the aggressive conditions of legacy chemical processes, the novel enzymatic method described in the patent operates under remarkably mild and controlled parameters that preserve both product integrity and equipment longevity. The core of this innovation lies in the use of a recombinant sulfonyltransferase which facilitates the reaction in an aqueous PBS buffer system at temperatures ranging strictly from 25-42°C, eliminating the need for energy-intensive heating or cooling cycles. This biocatalytic route avoids the use of hazardous chlorinating agents and strong mineral acids, thereby drastically reducing the generation of toxic byproducts and simplifying the downstream purification process. The specificity of the enzyme ensures that the sulfonyl group is transferred precisely to the target hydroxyl group, minimizing the formation of unwanted isomers and reducing the complexity of the impurity profile. This shift towards biomanufacturing represents a paradigm change in how high-strain ring systems are constructed, offering a cleaner, safer, and more predictable production environment for sensitive pharmaceutical intermediates.
Mechanistic Insights into Sulfonyltransferase-Mediated Cyclization
The mechanistic elegance of this biosynthetic pathway relies on a multi-enzyme cascade that mimics natural product biosynthesis to achieve high-fidelity ring closure. The process initiates with the generation of the active sulfonyl donor, PAPS, which is subsequently utilized by the sulfonyltransferase (Fic28) to modify the hydroxyl group of the amino alcohol substrate. This enzymatic sulfonation converts a poor leaving group into an excellent sulfonate ester intermediate, which is primed for the subsequent intramolecular nucleophilic attack. The precision of the enzyme active site ensures that this modification occurs with high regioselectivity, preventing side reactions that are common in non-enzymatic sulfonation attempts. Once the sulfonated intermediate is formed, the system is subjected to a pH adjustment above 10.0 using sodium hydroxide, which triggers the deprotonation of the neighboring amine group. This deprotonated amine then acts as a nucleophile, attacking the carbon bearing the sulfonate group to displace the leaving group and close the three-membered aziridine ring.
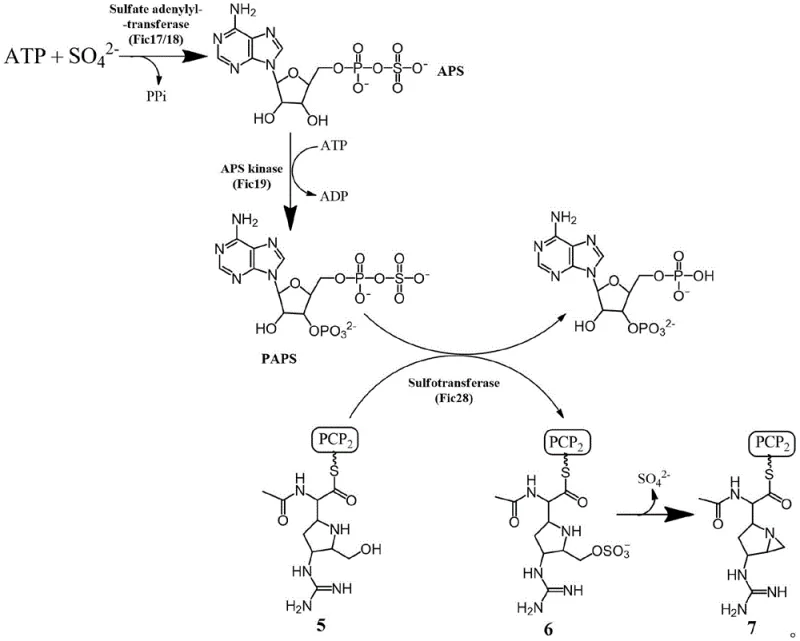
Controlling the impurity profile in aziridine synthesis is paramount for pharmaceutical applications, and this enzymatic method offers superior control compared to chemical alternatives. The mild reaction conditions prevent the thermal degradation of sensitive functional groups that might be present on complex substrate molecules, a common issue when using high-temperature gas-phase dehydration methods. Additionally, the use of a specific protein catalyst reduces the likelihood of non-specific alkylation or polymerization reactions that often plague chemical cyclization processes involving strong bases. The ability to conduct the reaction in a buffered aqueous system also facilitates easier quenching and extraction, minimizing the entrapment of inorganic salts that can be difficult to remove from the final organic product. This high level of control over the reaction trajectory ensures that the resulting aziridine intermediates meet the stringent purity specifications required for downstream drug synthesis, reducing the need for extensive recrystallization or chromatographic purification steps.
How to Synthesize Aziridine Efficiently
The implementation of this biosynthetic route requires a structured approach to enzyme preparation and reaction management to ensure consistent yields and product quality. The process begins with the heterologous expression of the fic28 gene in an E. coli host, followed by purification of the active sulfonyltransferase using affinity chromatography to remove host cell proteins. Once the enzyme is prepared, the synthesis is conducted by mixing the amino alcohol substrate with the sulfonyl donor PAPS and the enzyme in a phosphate-buffered saline solution. The detailed standardized synthesis steps see the guide below.
- Prepare the reaction system by diluting the substrate, PAPS, and sulfonyltransferase in PBS buffer to specific concentrations.
- Maintain the reaction mixture at a temperature between 25-42°C for a duration of 20 minutes to 2 hours to allow enzymatic sulfonation.
- Adjust the system pH to above 10.0 using 0.5mol/L NaOH to induce intramolecular nucleophilic substitution and form the aziridine ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this enzymatic technology offers tangible benefits that extend beyond mere technical feasibility into the realm of strategic cost management and risk mitigation. The elimination of hazardous reagents such as thionyl chloride or concentrated sulfuric acid reduces the costs associated with specialized storage, handling, and disposal of dangerous chemicals. Furthermore, the mild operating conditions translate to lower energy consumption for heating and cooling, contributing to a reduction in overall utility costs for the manufacturing facility. The enhanced selectivity of the enzymatic process means higher crude purity, which reduces the load on purification units and increases the overall throughput of the production line. These factors combine to create a more resilient supply chain that is less susceptible to regulatory shutdowns due to environmental violations or safety incidents.
- Cost Reduction in Manufacturing: The enzymatic process eliminates the need for expensive transition metal catalysts and corrosive reagents, leading to substantial cost savings in raw material procurement and waste treatment. By avoiding the use of heavy metals, the downstream processing does not require expensive metal scavenging steps, which further lowers the cost of goods sold. The reduced corrosion rate of reaction vessels extends the capital equipment lifecycle, deferring the need for costly replacements and maintenance shutdowns. Additionally, the higher specificity of the enzyme reduces the formation of byproducts, maximizing the yield of the desired intermediate from every batch of starting material.
- Enhanced Supply Chain Reliability: Adopting a green biocatalytic route minimizes the regulatory risks associated with the handling of toxic chlorinating agents, ensuring smoother operations during environmental audits. The stability of the enzymatic reaction conditions allows for more predictable batch cycles, reducing the variability in production lead times that often occurs with harsh chemical processes. This reliability is crucial for maintaining continuous supply to downstream pharmaceutical manufacturers who depend on just-in-time delivery of critical intermediates. The use of renewable biocatalysts also aligns with corporate sustainability goals, enhancing the brand value and marketability of the supply chain partners involved.
- Scalability and Environmental Compliance: The aqueous nature of the reaction system simplifies the scale-up process, as heat transfer and mixing are more manageable in water than in viscous organic solvents or gas-phase reactors. The significant reduction in hazardous waste generation facilitates easier compliance with increasingly strict global environmental regulations regarding industrial effluent. This environmental advantage reduces the liability and insurance costs associated with chemical manufacturing, making the operation more financially sustainable in the long term. The process is inherently safer, reducing the risk of catastrophic accidents, which protects the workforce and ensures uninterrupted production schedules.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfonyltransferase-mediated aziridine synthesis technology. These answers are derived directly from the experimental data and mechanistic understanding provided in the patent documentation to ensure accuracy and relevance for industry stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this biocatalytic route into existing manufacturing workflows. The information provided here aims to clarify the operational parameters and benefits for technical and commercial decision-makers.
Q: What are the advantages of enzymatic aziridine synthesis over chemical methods?
A: Enzymatic synthesis operates under mild conditions (25-42°C) and neutral pH initially, avoiding the harsh strong acids, bases, and high temperatures required by traditional chemical methods, which significantly reduces equipment corrosion and environmental waste.
Q: Which substrates are compatible with this sulfonyltransferase method?
A: The method has been validated for various amino alcohol structural analogs, including 2-Amino-1-Butanol and Phenylalaninol derivatives, demonstrating broad substrate specificity for generating diverse aziridine intermediates.
Q: How does this technology impact supply chain reliability?
A: By utilizing a biocatalytic route that eliminates the need for hazardous chlorinating agents and extreme reaction conditions, the process enhances operational safety and reduces the risk of production delays associated with strict environmental compliance checks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aziridine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced biocatalytic technologies to deliver high-quality pharmaceutical intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like this enzymatic aziridine synthesis can be successfully translated into robust industrial operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of aziridine intermediate meets the exacting standards required for drug substance manufacturing. Our commitment to technical excellence allows us to navigate the complexities of enzyme immobilization and cofactor regeneration, optimizing the process for maximum economic efficiency.
We invite procurement leaders and technical directors to engage with our Customized Cost-Saving Analysis service to evaluate how this green synthesis route can impact your specific supply chain economics. Please contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project requirements. By partnering with us, you gain access to a supply chain that is not only cost-effective but also aligned with the future of sustainable chemical manufacturing. Let us help you secure a reliable source of high-purity aziridine derivatives for your next generation of therapeutic agents.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
