Advanced Ruthenium-Catalyzed Synthesis of Polysubstituted Pyrroles for Pharmaceutical Applications
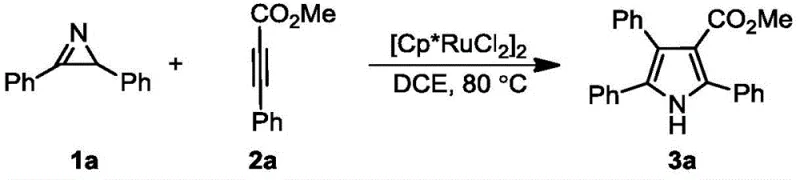
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct nitrogen-containing heterocycles, which serve as the backbone for countless bioactive molecules. Patent CN107522645B introduces a groundbreaking methodology for the preparation of polysubstituted pyrrole compounds, addressing critical bottlenecks in traditional synthetic routes. This technology leverages a ruthenium-catalyzed [3+2] cycloaddition reaction between readily available 2H-aziridine derivatives and monoalkyne compounds. By utilizing this innovative approach, manufacturers can achieve the construction of complex pyrrole scaffolds in a single operational step under relatively mild thermal conditions. The significance of this patent lies not only in its chemical elegance but also in its potential to streamline the supply chain for high-value pharmaceutical intermediates. As a leading entity in the chemical sector, understanding the nuances of such patented technologies is essential for maintaining a competitive edge in the global market for reliable pharmaceutical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrrole ring has relied heavily on classical methodologies such as the Hantzsch, Paal-Knorr, and Knorr reactions. While these methods have served the industry for decades, they are often plagued by significant drawbacks that hinder modern process efficiency. Traditional routes frequently require multi-step sequences involving the pre-synthesis of specific intermediates, which increases the overall material cost and extends the production lead time substantially. Furthermore, many conventional protocols necessitate harsh reaction conditions, including strong acids or bases and elevated temperatures, which can lead to poor atom economy and the generation of substantial chemical waste. The formation of regioisomers is another common challenge in older methodologies, complicating the purification process and reducing the overall yield of the desired target molecule. These inefficiencies create a bottleneck for procurement managers seeking cost reduction in API manufacturing, as the cumulative effect of low yields and complex workups drives up the final price of the active ingredient.
The Novel Approach
In stark contrast to legacy methods, the technology disclosed in CN107522645B offers a streamlined, one-step solution that fundamentally alters the economic landscape of pyrrole synthesis. This novel approach utilizes a direct [3+2] cycloaddition between 2H-aziridine derivatives and monoalkynes, catalyzed by specific ruthenium complexes. The reaction proceeds under mild conditions, typically ranging from 20°C to 120°C, with a preferred operating window of 80°C to 100°C, which significantly reduces energy consumption compared to high-temperature alternatives. The versatility of this method is demonstrated by its broad substrate scope, accommodating various functional groups such as esters, ketones, and diverse aromatic systems without the need for extensive protecting group strategies. By collapsing multiple synthetic steps into a single transformation, this process drastically simplifies the operational workflow and enhances the overall throughput of the manufacturing facility.
![General reaction scheme showing the ruthenium-catalyzed [3+2] cycloaddition of 2H-aziridine derivatives and monoalkynes to form polysubstituted pyrroles](/insights/img/polysubstituted-pyrrole-synthesis-ru-catalysis-pharma-supplier-20260304071502-01.webp)
Mechanistic Insights into Ruthenium-Catalyzed [3+2] Cycloaddition
The core of this technological advancement lies in the unique activation mode provided by the pentamethylcyclopentadienyl ruthenium catalysts. Mechanistically, the ruthenium center coordinates with the alkyne moiety of the monoalkyne substrate, increasing its electrophilicity and facilitating the nucleophilic attack by the nitrogen-containing aziridine ring. This coordination lowers the activation energy barrier for the ring-opening of the strained aziridine, allowing the [3+2] cyclization to proceed smoothly at moderate temperatures. The specific choice of catalyst, such as [Cp*RuCl2]2, is critical as it balances reactivity with stability, ensuring that the catalytic cycle turns over efficiently without premature deactivation. Understanding this mechanistic pathway is vital for R&D directors focused on purity and impurity profiles, as the controlled nature of the metal-ligand interaction minimizes the formation of unwanted byproducts that often arise from uncontrolled radical pathways in non-catalytic methods.
Furthermore, the reaction exhibits remarkable regioselectivity, which is a paramount concern when synthesizing complex pharmaceutical intermediates. The electronic and steric properties of the substituents on both the aziridine and the alkyne influence the orientation of the cycloaddition, yet the ruthenium catalyst helps to direct the formation of the desired polysubstituted pyrrole isomer with high fidelity. For instance, the patent data illustrates successful reactions with substrates bearing electron-withdrawing groups like nitro groups, as well as electron-rich systems like thiophenes, indicating a robust tolerance to electronic variance. This level of control over the reaction outcome ensures that the resulting impurity profile is clean and manageable, reducing the burden on downstream purification processes. Such predictability in chemical behavior is a key factor in scaling up complex pharmaceutical intermediates from the laboratory bench to commercial production volumes.
How to Synthesize Polysubstituted Pyrroles Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and reproducibility. The process begins with the preparation of the reaction vessel, where an inert atmosphere is established to prevent catalyst oxidation. The specific protocol involves charging the reactor with the ruthenium catalyst and a suitable solvent, such as 1,2-dichloroethane, before introducing the substrates. The molar ratio of the aziridine to the alkyne can be adjusted between 1:1 and 1:10, offering flexibility to drive the reaction to completion depending on the cost and availability of the starting materials. Following the reaction period of 12 to 24 hours at the optimized temperature, the workup procedure is straightforward, involving solvent removal and standard chromatographic purification. The detailed standardized synthesis steps for this specific transformation are outlined in the guide below.
- Prepare the reaction vessel by evacuating and backfilling with inert gas (argon or nitrogen) three times to ensure an oxygen-free environment.
- Add the ruthenium catalyst (e.g., [Cp*RuCl2]2) and solvent (e.g., 1,2-dichloroethane) to the vessel, followed by the 2H-aziridine derivative and monoalkyne substrates.
- Heat the mixture to 80°C-100°C for 12-24 hours, then remove solvent and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ruthenium-catalyzed methodology presents a compelling value proposition centered on operational efficiency and cost optimization. The primary driver of value is the simplification of the synthetic route; by eliminating the need for multi-step precursor synthesis, the total number of unit operations is significantly reduced. This reduction directly translates to lower labor costs, decreased equipment occupancy time, and a smaller physical footprint required for production. Additionally, the starting materials, specifically the 2H-aziridine derivatives, can be prepared from cheap and easily obtained oximes via Neber rearrangement, ensuring a stable and cost-effective supply of raw materials. The ability to source precursors from commodity chemical markets mitigates the risk of supply chain disruptions that often plague specialty intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the high atom economy of the [3+2] cycloaddition and the elimination of expensive reagents often required in traditional pyrrole synthesis. Since the reaction operates under mild thermal conditions, the energy demand for heating and cooling is substantially lower than that of high-temperature pyrolysis or reflux methods. Furthermore, the use of a robust ruthenium catalyst at low loading levels (0.05-0.1:1 molar ratio) ensures that the cost contribution of the precious metal remains minimal relative to the value of the final product. The simplified workup procedure, which avoids complex extraction or crystallization steps in favor of direct chromatography, further reduces the consumption of solvents and consumables, leading to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: From a logistics perspective, the broad substrate scope of this reaction enhances supply chain resilience by allowing for the use of diverse starting materials. If a specific alkyne or aziridine derivative becomes unavailable due to market fluctuations, the flexibility of the chemistry allows for the substitution of alternative analogs without requiring a complete process redevelopment. This adaptability is crucial for maintaining continuous production schedules for high-purity pharmaceutical intermediates. Moreover, the reaction's tolerance to various functional groups means that the same core process can be applied to synthesize a library of related compounds, maximizing the utility of the manufacturing infrastructure and reducing the need for dedicated production lines for different products.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory experiments to multi-ton commercial production is facilitated by the homogeneous nature of the catalytic system and the use of common industrial solvents like dichloroethane or toluene. The mild reaction conditions reduce the safety risks associated with high-pressure or high-temperature operations, making the process easier to validate for Good Manufacturing Practice (GMP) compliance. Additionally, the high selectivity of the reaction minimizes the generation of hazardous byproducts, simplifying waste treatment and disposal procedures. This alignment with green chemistry principles not only reduces environmental liability but also aligns with the increasingly stringent regulatory requirements faced by modern chemical manufacturers globally.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this patented synthesis method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering practical guidance for process chemists and engineering teams. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production workflows and for troubleshooting potential issues during scale-up activities.
Q: What catalyst system is optimal for this pyrrole synthesis?
A: The patent specifies metal ruthenium salts such as [Cp*RuCl2]2, Cp*Ru(COD)Cl, or [Cp*RuCl]4 as highly effective catalysts for driving the [3+2] cycloaddition reaction efficiently.
Q: What is the substrate scope for the aziridine component?
A: The method demonstrates excellent tolerance for various substituents on the aziridine, including phenyl, naphthyl, thienyl, and alkyl groups, as well as electron-withdrawing groups like nitro.
Q: How are the final pyrrole products purified?
A: Post-reaction workup involves solvent removal via rotary evaporation followed by standard silica gel column chromatography using petroleum ether and ethyl acetate mixtures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyrroles Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the ruthenium-catalyzed [3+2] cycloaddition in driving innovation within the pharmaceutical sector. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent concept to industrial reality is seamless and efficient. We are committed to delivering high-purity polysubstituted pyrroles that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex heterocyclic synthesis allows us to provide tailored solutions that address the specific needs of global drug developers and fine chemical manufacturers.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that evaluates the economic impact of switching to this novel synthetic route for your specific target molecules. We encourage you to contact us today to obtain specific COA data for our reference standards and to discuss route feasibility assessments for your custom synthesis requirements. Let us help you optimize your supply chain and accelerate your time-to-market with our reliable and scalable manufacturing capabilities.
