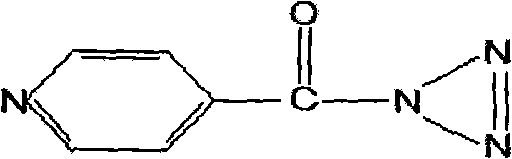
Advanced Synthesis of Isonicotinoyl Azide for High-Purity Agrochemical and Pharmaceutical Intermediates
The chemical landscape for synthesizing high-value heterocyclic intermediates has long been dominated by processes that prioritize laboratory feasibility over industrial efficiency. Patent CN101735144A introduces a transformative approach to the production of isonicotinoyl azide, a critical building block for advanced plant growth regulators and pharmaceutical agents. This technology addresses the historical bottlenecks of solvent dependency and thermal instability by leveraging a precise aqueous-phase nitrosation strategy. By shifting away from volatile organic extraction methods, this innovation not only enhances the purity profile of the final crystal but also fundamentally alters the economic equation for manufacturers seeking reliable agrochemical intermediate supplier partnerships. The structural integrity of the target molecule, characterized by its reactive azide functionality attached to the pyridine ring, demands meticulous handling which this new protocol provides through controlled crystallization dynamics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isonicotinoyl azide has been plagued by operational complexities that render it unsuitable for modern, high-volume manufacturing requirements. Early methodologies, such as those established by Carrana in the 1950s, relied heavily on the use of diethyl ether for extraction, a process that is not only hazardous due to the flammability of the solvent but also inefficient in terms of mass transfer. These legacy techniques often resulted in significant product loss during the multiple washing and drying stages, with yields frequently stagnating below acceptable commercial thresholds. Furthermore, the sensitivity of the azide group to hydrolysis meant that prolonged exposure to aqueous acidic or basic conditions during workup led to rapid decomposition, creating an impurity profile that was difficult to manage without expensive purification steps. The reliance on organic solvents also introduced substantial environmental liabilities, requiring complex recovery systems and waste treatment protocols that inflated the overall cost of goods sold.
The Novel Approach
In stark contrast, the methodology outlined in the referenced patent revolutionizes the production workflow by eliminating organic solvents entirely and utilizing a direct precipitation mechanism. This novel approach capitalizes on the solubility characteristics of the isonicotinoyl azide salt, allowing the product to crystallize directly from the aqueous reaction matrix upon neutralization. By carefully controlling the addition of sodium carbonate to reach a neutral pH, the process avoids the harsh conditions that typically trigger decomposition, thereby locking in high purity levels without the need for chromatographic separation. The transition from a multi-day extraction protocol to a streamlined 2 to 4-hour continuous process represents a paradigm shift in process chemistry, enabling manufacturers to achieve cost reduction in agrochemical intermediate manufacturing through drastically simplified unit operations. This solvent-free design not only mitigates safety risks associated with volatile organics but also ensures a consistent, reproducible output that meets the stringent specifications required by downstream formulators.
Mechanistic Insights into Aqueous Nitrosation and Crystallization
The core of this technological advancement lies in the precise manipulation of reaction kinetics during the nitrosation phase, where isoniazid is converted into the diazonium intermediate before forming the final azide. Maintaining the reaction temperature within the narrow window of 0°C to 5.0°C is critical, as thermal excursions beyond this range accelerate the decomposition of nitrous acid and promote unwanted side reactions that degrade the pyridine scaffold. The stoichiometric balance between isoniazid and sodium nitrite, optimized at a molar ratio of approximately 1:1.2, ensures complete conversion of the starting material while minimizing the presence of residual nitrites that could contaminate the final product. This kinetic control is further supported by the specific concentration of hydrochloric acid, which facilitates the formation of the soluble salt precursor necessary for the subsequent crystallization step. Understanding these mechanistic nuances allows process chemists to replicate the high yields reported in the patent, ensuring that the reactive azide functionality remains intact throughout the synthesis.
Impurity control is inherently built into the physical chemistry of the precipitation step, where the choice of neutralizing agent and its concentration plays a decisive role in the quality of the isolated solid. The patent specifies the use of sodium carbonate solutions with a concentration between 10% and 19%, a parameter that is vital for preventing local alkalinity spikes that could hydrolyze the sensitive azide bond. By slowly adding the base under vigorous stirring, the system maintains a homogeneous pH environment that favors the nucleation of pure isonicotinoyl azide crystals rather than the formation of amorphous oils or degradation byproducts. This direct crystallization from the reaction liquor effectively acts as a purification step, excluding soluble inorganic salts and organic impurities that remain in the mother liquor. Consequently, the resulting white amorphous crystals exhibit a sharp melting point and high chemical purity, reducing the need for downstream recrystallization and validating the process as a robust solution for producing high-purity OLED material or pharmaceutical precursors.
How to Synthesize Isonicotinoyl Azide Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters identified through extensive experimental design, particularly regarding temperature and reagent concentrations. The process begins with the dissolution of isoniazid in a specific concentration of hydrochloric acid, followed by the controlled addition of sodium nitrite to generate the reactive intermediate in situ. The final step involves the careful neutralization of the acidic mixture to induce precipitation, a operation that transforms the soluble species into the isolable solid product. For detailed operational parameters, safety guidelines, and equipment specifications required to execute this synthesis safely and effectively, please refer to the standardized procedure outlined below.
- Dissolve isoniazid in aqueous hydrochloric acid (26%-34% concentration) at a molar ratio of 1: 2.5 to 1:3.5, maintaining the temperature at 0°C.
- Dropwise add aqueous sodium nitrite solution (molar ratio 1: 1.0 to 1:1.5) while keeping the reaction temperature strictly between 0°C and 5.0°C, then stir for 1-2 hours.
- Slowly add 10%-19% aqueous sodium carbonate to adjust pH to 7, inducing direct crystallization of the product, followed by filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solvent-free synthesis technology offers profound strategic benefits that extend far beyond simple unit cost metrics. The elimination of diethyl ether and other volatile organic compounds removes the need for expensive solvent recovery infrastructure and significantly lowers the regulatory burden associated with VOC emissions. This simplification of the manufacturing process translates directly into enhanced supply chain reliability, as the production timeline is compressed from several days to merely a few hours, allowing for rapid response to market demand fluctuations. Furthermore, the high stability of the yield means that production planning becomes more predictable, reducing the risk of batch failures that can disrupt downstream formulation schedules. By partnering with a supplier who utilizes this advanced methodology, organizations can secure a steady flow of critical intermediates without the volatility associated with legacy chemical processes.
- Cost Reduction in Manufacturing: The economic impact of removing organic solvents from the process cannot be overstated, as it eliminates the capital and operational expenditures linked to solvent storage, handling, and distillation. Without the need for energy-intensive solvent recovery loops, the utility consumption per kilogram of product is drastically reduced, leading to substantial cost savings in utility bills and waste disposal fees. Additionally, the high yield efficiency ensures that raw material utilization is maximized, meaning less starting material is wasted to side reactions or purification losses. This lean manufacturing approach results in a significantly lower cost base, providing a competitive edge in pricing strategies for the final agrochemical or pharmaceutical products.
- Enhanced Supply Chain Reliability: The shortened production cycle time of 2 to 4 hours, compared to the 3 to 7 days required by conventional methods, dramatically improves the agility of the supply chain. This rapid turnaround capability allows manufacturers to operate with lower inventory buffers while still maintaining high service levels, effectively reducing the working capital tied up in stock. The robustness of the aqueous process also minimizes the risk of production stoppages due to solvent supply shortages or environmental compliance issues, ensuring continuous availability of the intermediate. For global buyers, this reliability is crucial for maintaining uninterrupted production lines and meeting strict delivery commitments to end-users.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the absence of flammable organic solvents significantly de-risks the scale-up process, making it easier to transition from pilot plant to commercial scale production. The aqueous waste stream generated is easier to treat and dispose of compared to mixed organic-aqueous waste streams, aligning with increasingly stringent global environmental regulations. This green chemistry approach not only future-proofs the manufacturing site against regulatory changes but also enhances the corporate sustainability profile of the supply chain. Companies prioritizing ESG goals will find this solvent-free methodology highly attractive as it demonstrates a commitment to safer and cleaner chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of isonicotinoyl azide, based on the specific advancements detailed in the patent literature. These insights are designed to clarify the operational benefits and safety considerations for technical teams evaluating this synthesis route for potential integration into their supply chains. Understanding these details is essential for making informed decisions about sourcing strategies and process optimization.
Q: How does the new aqueous synthesis method improve yield compared to traditional ether extraction?
A: Traditional methods utilizing ether extraction often suffer from product hydrolysis and emulsion formation, limiting yields to around 70%. The novel aqueous method described in patent CN101735144A utilizes direct precipitation at neutral pH, preventing hydrolysis and achieving stable yields up to 97%.
Q: Is the process scalable for industrial production without organic solvents?
A: Yes, the process is specifically designed for industrial scale-up. By eliminating volatile organic solvents like ether and relying on water-based crystallization, the method significantly reduces fire hazards and environmental waste, making it ideal for large-scale manufacturing of agrochemical intermediates.
Q: What are the critical control parameters for ensuring product stability?
A: Temperature control is paramount; the nitrosation step must remain between 0°C and 5°C to prevent side reactions. Additionally, the concentration of sodium carbonate during neutralization must not exceed 19% to avoid alkaline decomposition of the sensitive azide group.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isonicotinoyl Azide Supplier
The technological breakthroughs presented in patent CN101735144A highlight the immense potential for optimizing the production of complex heterocyclic intermediates through intelligent process design. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like this aqueous nitrosation method are translated into reliable industrial reality. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards. We understand that consistency is key for our partners, and our advanced manufacturing facilities are equipped to handle the precise temperature and pH controls required for this sensitive chemistry.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized synthesis capabilities, we can help you reduce lead time for high-purity agrochemical intermediates while simultaneously lowering your total cost of ownership. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. Let us demonstrate how our engineering expertise can become a catalyst for your supply chain efficiency and product excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
