Scalable Synthesis of Dydrogesterone Intermediates for Commercial Pharmaceutical Production
Scalable Synthesis of Dydrogesterone Intermediates for Commercial Pharmaceutical Production
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical hormonal therapies, and the recent disclosure in patent CN115894593A offers a transformative approach to producing Dydrogesterone and its key intermediates. This technical breakthrough addresses long-standing inefficiencies in steroid synthesis by introducing a route that begins with dideoxynol and proceeds through a series of highly controlled oxidation, decarboxylation, and cyclization steps. For R&D directors and procurement specialists, the significance lies in the method's ability to bypass the low-yield bottlenecks that have historically plagued progesterone-derived syntheses. By leveraging mild reaction conditions and economically accessible raw materials, this new methodology not only enhances the total yield of the final product but also ensures good reproducibility across different batch sizes. The strategic implementation of these chemical transformations represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-purity pharmaceutical intermediates, directly impacting the supply chain reliability for global hormone therapy markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
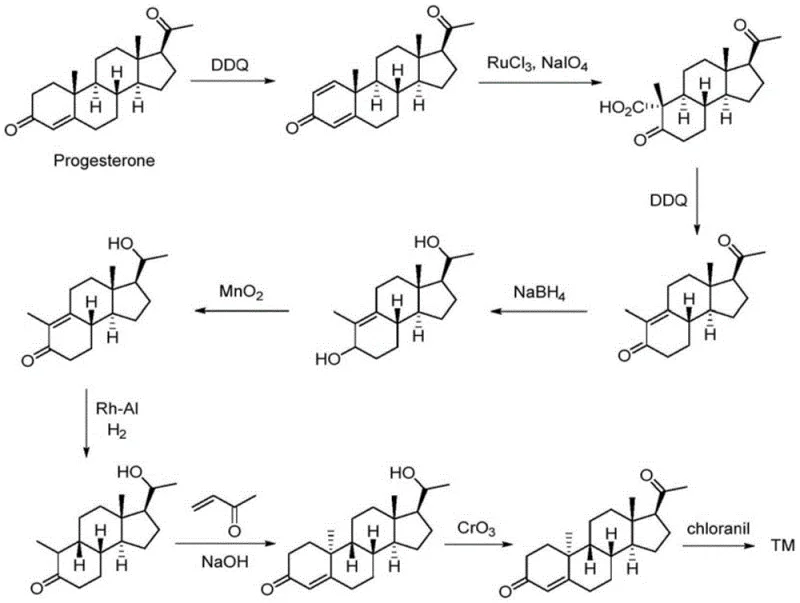
Historically, the synthesis of Dydrogesterone has been hindered by complex multi-step sequences that rely heavily on inefficient transformations and specialized equipment. For instance, prior art such as patent WO2018/109622 describes a route originating from progesterone that involves dehydrogenation and oxidation steps, yet suffers from a total yield lower than 5%, rendering it economically unviable for large-scale production. Another existing method, disclosed in CN110818760A, depends on two-step illumination reactions which require complex photochemical reactors and still result in low yields, making the process unsuitable for industrial amplification. These conventional strategies often involve cutting the A ring through oxidation and regulating chirality via carbon-carbon bond manipulation, which introduces significant risks of incomplete reactions and poor selectivity. The reliance on such harsh or specialized conditions creates substantial supply chain vulnerabilities, as the equipment required is not standard in most fine chemical manufacturing facilities, leading to extended lead times and increased capital expenditure for producers attempting to scale these legacy routes.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the recent patent data introduces a streamlined strategy that avoids the pitfalls of photochemistry and excessive oxidation-reduction cycles. This new pathway utilizes a cyclohexanone oxidation strategy that significantly simplifies the operational complexity, allowing for the use of standard reactor vessels rather than specialized illumination equipment. By focusing on the construction of the intermediate through halogenation and elimination reactions followed by a Robinson cyclization, the method achieves a drastic improvement in reaction efficiency and selectivity. The avoidance of multi-step oxidation-reduction operations not only reduces the consumption of expensive reagents but also minimizes the generation of by-products that complicate downstream purification. This shift in synthetic logic means that manufacturers can achieve higher throughput with fewer processing steps, directly translating to reduced operational costs and a more resilient supply chain capable of meeting the rigorous demands of the global pharmaceutical market without the need for prohibitive infrastructure investments.
Mechanistic Insights into FeCl3-Catalyzed Cyclization and Oxidative Rearrangement
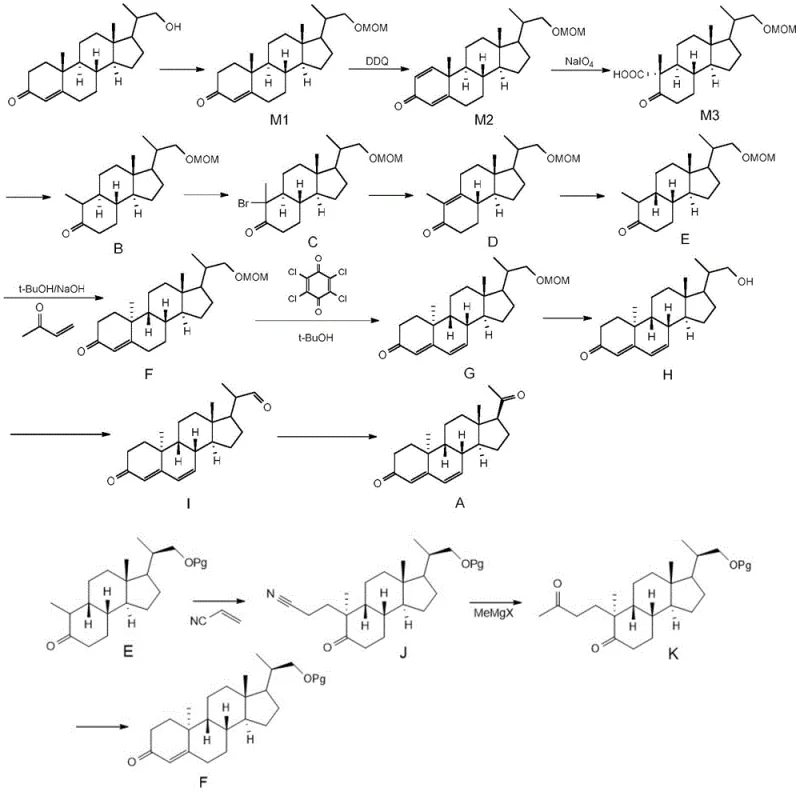
The core of this synthetic advancement lies in the precise control of stereochemistry and functional group transformations during the construction of the steroid backbone. The process begins with the protection of the hydroxyl group in dideoxynol, followed by a selective oxidation using DDQ to introduce unsaturation, and subsequent oxidative cleavage with sodium periodate to open the ring system. A critical mechanistic step involves the alpha-halogenation of the ketone intermediate using N-bromosuccinimide, which sets the stage for a subsequent elimination reaction driven by lithium carbonate and lithium bromide. This elimination is crucial for establishing the correct double bond geometry required for the final cyclization. The process then employs catalytic hydrogenation to reduce specific unsaturated bonds without affecting the sensitive ketone functionalities, ensuring that the chiral centers remain intact throughout the sequence. This level of mechanistic precision is vital for R&D teams focused on impurity control, as it prevents the formation of diastereomers that are difficult to separate and could compromise the safety profile of the final active pharmaceutical ingredient.
Furthermore, the final stages of the synthesis involve a sophisticated oxidative rearrangement that constructs the final Dydrogesterone structure with high fidelity. This step utilizes a copper salt catalyst in the presence of a ligand such as phenanthroline under an oxygen atmosphere, which facilitates the migration of functional groups to their thermodynamically stable positions. The use of IBX oxidation in preceding steps ensures that the hydroxyl groups are converted to ketones with minimal over-oxidation, maintaining the integrity of the steroid nucleus. For technical teams, understanding this mechanism is key to optimizing process parameters such as temperature and solvent choice, as the reaction is sensitive to moisture and oxygen levels. The ability to control these variables allows for the consistent production of high-purity intermediates, reducing the burden on quality control laboratories and ensuring that every batch meets the stringent specifications required for regulatory approval in major pharmaceutical markets.
How to Synthesize Dydrogesterone Efficiently
The implementation of this synthesis route requires a clear understanding of the sequential transformations that convert the starting material into the final high-value intermediate. The process is designed to be operationally simple, utilizing common solvents like ethanol, toluene, and DMF which are readily available in most chemical supply chains. The initial steps involve the protection of the hydroxyl group followed by oxidation and ring opening, which set the foundation for the subsequent carbon-carbon bond forming reactions. Detailed standardized synthesis steps are critical for ensuring reproducibility, particularly during the halogenation and elimination phases where reaction times and temperatures must be strictly controlled to prevent side reactions. The final cyclization step using methyl vinyl ketone is exothermic and requires careful addition rates to maintain safety and yield. By adhering to these optimized conditions, manufacturers can achieve the high yields reported in the patent data, such as the 99% yield in the protection step and 90% in the hydrogenation step, which are indicative of a robust and scalable process.
- Perform keto alpha-halogenation on Compound B using N-bromosuccinimide in carbon tetrachloride to yield Compound C.
- Execute heating elimination on Compound C with lithium carbonate and lithium bromide in DMF to form Compound D.
- Conduct catalytic hydrogenation on Compound D followed by Robinson cyclization with methyl vinyl ketone to obtain Intermediate F.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this new synthesis route offers profound advantages that directly address the cost and reliability concerns of procurement managers and supply chain heads. The elimination of photochemical steps removes the need for expensive and specialized reactor equipment, which significantly lowers the barrier to entry for contract manufacturing organizations and reduces the capital expenditure required for production scale-up. Furthermore, the use of economically accessible raw materials such as dideoxynol and common reagents like N-bromosuccinimide ensures that the supply chain is not dependent on scarce or volatile commodity markets. This stability is crucial for maintaining continuous production schedules and avoiding delays that can arise from the sourcing of exotic catalysts or precursors. The simplified workup procedures, which often involve standard extraction and chromatography techniques, also reduce the consumption of solvents and energy, contributing to a lower overall cost of goods sold and a smaller environmental footprint for the manufacturing facility.
- Cost Reduction in Manufacturing: The strategic design of this route eliminates the need for expensive transition metal catalysts in several key steps, replacing them with more affordable reagents like copper chloride and organic bases. This substitution drastically reduces the raw material costs associated with the synthesis, as precious metals often require complex recovery and recycling processes that add to the operational overhead. Additionally, the high yields achieved in each step mean that less starting material is wasted, further driving down the cost per kilogram of the final intermediate. The reduction in processing steps also translates to lower labor costs and reduced utility consumption, as fewer reactors are needed to complete the synthesis. These cumulative savings allow for a more competitive pricing structure, enabling suppliers to offer high-purity pharmaceutical intermediates at a price point that supports the economic viability of the final drug product in cost-sensitive markets.
- Enhanced Supply Chain Reliability: By relying on reagents that are commercially available in bulk quantities, such as lithium carbonate and sodium hydroxide, the process mitigates the risk of supply disruptions that can occur with specialized chemicals. The robustness of the reaction conditions, which tolerate a wider range of temperatures and solvent purities, ensures that production can continue even if there are minor fluctuations in raw material quality. This resilience is vital for supply chain heads who must guarantee uninterrupted delivery to pharmaceutical clients facing strict regulatory deadlines. The ability to scale the process from laboratory to multi-ton production without significant re-engineering means that suppliers can respond quickly to increases in demand, providing a reliable source of critical intermediates that supports the global availability of essential hormone therapies.
- Scalability and Environmental Compliance: The avoidance of photochemical reactions and harsh oxidation conditions simplifies the waste management profile of the manufacturing process, making it easier to comply with increasingly stringent environmental regulations. The solvents used in the process, such as ethanol and ethyl acetate, are easier to recover and recycle compared to more hazardous alternatives, reducing the volume of hazardous waste generated. This environmental compatibility not only lowers disposal costs but also enhances the sustainability credentials of the manufacturer, which is becoming a key factor in supplier selection for major pharmaceutical companies. The process is inherently designed for scale-up, with exothermic reactions that can be safely managed in large reactors, ensuring that the transition from pilot plant to commercial production is smooth and predictable without the need for extensive process re-validation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, based on the detailed data provided in the patent documentation. These answers are designed to clarify the operational benefits and technical feasibility for stakeholders evaluating this technology for potential adoption. Understanding these details is essential for making informed decisions about process integration and supply chain partnerships. The information reflects the specific advantages of the new method over prior art, focusing on yield, scalability, and regulatory compliance.
Q: How does this new method improve upon prior art photochemical routes?
A: Unlike prior art requiring complex illumination equipment and suffering from low yields, this method utilizes thermal chemical steps like Robinson cyclization which are significantly easier to scale and control in standard reactor vessels.
Q: What are the key purity advantages of this synthesis route?
A: The route avoids harsh oxidation-reduction cycles that typically generate complex impurity profiles, instead employing selective halogenation and elimination steps that maintain stereochemical integrity and simplify downstream purification.
Q: Is the process suitable for multi-ton commercial manufacturing?
A: Yes, the elimination of photochemical steps and the use of common reagents like NBS and DDQ make the process highly adaptable for large-scale industrial production without specialized infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dydrogesterone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis routes like the one described in CN115894593A to ensure the consistent supply of high-quality pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch of Dydrogesterone intermediate meets the highest global standards. Our infrastructure is designed to handle complex organic syntheses with precision, allowing us to deliver the reliability and quality that top-tier pharmaceutical companies demand for their critical drug pipelines.
We invite you to engage with our technical procurement team to discuss how we can support your specific manufacturing needs with this optimized synthesis route. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how this method can reduce your overall production costs while improving supply security. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements. Our goal is to be your strategic partner in overcoming synthesis challenges and ensuring the uninterrupted availability of essential medical treatments.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
