Industrial Scale Synthesis of Dydrogesterone from Phytosterol Degradation Products
Introduction to Patent CN115160395A
The pharmaceutical industry continuously seeks robust and scalable pathways for the production of critical hormonal therapeutics, and the synthesis of Dydrogesterone represents a significant area of innovation. Patent CN115160395A introduces a groundbreaking methodology that fundamentally shifts the paradigm from traditional photochemical isomerization to a linear, chemically driven synthesis originating from phytosterol degradation products. This novel approach addresses long-standing inefficiencies in steroid manufacturing by leveraging a sequence of high-yielding transformations, including intramolecular esterification, Wittig olefination, and Robinson annulation. For R&D directors and process chemists, this patent offers a compelling alternative to legacy routes that have historically suffered from poor atom economy and hazardous reaction conditions. By utilizing readily available raw materials and avoiding the bottlenecks of light-induced rearrangements, this technology promises to enhance the reliability of the supply chain for this essential progestogen.
The strategic value of this invention lies in its ability to bypass the stereochemical complexities associated with the 19-position methyl configuration inversion, which has traditionally required energy-intensive photochemical reactors. Instead, the disclosed route constructs the necessary molecular architecture through precise carbon-carbon bond-forming reactions and functional group interconversions. This not only simplifies the process control parameters but also significantly reduces the impurity profile typically associated with radical-based photochemical processes. As we delve deeper into the technical specifics, it becomes evident that this method is not merely an incremental improvement but a substantial leap forward in the manufacturing logic of complex steroid intermediates, aligning perfectly with modern green chemistry principles and industrial safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Dydrogesterone has been heavily reliant on photochemical ring-opening and reclosing cycles to achieve the necessary stereochemical inversion at the C19 position. As illustrated in early literature such as Recueil des Travaux Chimiques des Pays-Bas (1961), these methods typically employ 3-hydroxy-pregna-5,7-diene-20-ketone as a starting material, subjecting it to high-pressure mercury lamp irradiation in tetrahydrofuran. While conceptually elegant, these photochemical steps are plagued by notoriously low yields, often reported around 10.6%, due to competing Norrish type reactions and the formation of complex isomeric mixtures. Furthermore, subsequent iterations involving ethylene glycol protection and bromination-debromination sequences, as seen in later patents, introduce additional unit operations that degrade overall process efficiency.
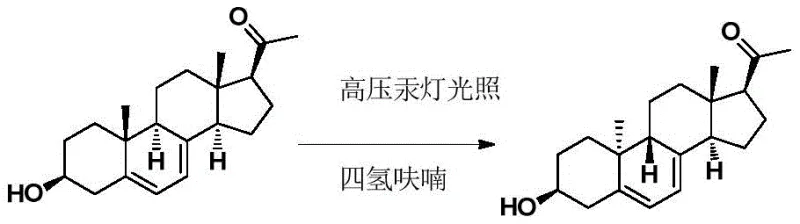
The reliance on high-energy UV irradiation creates significant engineering challenges, including heat management, reactor transparency maintenance, and the handling of large volumes of flammable solvents like THF. The low conversion rates in the critical light step mean that vast quantities of unreacted starting material must be recovered and recycled, increasing both the capital expenditure for separation equipment and the operational costs associated with solvent loss. Additionally, the use of bromine and subsequent debromination steps generates hazardous waste streams that require rigorous environmental controls. These factors collectively render conventional photochemical routes suboptimal for modern, cost-sensitive, and environmentally conscious pharmaceutical manufacturing, creating a pressing need for a more direct and chemically controlled synthetic strategy.
The Novel Approach
In stark contrast to the stochastic nature of photochemistry, the novel route disclosed in CN115160395A employs a deterministic, step-wise construction of the Dydrogesterone skeleton starting from a phytosterol degradation product. This linear synthesis avoids the pitfalls of radical intermediates by utilizing well-established ionic and organometallic reactions. The process initiates with an intramolecular esterification to protect the carbonyl functionality, followed by a Wittig reaction to install the side chain olefin with high regioselectivity. This is succeeded by a Grignard reagent-mediated ring-opening of the lactone, which elegantly sets up the carbon framework for subsequent functionalization without the need for harsh halogenation reagents.
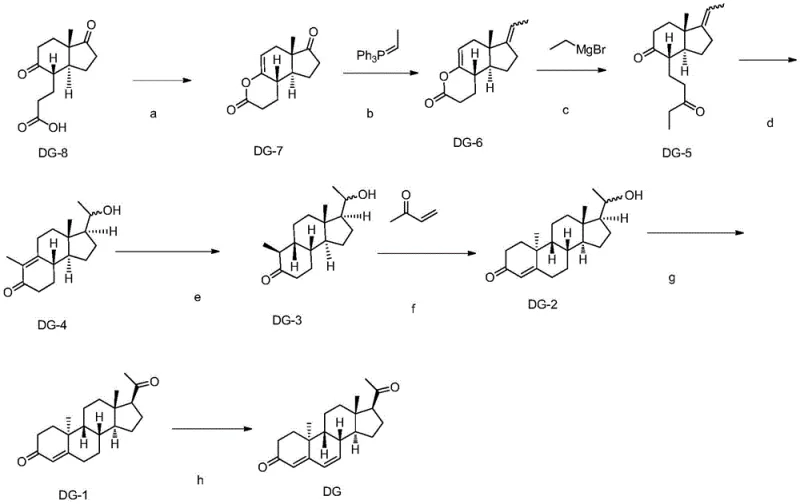
The true brilliance of this approach is exemplified in the later stages, where a Robinson annulation reaction is utilized to construct the A-ring enone system, a structural motif critical for progestogenic activity. By coupling a saturated ketone intermediate with methyl vinyl ketone under basic conditions, the process efficiently builds the six-membered ring with the correct conjugation. The final steps involve mild oxidation and dehydrogenation using reagents like DDQ or chloranil to establish the requisite double bonds. This sequence not only delivers high purity but also operates under much milder thermal conditions compared to the extreme environments of photochemical reactors. The result is a process that is inherently safer, easier to scale, and capable of delivering consistent quality, making it an ideal candidate for reliable API intermediate supplier operations.
Mechanistic Insights into the Synthetic Cascade
The mechanistic elegance of this synthesis lies in the careful orchestration of functional group transformations that maintain stereochemical integrity throughout the cascade. The initial intramolecular esterification of the starting material DG-8 serves a dual purpose: it protects the ketone functionality from unwanted nucleophilic attack in subsequent steps and simultaneously rigidifies the molecular conformation, which can influence the stereoselectivity of downstream reactions. Following this, the Wittig reaction utilizes a phosphorus ylide to convert the carbonyl group into an exocyclic alkene. This step is crucial as it extends the carbon chain necessary for the eventual formation of the C17 side chain, doing so with high fidelity and minimal byproduct formation compared to older alkylation methods.
Subsequent to the olefin construction, the Grignard addition plays a pivotal role in opening the lactone ring. The nucleophilic attack by the ethyl magnesium bromide on the carbonyl carbon of the lactone results in ring cleavage, generating a keto-alcohol intermediate that is primed for the hydroboration step. The hydroboration-oxidation sequence is particularly noteworthy for its anti-Markovnikov selectivity, ensuring that the hydroxyl group is installed at the less substituted carbon, which is essential for the correct positioning of oxygen functionality in the final molecule. This is followed by a catalytic hydrogenation step using Raney-Ni, which selectively reduces the double bond formed during the earlier stages without affecting other sensitive functionalities, thereby setting the stage for the ring-closing metathesis-like behavior of the Robinson annulation.
The Robinson annulation itself is a tandem Michael addition-aldol condensation sequence that forms the A-ring. The base-catalyzed addition of the enolate derived from the steroid nucleus to methyl vinyl ketone creates a new carbon-carbon bond, followed by an intramolecular aldol condensation and dehydration to yield the conjugated enone system. Finally, the introduction of unsaturation in the B-ring via dehydrogenation with DDQ or tetrachlorobenzoquinone completes the structural requirements of Dydrogesterone. Each step in this mechanism is designed to maximize yield and minimize impurity generation, ensuring that the final product meets the stringent purity specifications required for pharmaceutical applications. This level of mechanistic control is what distinguishes this patent from prior art, offering a clear path to high-purity Dydrogesterone.
How to Synthesize Dydrogesterone Efficiently
Implementing this synthesis requires precise control over reaction parameters, particularly temperature and stoichiometry, to ensure optimal yields at each stage. The process begins with the reflux of the starting material in acetic anhydride with sodium acetate, a straightforward operation that sets the foundation for the entire sequence. As the synthesis progresses through the Wittig and Grignard steps, strict anhydrous conditions and temperature control (e.g., maintaining 0°C during Grignard addition) are paramount to prevent side reactions. The subsequent hydroboration and hydrogenation steps utilize common catalysts and reagents, making them accessible for standard chemical processing facilities. For a detailed breakdown of the specific operational parameters, reagent ratios, and workup procedures for each transformation, please refer to the standardized synthesis guide below.
- Perform intramolecular esterification of DG-8 using acetic anhydride and sodium acetate to form the ketal intermediate DG-7.
- Execute a Wittig reaction on DG-7 to construct the side chain olefin, followed by Grignard reagent ring-opening to yield DG-5.
- Conduct hydroboration and oxidation to form ketene DG-4, followed by hydrogenation to saturate the double bond yielding DG-3.
- Complete the synthesis via Robinson annulation with methyl vinyl ketone to form DG-2, followed by oxidation and final dehydrogenation to obtain Dydrogesterone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers transformative benefits that directly impact the bottom line and operational resilience. The primary advantage stems from the elimination of the photochemical isomerization step, which has historically been a major bottleneck in Dydrogesterone production. By replacing this low-yield, high-energy process with a series of high-yielding thermal reactions, manufacturers can drastically simplify their production infrastructure. There is no longer a need for specialized photochemical reactors or the extensive safety protocols associated with high-pressure mercury lamps and large volumes of volatile solvents. This simplification translates directly into reduced capital expenditure and lower operational overheads, making the manufacturing process more economically viable.
- Cost Reduction in Manufacturing: The economic implications of this route are profound, primarily driven by the significant improvement in overall yield and the use of inexpensive, readily available raw materials. Unlike traditional routes that suffer from cumulative yield losses due to multiple protection-deprotection cycles and inefficient photo-steps, this linear pathway maintains high mass efficiency throughout. The avoidance of expensive halogenating agents like bromine and the reduction in solvent consumption further contribute to substantial cost savings. Additionally, the simplified purification requirements resulting from fewer isomeric byproducts reduce the burden on downstream processing, lowering the cost of goods sold (COGS) and enhancing profit margins for the final API.
- Enhanced Supply Chain Reliability: Supply chain stability is critically dependent on the availability of raw materials and the robustness of the manufacturing process. This method utilizes phytosterol degradation products as the starting point, which are abundant byproducts of the vegetable oil industry, ensuring a stable and sustainable feedstock supply. Unlike specialized photochemical precursors that may have limited suppliers, these degradation products are commoditized, reducing the risk of supply disruptions. Furthermore, the use of standard chemical transformations means that the process can be easily transferred between different manufacturing sites or scaled up rapidly to meet surges in demand, providing a buffer against market volatility and ensuring continuous availability for downstream drug formulation.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden complexities, but this route is inherently designed for industrial magnification. The reactions involved, such as esterification, hydrogenation, and Robinson annulation, are well-understood unit operations that scale predictably from pilot plants to multi-ton reactors. Moreover, the environmental footprint of this process is significantly smaller than that of conventional methods. By eliminating bromination steps and reducing solvent usage, the generation of hazardous waste is minimized, simplifying compliance with increasingly stringent environmental regulations. This 'green' profile not only reduces waste disposal costs but also aligns with the sustainability goals of modern pharmaceutical companies, enhancing the brand value of the supply chain partners.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is essential for stakeholders evaluating its potential for integration into their supply chains. The following questions address common inquiries regarding the process efficiency, raw material sourcing, and scalability, drawing directly from the technical disclosures and comparative data provided in the patent literature. These insights are intended to clarify the operational advantages and assist in decision-making processes regarding the adoption of this technology for commercial production.
Q: What are the primary advantages of this new Dydrogesterone synthesis route over traditional photochemical methods?
A: The new route eliminates the reliance on low-yield photochemical isomerization steps (often <22% yield) and avoids complex protection/deprotection sequences involving bromination. It utilizes easily available phytosterol degradation products as starting materials, offering higher overall conversion rates and milder reaction conditions suitable for industrial scaling.
Q: How does the Robinson annulation step contribute to the structural integrity of the final API?
A: The Robinson annulation reaction (Step F) is critical for constructing the A-ring enone system characteristic of progestogens. By reacting the saturated ketone intermediate DG-3 with methyl vinyl ketone under basic conditions, the process efficiently builds the six-membered ring with the correct conjugation, ensuring the biological activity required for the final Dydrogesterone molecule.
Q: Is this synthesis method scalable for commercial production of pharmaceutical intermediates?
A: Yes, the method is specifically designed for industrial production. It replaces hazardous photochemical steps and difficult-to-control bromination reactions with standard organic transformations like esterification, Grignard addition, and catalytic hydrogenation. These steps are well-understood in chemical engineering, allowing for robust scale-up from pilot plant to multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dydrogesterone Supplier
The technological advancements detailed in patent CN115160395A represent a significant opportunity for the pharmaceutical industry to secure a more stable and cost-effective supply of Dydrogesterone. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this innovative laboratory-scale chemistry into robust commercial reality. Our team of process chemists is adept at optimizing complex steroid syntheses, ensuring that the transition from pilot scale to full-scale production is seamless and efficient. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, coupled with stringent purity specifications and rigorous QC labs that guarantee every batch meets the highest international standards.
We invite procurement leaders and R&D directors to collaborate with us to leverage this superior synthesis route for your Dydrogesterone requirements. By partnering with us, you gain access to a supply chain that is not only cost-competitive but also resilient and environmentally responsible. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume needs. Let us provide you with specific COA data and route feasibility assessments to demonstrate how we can drive value and efficiency in your Dydrogesterone supply chain today.
