Scalable Low-Dimer Synthesis of Aripiprazole Intermediates via Zinc Chloride Catalysis
The pharmaceutical landscape for atypical antipsychotics has been significantly shaped by the commercial success of Aripiprazole, a dopamine-serotonin system stabilizer approved for the treatment of schizophrenia. As demand for this high-value active pharmaceutical ingredient continues to grow, the efficiency of its supply chain relies heavily on the optimization of intermediate synthesis. Patent CN103787965A introduces a transformative methodology that addresses the longstanding challenge of dimer impurity formation during the alkylation of 7-hydroxyquinolinone. By shifting away from traditional dibromobutane reagents and leveraging a zinc chloride catalyzed ring-opening of tetrahydrofuran, this technology offers a pathway to higher purity intermediates with drastically simplified downstream processing. For procurement and R&D leaders, this represents a critical opportunity to enhance supply security while mitigating the risks associated with complex impurity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Aripiprazole has relied heavily on the alkylation of 7-hydroxyquinolinone using 1,4-dibromobutane. While conceptually straightforward, this approach suffers from a fundamental lack of chemoselectivity, as the dibromide possesses two reactive leaving groups of similar activity. Consequently, the reaction inevitably produces significant quantities of the quinolinone dimer impurity, often exceeding 10% of the crude mixture. This dimer, structurally characterized by two quinolinone units linked by a butylene chain, is notoriously difficult to separate from the desired mono-alkylated intermediate due to similar physicochemical properties. The removal process typically necessitates extensive chromatography or repeated recrystallizations using large volumes of volatile solvents like toluene, which not only depresses overall yield to as low as 39% but also creates substantial environmental and safety burdens.
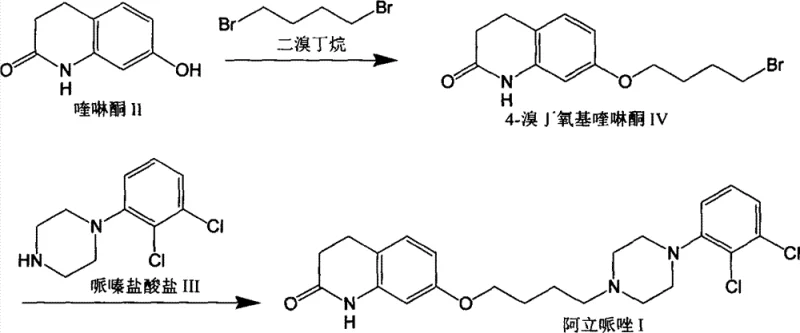
The Novel Approach
The patented innovation circumvents these selectivity issues by employing a strategic ring-opening of tetrahydrofuran (THF) with p-toluenesulfonyl chloride. This reaction, catalyzed by zinc chloride, generates 4-chlorobutyl paratoluenesulfonate, an asymmetric electrophile where the tosylate group is a far superior leaving group compared to the terminal chloride. This differentiation in leaving group ability ensures that the subsequent etherification with 7-hydroxyquinolinone occurs exclusively at the tosylate end, effectively suppressing the formation of the dimer impurity at the source. By preventing the double alkylation event entirely, the process yields an intermediate with exceptional purity, eliminating the need for the tedious and yield-eroding purification steps required by legacy methods. This streamlined approach not only improves the quality of the final API but also significantly enhances the throughput of the manufacturing facility.
Mechanistic Insights into Zinc Chloride Catalyzed Etherification
The core of this synthetic advancement lies in the Lewis acid catalyzed ring opening of the cyclic ether. Zinc chloride acts as a potent Lewis acid, coordinating with the oxygen atom of the tetrahydrofuran ring to increase its electrophilicity and facilitate nucleophilic attack by the chloride ion derived from the sulfonyl chloride or added salts. This results in the cleavage of the C-O bond and the formation of the linear 4-chlorobutyl chain bearing a tosylate moiety. The mechanistic precision here is vital; unlike the statistical distribution of products seen with symmetrical dihalides, this ring-opening strategy provides a single, well-defined reactive species. The subsequent reaction with 7-hydroxyquinolinone proceeds via a standard SN2 mechanism where the phenoxide anion attacks the carbon bearing the tosylate group. Because the terminal chloride is a poor leaving group under these specific mild conditions, further alkylation is kinetically inhibited, ensuring the reaction stops cleanly at the mono-substituted stage.
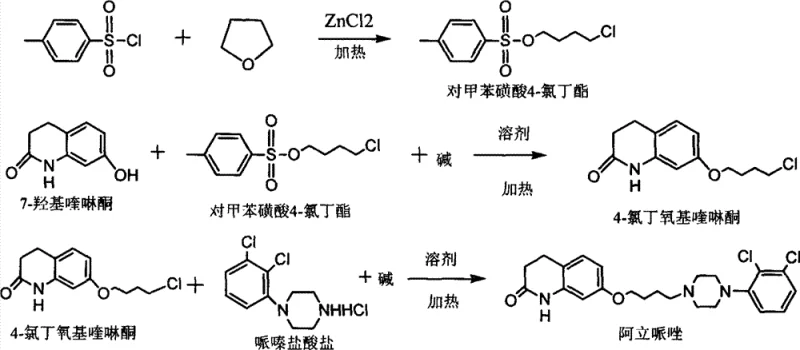
Furthermore, the control over impurity generation extends to the final coupling step with piperazine. In conventional routes, residual dimer impurities from the first step carry through to the final amination, contaminating the final API and requiring rigorous final purification. In this novel pathway, the high purity of the 4-chlorobutoxyquinolinone intermediate ensures that the final nucleophilic substitution with piperazine hydrochloride proceeds with minimal side reactions. The result is a final product with dimer content reduced to less than 0.05%, a specification that is difficult to achieve consistently with dibromobutane-based chemistry. This level of control over the impurity profile is critical for regulatory compliance and reduces the burden on quality control laboratories, allowing for faster batch release times.
How to Synthesize Aripiprazole Efficiently
The implementation of this synthesis route requires careful attention to the stoichiometry of the ring-opening step and the selection of appropriate bases for the etherification. The process begins with the activation of THF, followed by a controlled addition of the sulfonyl chloride to manage exotherms. Detailed operational parameters, including specific temperature ramps and workup procedures to isolate the tosylate intermediate, are essential for reproducibility. For a comprehensive breakdown of the reaction conditions, molar ratios, and isolation techniques required to achieve the reported high yields and purity, please refer to the standardized synthesis guide below.
- React tetrahydrofuran with p-toluenesulfonyl chloride under zinc chloride catalysis at 50-120°C to obtain 4-chlorobutyl paratoluenesulfonate.
- Condense the resulting tosylate ester with 7-hydroxyquinolinone in the presence of a base and solvent to generate 4-chlorobutoxyquinolinone.
- Perform nucleophilic substitution of the chlorobutoxy intermediate with piperazine hydrochloride in alkaline conditions to yield high-purity Aripiprazole.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the shift to a THF-based synthesis strategy offers profound advantages in terms of cost structure and supply chain resilience. The reliance on commodity chemicals rather than specialized, high-cost halogenated intermediates decouples the production cost from the volatility of niche reagent markets. Additionally, the drastic reduction in purification complexity translates directly into lower operational expenditures, as less solvent is consumed and less waste is generated per kilogram of product. This efficiency gain allows manufacturers to offer more competitive pricing while maintaining healthy margins, a critical factor in the highly price-sensitive generic pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of expensive reagents like 1-chloro-4-bromobutane, which are prone to instability and high procurement costs, results in a direct reduction in raw material expenses. Moreover, by avoiding the formation of difficult-to-remove dimer impurities, the process removes the need for costly chromatographic purification or multiple recrystallization cycles. This simplification of the downstream processing significantly lowers utility consumption, solvent recovery costs, and labor hours, leading to a substantially more economical manufacturing process overall.
- Enhanced Supply Chain Reliability: Tetrahydrofuran and p-toluenesulfonyl chloride are bulk commodities produced by numerous global suppliers, ensuring a stable and continuous supply of starting materials. Unlike specialized brominated intermediates which may have limited sources and long lead times, the raw materials for this route are readily available in multi-ton quantities. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages, providing procurement managers with greater flexibility and security in their sourcing strategies.
- Scalability and Environmental Compliance: The process operates under mild thermal conditions and utilizes common organic solvents that are easily recovered and recycled, aligning with modern green chemistry principles. The absence of heavy metal catalysts or hazardous bromine reagents simplifies waste treatment and reduces the environmental footprint of the manufacturing site. This inherent safety and environmental compatibility facilitates easier regulatory approval for new manufacturing sites and supports the sustainable scale-up of production capacity to meet growing global demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis pathway. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the operational benefits and chemical rationale behind the technology.
Q: How does the THF-based route reduce dimer impurities compared to dibromobutane methods?
A: Traditional methods using 1,4-dibromobutane suffer from non-selective alkylation at both bromine sites, leading to dimer formation greater than 10%. The patented THF route utilizes a ring-opening strategy that generates a mono-functionalized electrophile (4-chlorobutyl tosylate), effectively preventing the double alkylation that causes dimerization, thereby reducing dimer content to below 0.05% in the final product.
Q: What are the cost advantages of using tetrahydrofuran over 1-chloro-4-bromobutane?
A: Tetrahydrofuran (THF) is a widely available commodity chemical with significantly lower procurement costs compared to specialized halogenated derivatives like 1-chloro-4-bromobutane or 4-bromo-1-butanol. Furthermore, the elimination of tedious purification steps required to remove dimers drastically reduces solvent consumption and processing time, leading to substantial overall manufacturing cost savings.
Q: Is the zinc chloride catalyzed ring opening safe for industrial scale-up?
A: Yes, the process operates under mild conditions (50-120°C) and avoids the use of unstable reagents like 4-bromo-1-butanol, which can cyclize back to THF or decompose. The use of common solvents like toluene and standard inorganic bases ensures that the reaction profile is robust and safe for large-scale commercial production without requiring exotic high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aripiprazole Supplier
The transition to this advanced THF-based synthesis route exemplifies the kind of process innovation that drives value in the pharmaceutical intermediate sector. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such technologies to market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of Aripiprazole intermediate meets the highest international standards for safety and efficacy.
We invite forward-thinking procurement directors and supply chain heads to collaborate with us on optimizing their API supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing processes can enhance your product's competitiveness in the global marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
