Advanced Lewis Acid-Catalyzed Synthesis of Polyene Esters for Commercial Carotenoid Production
Advanced Lewis Acid-Catalyzed Synthesis of Polyene Esters for Commercial Carotenoid Production
The chemical landscape for synthesizing high-value carotenoids and polyene esters has historically been dominated by phosphorus-based methodologies, yet Patent CN1169418A introduces a transformative approach that leverages Lewis acid-catalyzed condensation to achieve superior efficiency and purity. This groundbreaking technology replaces traditional Wittig or Horner reactions with a streamlined process involving the reaction of acetalized polyenals with vinyl ketene acetals, fundamentally altering the impurity profile and economic viability of producing compounds like beta-apocarotenoids and crocetin derivatives. By utilizing specific Lewis acids such as zinc chloride or boron trifluoride etherate, the process ensures high gamma-selectivity, effectively suppressing the formation of telomers that have plagued previous synthetic attempts in this domain. The strategic implementation of this method allows for the precise construction of conjugated polyene chains essential for food colorants and pharmaceutical intermediates without the burden of heavy metal or phosphine waste. 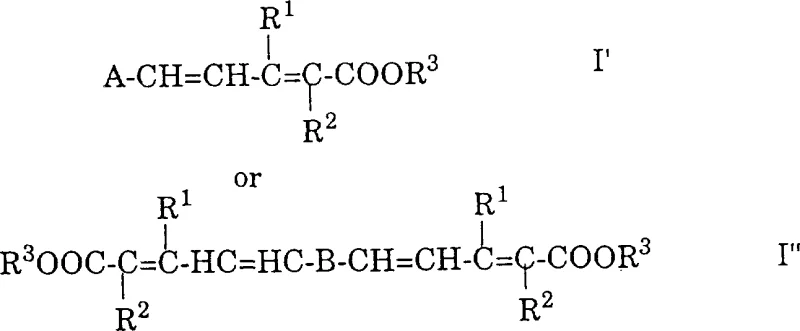 This innovation represents a significant leap forward for manufacturers seeking a reliable carotenoid intermediate supplier, as it directly addresses the critical pain points of waste management and downstream purification complexity inherent in older technologies.
This innovation represents a significant leap forward for manufacturers seeking a reliable carotenoid intermediate supplier, as it directly addresses the critical pain points of waste management and downstream purification complexity inherent in older technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for extending polyene chains, particularly the Wittig and Horner-Wadsworth-Emmons reactions, have long been the industry standard but suffer from severe drawbacks that impact both operational expenditure and environmental compliance. These classical methods invariably generate stoichiometric amounts of phosphine oxides as byproducts, which are notoriously difficult to separate from the desired polyene products due to similar polarity and solubility characteristics. The removal of these phosphorus residues often requires extensive chromatography or multiple recrystallization steps, leading to substantial yield losses and increased solvent consumption that drives up the overall cost of goods sold. Furthermore, the reagents required for these transformations, such as phosphonium salts and strong bases, can be hazardous to handle on a large scale and contribute to a higher E-factor for the manufacturing process. For procurement managers focused on cost reduction in fine chemical manufacturing, the disposal costs associated with phosphorus waste and the low atom economy of these reactions present a persistent financial drain that erodes profit margins over time.
The Novel Approach
In stark contrast, the methodology disclosed in Patent CN1169418A utilizes a Lewis acid-catalyzed condensation between polyene O,O-dialkyl acetals and vinyl ketene acetals to construct the carbon skeleton with remarkable precision and cleanliness. This novel approach operates under mild conditions, typically between -20°C and room temperature, using catalytic amounts of Lewis acids like zinc chloride or titanium tetrachloride to activate the acetal electrophile. The reaction proceeds with exclusive gamma-selectivity, meaning the carbon chain extension occurs specifically at the desired position without generating the complex mixtures of isomers often seen in free-radical or uncatalyzed condensations. Following the initial coupling, a simple base-induced elimination step removes the alkoxy group to restore the conjugated double bond system, yielding the final polyene ester or acid in high purity. This pathway eliminates the need for phosphorus reagents entirely, thereby removing the associated purification bottlenecks and allowing for a more direct isolation of the product, often through simple crystallization from alcohols like ethanol or methanol.
Mechanistic Insights into Lewis Acid-Catalyzed Acetal Condensation
The core mechanistic advantage of this process lies in the activation of the polyene acetal by the Lewis acid catalyst, which generates a highly reactive oxocarbenium ion intermediate that is susceptible to nucleophilic attack by the vinyl ketene acetal. Unlike previous attempts using dienol ethers which resulted in uncontrolled polymerization or telomerization, the specific electronic properties of the vinyl ketene acetal ensure that the reaction terminates after a single addition step. The vinyl ketene acetal acts as a masked enolate equivalent, attacking the activated acetal at the gamma-position to form a saturated delta-alkoxy intermediate, which is a key structural feature distinguishing this method from direct olefination techniques. 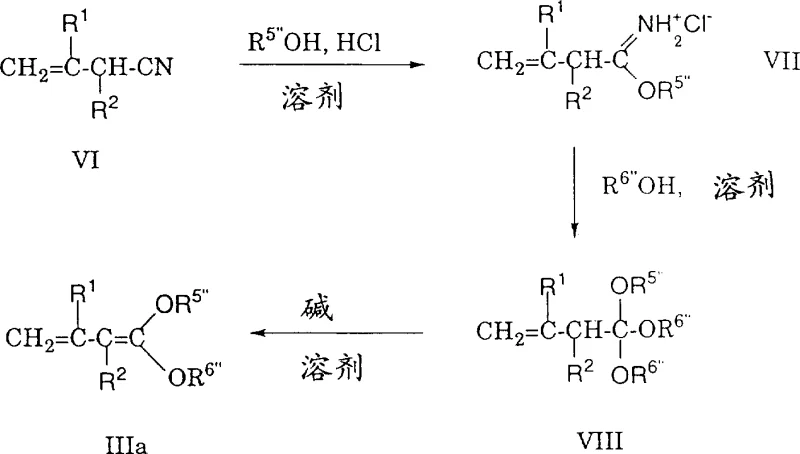 The subsequent hydrolysis and base treatment facilitate the elimination of the alkoxy group, driven by the thermodynamic stability of the newly formed conjugated polyene system. This two-step sequence (condensation followed by elimination) provides exceptional control over the stereochemistry of the double bonds, predominantly yielding the all-E isomer which is critical for the biological activity and color properties of carotenoids. The ability to tune the reaction outcome by selecting different vinyl ketene acetals (e.g., those with silyl groups or cyclic acetals) offers chemists a versatile toolkit for synthesizing a wide array of polyene derivatives including apocarotenoids and symmetrical diesters like crocetin.
The subsequent hydrolysis and base treatment facilitate the elimination of the alkoxy group, driven by the thermodynamic stability of the newly formed conjugated polyene system. This two-step sequence (condensation followed by elimination) provides exceptional control over the stereochemistry of the double bonds, predominantly yielding the all-E isomer which is critical for the biological activity and color properties of carotenoids. The ability to tune the reaction outcome by selecting different vinyl ketene acetals (e.g., those with silyl groups or cyclic acetals) offers chemists a versatile toolkit for synthesizing a wide array of polyene derivatives including apocarotenoids and symmetrical diesters like crocetin.
Impurity control is inherently built into this mechanism because the reaction conditions do not support the propagation of chain growth beyond the intended single coupling event. The use of protected hydroxyl groups on the cyclohexene ring, such as methoxy or silyl ethers, remains stable under the Lewis acid conditions employed, allowing for the synthesis of functionalized carotenoids without the need for orthogonal protection strategies that add synthetic steps. Furthermore, the intermediate delta-alkoxy esters can often be isolated and characterized, providing a valuable checkpoint for quality control before the final elimination step. This level of mechanistic transparency is invaluable for R&D directors who require robust analytical data to support regulatory filings for food additives or pharmaceutical ingredients. The process minimizes the formation of Z-isomers and other geometric impurities, which are common contaminants in thermal isomerization processes, thus ensuring a final product specification that meets the stringent requirements of global markets for high-purity carotenoid intermediates.
How to Synthesize Polyene Esters Efficiently
The practical execution of this synthesis begins with the preparation of the vinyl ketene acetal reagent, which can be derived from readily available nitriles through a sequence involving iminoalkyl ester formation and subsequent elimination. 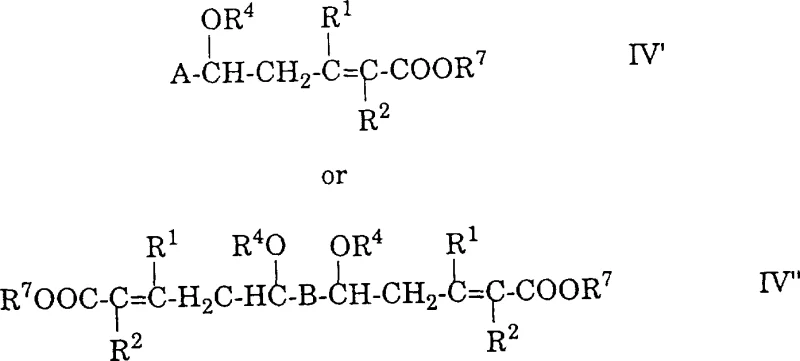 Once the reagents are prepared, the polyene acetal is dissolved in an aprotic solvent such as tert-butyl methyl ether or dichloromethane and cooled to temperatures around -20°C to 0°C before the addition of the Lewis acid catalyst. The reaction mixture is then allowed to warm to room temperature or slightly elevated temperatures up to 50°C to drive the condensation to completion, monitored by TLC or HPLC to ensure full conversion of the starting acetal. After the coupling phase, the reaction is quenched with water or a weak acid solution to hydrolyze any remaining silyl groups or orthoester intermediates, followed by extraction and concentration to isolate the crude delta-alkoxy intermediate. The detailed standardized synthesis steps for optimizing yield and purity in this specific transformation are outlined below.
Once the reagents are prepared, the polyene acetal is dissolved in an aprotic solvent such as tert-butyl methyl ether or dichloromethane and cooled to temperatures around -20°C to 0°C before the addition of the Lewis acid catalyst. The reaction mixture is then allowed to warm to room temperature or slightly elevated temperatures up to 50°C to drive the condensation to completion, monitored by TLC or HPLC to ensure full conversion of the starting acetal. After the coupling phase, the reaction is quenched with water or a weak acid solution to hydrolyze any remaining silyl groups or orthoester intermediates, followed by extraction and concentration to isolate the crude delta-alkoxy intermediate. The detailed standardized synthesis steps for optimizing yield and purity in this specific transformation are outlined below.
- Prepare the vinyl ketene acetal reagent (Formula III) from the corresponding nitrile via iminoalkyl ester and orthoester intermediates, ensuring high purity for optimal coupling.
- React the polyene O,O-dialkyl acetal (Formula II) with the vinyl ketene acetal in the presence of a Lewis acid catalyst like zinc chloride or boron trifluoride etherate at temperatures between -20°C and room temperature.
- Hydrolyze the reaction mixture if necessary, then treat the resulting delta-alkoxy intermediate with a strong base such as sodium alkoxide to eliminate alcohol and form the conjugated polyene ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Lewis acid-catalyzed technology offers profound advantages that extend far beyond the laboratory bench, directly impacting the bottom line through reduced raw material costs and simplified logistics. The elimination of phosphorus-based reagents means that manufacturers no longer need to source expensive and potentially regulated phosphonium salts, replacing them with more commodity-grade alcohols and nitriles that are widely available in the global chemical market. This shift significantly de-risks the supply chain by reducing dependency on specialized reagent suppliers and mitigating the volatility associated with the pricing of organophosphorus compounds. Additionally, the simplified workup procedure, which often relies on crystallization rather than column chromatography, drastically reduces solvent consumption and waste disposal fees, contributing to a more sustainable and cost-effective manufacturing operation. The ability to produce high-purity intermediates with fewer processing steps translates to shorter cycle times and increased throughput capacity, allowing suppliers to respond more agilely to fluctuating market demands for carotenoid colorants and nutritional supplements.
- Cost Reduction in Manufacturing: The removal of stoichiometric phosphine oxide byproducts eliminates the need for costly and time-consuming purification steps such as silica gel chromatography, which is a major driver of operational expenses in traditional polyene synthesis. By relying on crystallization and simple aqueous washes for purification, the process significantly lowers the consumption of high-purity solvents and stationary phases, resulting in substantial cost savings per kilogram of finished product. Furthermore, the catalytic nature of the Lewis acid means that only small quantities of expensive metals like zinc or titanium are required, unlike the stoichiometric amounts of base and phosphorus reagents needed in Wittig chemistry. This improvement in atom economy ensures that a higher percentage of the raw material mass ends up in the final product, maximizing the return on investment for every ton of feedstock purchased by the procurement team.
- Enhanced Supply Chain Reliability: The starting materials for this process, including polyene aldehydes and simple nitriles, are established commodities with robust global supply networks, ensuring consistent availability and reducing the risk of production stoppages due to raw material shortages. The mild reaction conditions, which do not require cryogenic temperatures below -40°C or high-pressure equipment, allow the synthesis to be performed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. This compatibility with existing infrastructure means that technology transfer and scale-up can be achieved rapidly without the need for significant capital expenditure on specialized hardware. Consequently, supply chain leaders can secure longer-term contracts with greater confidence, knowing that the manufacturing process is resilient to external shocks and capable of maintaining continuous production schedules to meet delivery deadlines.
- Scalability and Environmental Compliance: As regulatory pressures regarding chemical waste and environmental impact intensify globally, this phosphorus-free synthesis route positions manufacturers favorably for compliance with strict environmental standards such as REACH and EPA guidelines. The absence of phosphorus waste streams simplifies wastewater treatment protocols and reduces the liability associated with hazardous waste disposal, making the facility more attractive to investors and partners who prioritize ESG (Environmental, Social, and Governance) criteria. The process is inherently scalable, as demonstrated by the patent examples which show successful execution from gram to multi-gram scales with consistent yields and purity profiles. This scalability ensures that the technology can support commercial production volumes ranging from hundreds of kilograms to multi-ton annual capacities, providing a stable foundation for long-term business growth and market expansion in the competitive fine chemicals sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Lewis acid-catalyzed polyene synthesis technology, drawing directly from the experimental data and claims within Patent CN1169418A. These insights are designed to clarify the operational parameters and quality outcomes expected when transitioning from conventional phosphorus-based methods to this advanced acetal condensation strategy. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios for carotenoid and vitamin production. The answers reflect the specific reaction conditions, selectivity profiles, and purification advantages documented in the patent literature.
Q: What is the primary advantage of this Lewis acid method over traditional Wittig reactions?
A: The primary advantage is the elimination of phosphorus-containing waste products like triphenylphosphine oxide, which simplifies purification and significantly reduces environmental disposal costs while improving atom economy.
Q: How does the process ensure gamma-selectivity during the condensation step?
A: The use of specific vinyl ketene acetals in the presence of Lewis acids directs the nucleophilic attack exclusively to the gamma-position of the diene system, preventing the formation of unwanted telomers or alpha-coupled byproducts.
Q: Can this method be scaled for industrial production of carotenoids?
A: Yes, the reaction conditions utilize mild temperatures ranging from -20°C to 50°C and common solvents like tert-butyl methyl ether or ethanol, making the process highly amenable to large-scale commercial manufacturing with robust safety profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyene Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Lewis acid-catalyzed synthesis described in Patent CN1169418A and have integrated these advanced methodologies into our state-of-the-art production facilities to serve the global demand for high-performance carotenoid intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this technology are fully realized in large-scale manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs equipped with HPLC and UV-Vis spectroscopy to guarantee that every batch of polyene ester or acid meets the exacting standards required for food, feed, and pharmaceutical applications. Our commitment to quality is matched by our dedication to process safety and environmental stewardship, making us a preferred partner for multinational corporations seeking a reliable polyene esters supplier who can deliver consistency and compliance.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific product requirements and volume needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to this phosphorus-free technology for your supply chain. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments that demonstrate our capability to produce complex polyene structures with superior efficiency. Let us collaborate to optimize your sourcing strategy and secure a competitive edge in the market through the adoption of cutting-edge chemical manufacturing technologies that define the future of the industry.
