Advanced Lewis Acid Catalysis For Commercial Scale Polyene Ester Production
The chemical industry continuously seeks more efficient pathways for synthesizing complex polyene structures, particularly those serving as critical intermediates for carotenoids and vitamin derivatives. Patent CN1124261C introduces a transformative methodology for the manufacture of polyene esters and acids, moving away from traditional phosphorus-based coupling reactions. This innovation leverages a Lewis acid-catalyzed condensation between acetalized polyenals and vinyl ketene acetals, followed by a strategic hydrolysis and base-induced elimination sequence. The significance of this patent lies in its ability to construct conjugated systems with high stereoselectivity while bypassing the environmental and purification burdens associated with Wittig or Horner-Wadsworth-Emmons reactions. For R&D directors and procurement specialists, understanding this shift is crucial for evaluating long-term supply chain stability and cost structures in fine chemical manufacturing. The process allows for the precise assembly of carbon chains essential for high-value colorants used in food, animal nutrition, and pharmaceutical applications.
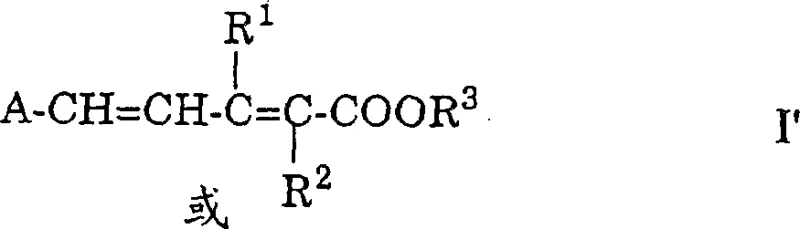
Traditional methods for extending polyene chains have long relied on organophosphorus chemistry, which, while effective, introduces significant downstream processing challenges. The limitations of conventional Wittig reactions include the stoichiometric generation of phosphine oxides, which are notoriously difficult to separate from the desired product without extensive chromatography or recrystallization steps. Furthermore, the reagents required for these traditional pathways often suffer from stability issues and high costs, impacting the overall economic viability of large-scale production. In contrast, the novel approach detailed in the patent utilizes readily available vinyl ketene acetals and polyene acetals. This shift not only simplifies the reagent profile but also fundamentally changes the impurity landscape of the reaction. By avoiding phosphorus entirely, the process reduces the load on waste treatment facilities and minimizes the risk of heavy metal or toxic residue contamination in the final active pharmaceutical ingredients or food additives.
The novel approach described in the patent achieves chain extension through a highly specific gamma-position coupling, a selectivity that was historically difficult to maintain without forming telomers or unwanted alpha-products. Previous attempts to couple acetals with dienol ethers often resulted in mixtures that required complex separation techniques, rendering them unsuitable for industrial synthesis. The use of vinyl ketene acetals, particularly silylated analogues, overcomes these historical barriers by directing the reaction exclusively to the desired position. This specificity is critical for maintaining the integrity of the conjugated system, which dictates the optical and biological properties of the final carotenoid. The subsequent base-induced elimination of the alkoxy group restores the conjugation without the need for additional oxidation or dehydration steps, streamlining the synthetic route. This efficiency translates directly into reduced processing time and lower energy consumption per kilogram of product manufactured.
Mechanistic Insights into Lewis Acid-Catalyzed Condensation and Elimination
The core mechanism involves the activation of the polyene acetal by a Lewis acid, such as zinc chloride, titanium tetrachloride, or boron trifluoride etherate, which generates a reactive carbocation intermediate. This intermediate is then attacked by the vinyl ketene acetal at the gamma-position, forming a new carbon-carbon bond while preserving the stereochemistry of the existing double bonds. The reaction conditions are meticulously controlled, typically ranging from -40°C to room temperature, to prevent polymerization or isomerization of the sensitive polyene chain. Following the coupling, the intermediate undergoes hydrolysis to reveal a carbonyl or ester functionality, depending on the specific vinyl ketene acetal used. The final and perhaps most critical step is the treatment with a strong base, such as sodium methoxide or potassium tert-butoxide. This step cleaves the delta-alkoxy group, triggering an elimination reaction that establishes the final conjugated double bond. Remarkably, this base-catalyzed elimination favors the thermodynamically stable all-E configuration, ensuring high purity without the need for isomer separation.
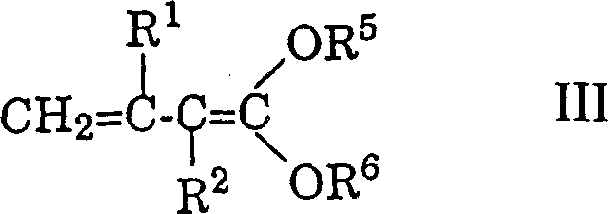
Impurity control is inherently built into this mechanistic pathway through the use of crystallization rather than chromatography for purification. The patent examples demonstrate that the intermediate acetals and final esters can often be isolated as crystalline solids directly from the reaction mixture or after simple solvent exchanges. This physical property is a direct result of the high regioselectivity of the coupling reaction, which minimizes the formation of structural isomers that would otherwise depress the melting point and hinder crystallization. For quality control teams, this means that standard analytical techniques like HPLC and UV-Vis spectroscopy can confirm purity levels exceeding 95% without complex sample preparation. The avoidance of phosphorus by-products also means that ICP-MS testing for residual metals is simplified, as the primary concern shifts to the removal of the Lewis acid catalyst, which can be achieved through standard aqueous workups. This robust impurity profile is essential for meeting the stringent regulatory requirements of the food and pharmaceutical industries.
How to Synthesize Polyene Esters Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for reproducing these high-value intermediates in a laboratory or pilot plant setting. The process begins with the preparation of the polyene acetal educts, which can be synthesized from corresponding aldehydes using trialkyl orthoformates under acidic conditions. These acetals are stable and can be stored, allowing for flexible production scheduling. The coupling reaction is then performed in aprotic solvents like tert-butyl methyl ether or toluene, with careful addition of the Lewis acid catalyst to control exotherms. Following the coupling, the reaction mixture is quenched and hydrolyzed, often in the same pot, to generate the beta-alkoxy ester intermediate. The final elimination step requires the use of anhydrous alcoholic solvents and alkali metal alkoxides, with temperature control being paramount to prevent degradation of the polyene chain. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- React polyene O,O-dialkylacetal with vinyl ketene acetal in the presence of a Lewis acid catalyst such as zinc chloride or boron trifluoride etherate.
- Hydrolyze the reaction mixture using aqueous acid or water depending on the specific vinyl ketene acetal analogue employed.
- Cleave the delta-position alkoxy group under strongly basic conditions using alkali metal alcoholates to form the final conjugated polyene ester.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthetic route offers substantial strategic advantages regarding raw material sourcing and cost management. The key reagents, including polyene acetals and vinyl ketene acetals, are derived from commodity chemicals that are widely available in the global market, reducing the risk of supply bottlenecks associated with specialized organometallic reagents. The elimination of phosphorus-based chemistry removes the need for expensive phosphonium salts and the associated waste disposal costs, leading to a leaner cost structure. Furthermore, the ability to purify products via crystallization rather than column chromatography significantly reduces the consumption of silica gel and organic solvents, which are major cost drivers in fine chemical manufacturing. These factors combine to create a more resilient supply chain that is less susceptible to fluctuations in the price of specialized reagents or regulatory changes regarding hazardous waste.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the simplification of the purification workflow. By avoiding the generation of stoichiometric phosphine oxide waste, the need for extensive extraction and chromatographic separation is drastically reduced. This reduction in downstream processing translates to lower labor costs, reduced solvent usage, and decreased energy consumption for solvent recovery. Additionally, the high yield and selectivity of the reaction minimize the loss of valuable polyene starting materials, ensuring that the maximum amount of raw material is converted into saleable product. The use of catalytic amounts of Lewis acids, which can often be recovered or neutralized easily, further contributes to the economic efficiency of the process compared to stoichiometric reagent consumption in traditional methods.
- Enhanced Supply Chain Reliability: Supply chain reliability is bolstered by the use of stable intermediate acetals that can be stockpiled without significant degradation. Unlike reactive ylides used in Wittig reactions which must be generated in situ, the acetals and ketene acetals described in this patent have sufficient shelf life to allow for bulk purchasing and inventory management. This stability reduces the pressure on just-in-time manufacturing schedules and provides a buffer against unexpected disruptions in the supply of fresh reagents. Moreover, the reaction conditions are compatible with standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, meaning that production does not require specialized equipment that could become a single point of failure in the manufacturing network.
- Scalability and Environmental Compliance: Scalability is a inherent strength of this methodology due to its reliance on unit operations that are easily transferred from laboratory to industrial scale. The exothermic nature of the Lewis acid catalysis is manageable with standard cooling systems, and the crystallization steps are robust enough to handle large batch sizes without loss of purity. From an environmental compliance standpoint, the process aligns with green chemistry principles by reducing the use of hazardous phosphorus compounds and minimizing solvent waste. The aqueous workups generate waste streams that are easier to treat than those containing organophosphorus residues, facilitating compliance with increasingly strict environmental regulations in major manufacturing hubs. This environmental profile enhances the marketability of the final product to eco-conscious consumers and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this polyene synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this route into their existing production portfolios. The focus is on practical considerations such as isomer control, purification methods, and reagent stability which are critical for successful technology transfer.
Q: How does this process improve upon traditional Wittig reactions for carotenoid synthesis?
A: This method eliminates the need for phosphorus-containing reagents, thereby avoiding the generation of difficult-to-remove phosphine oxide by-products and simplifying the purification process significantly.
Q: What is the stereoselectivity of the double bonds formed in this reaction?
A: The base-induced elimination step predominantly yields the desired all-E isomers, minimizing the formation of Z-isomers and other undesired geometric configurations often seen in acid-catalyzed variants.
Q: Can this process be scaled for industrial production of food colorants?
A: Yes, the process utilizes crystallization steps for purification and avoids chromatographic separation in later stages, making it highly suitable for commercial scale-up of complex carotenoids.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyene Esters Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex patent methodologies into commercial reality for the global fine chemical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this Lewis acid-catalyzed process are fully realized in large-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of polyene ester or carotenoid intermediate meets the exacting standards required by the pharmaceutical and food industries. Our commitment to quality ensures that the high stereoselectivity and purity described in the patent are consistently delivered to our partners, supporting their downstream formulation and product development efforts.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to this phosphorus-free methodology can impact your overall production costs. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your target molecules. Our goal is to provide not just a chemical product, but a strategic partnership that enhances your competitiveness in the market through superior technology and reliable supply.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
