Advanced Industrial Synthesis of Voglibose: Enhancing Purity and Scalability for Global Pharma Supply Chains
Advanced Industrial Synthesis of Voglibose: Enhancing Purity and Scalability for Global Pharma Supply Chains
The pharmaceutical landscape for Type II Diabetes Mellitus treatment continues to evolve, with alpha-glucosidase inhibitors like voglibose playing a pivotal role in managing postprandial hyperglycemia. A recent technological breakthrough, documented in patent CN113214094A, introduces a highly efficient eleven-step synthetic methodology that fundamentally addresses the historical bottlenecks of raw material scarcity and low overall yields. This novel approach leverages glucose monohydrate and sodium acetate as foundational feedstocks, transforming them through a series of meticulously optimized reactions including acetylation, glycosylation, and a critical intramolecular aldol condensation. For R&D directors and supply chain strategists, this patent represents a significant shift towards more sustainable and economically viable manufacturing protocols, offering a robust alternative to legacy processes that often suffer from complex purification requirements and prohibitive input costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of voglibose has been plagued by significant technical and economic hurdles that impede widespread accessibility and cost-effective production. Traditional synthetic routes frequently rely on expensive, hard-to-source chiral starting materials that drive up the initial input costs substantially, creating a barrier for generic manufacturers aiming to compete in price-sensitive markets. Furthermore, existing methodologies often involve harsh reaction conditions that necessitate rigorous environmental controls and specialized equipment, thereby increasing operational expenditures and safety risks. A major pain point in conventional synthesis is the reliance on extensive column chromatography for purification at multiple stages, which not only consumes vast quantities of solvents but also drastically reduces throughput and scalability. These inefficiencies result in low overall yields and a final product with a high market price, limiting patient access and straining procurement budgets for healthcare systems globally.
The Novel Approach
In stark contrast, the methodology outlined in CN113214094A presents a streamlined, cost-optimized pathway that prioritizes atom economy and operational simplicity without compromising on stereochemical integrity. By initiating the synthesis with ubiquitous glucose monohydrate, the process immediately eliminates the dependency on exotic raw materials, establishing a foundation for substantial cost reduction in API manufacturing. The route is characterized by mild reaction conditions, such as the use of a dimethyl sulfoxide-acetic anhydride system for oxidation, which is notably insensitive to air and moisture, thus simplifying reactor requirements. Crucially, the process design enables the purification of key intermediates, such as Compound II, through simple recrystallization from ethanol-cyclohexane rather than chromatography, achieving a remarkable yield of 95.8%. This strategic shift from chromatographic separation to crystallization-based purification significantly enhances the potential for commercial scale-up of complex pharmaceutical intermediates.
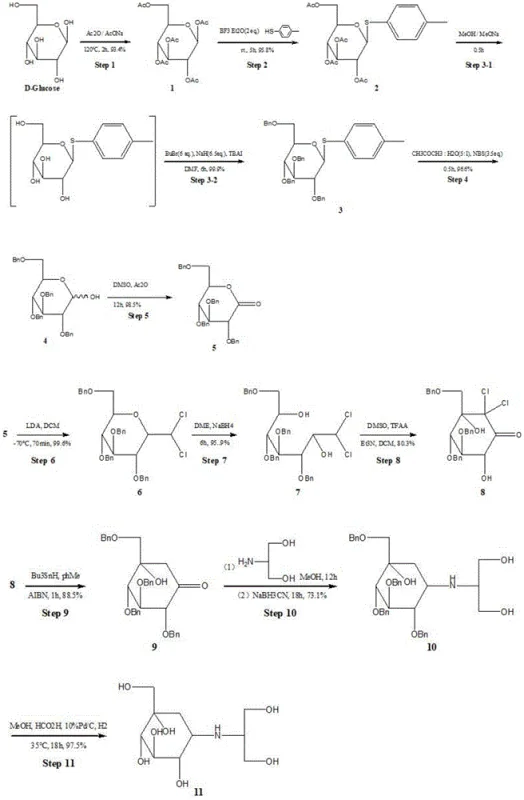
Mechanistic Insights into the Carbocyclic Core Construction
The chemical elegance of this synthesis lies in its strategic manipulation of the carbohydrate scaffold to construct the requisite carbocyclic ring system with high precision. The pathway begins with the peracetylation of glucose, followed by a Lewis acid-catalyzed glycosylation using p-methylthiophenol, which serves as a robust protecting group for the anomeric position while facilitating subsequent stereocontrol. Following the installation of benzyl protecting groups to mask the remaining hydroxyl functionalities, the anomeric thio-group is oxidatively removed to reveal a hemiacetal, which is then oxidized to a gluconolactone. This lactone serves as the electrophilic partner for a subsequent nucleophilic addition using Lithium Diisopropylamide (LDA), setting the stage for the critical ring-closing event. The transformation of the linear chain back into a cyclic structure via intramolecular aldol condensation is the cornerstone of this synthesis, effectively forging the six-membered carbocyclic core that defines the voglibose architecture.
Stereochemical control is rigorously maintained throughout the sequence, particularly during the final functionalization stages. The reductive amination step utilizes sodium cyanoborohydride to introduce the nitrogen-containing side chain with exceptional selectivity, yielding the unique alpha-configuration product essential for biological activity. This step avoids the formation of difficult-to-separate epimeric mixtures, a common issue in amine syntheses, thereby ensuring a high-purity profile for the final active ingredient. Furthermore, the final deprotection step employs a methanol-formic acid solvent system with 10% Pd/C, which offers superior solubility characteristics compared to traditional palladium black catalysts. This modification not only accelerates the hydrogenolysis reaction but also simplifies the post-treatment workup to a mere neutralization and filtration, demonstrating a deep understanding of process chemistry aimed at maximizing efficiency and minimizing waste generation.
How to Synthesize Voglibose Efficiently
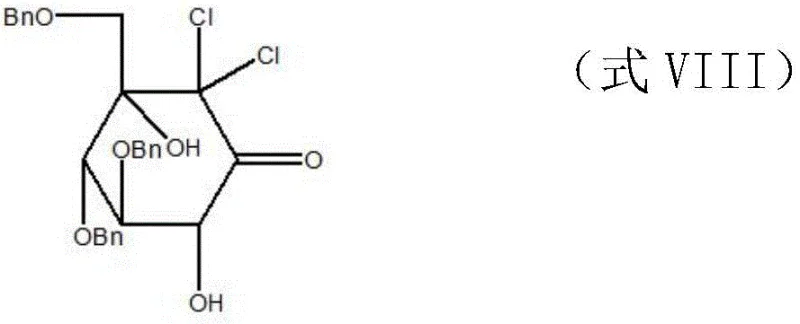
The execution of this eleven-step synthesis requires precise control over reaction parameters, particularly temperature and stoichiometry, to replicate the high yields reported in the patent literature. The process flows logically from simple protection strategies to complex skeletal rearrangements, demanding careful monitoring during the LDA addition and aldol condensation phases to prevent side reactions. Operators must adhere strictly to the specified molar ratios, such as the 1:5 to 1:6 ratio of glucose to acetic anhydride in the initial step, to ensure complete conversion. For a detailed breakdown of the specific operational parameters, reagent quantities, and workup procedures for each of the eleven steps, please refer to the standardized synthesis guide below.
- Acetylation of glucose monohydrate followed by Lewis acid-catalyzed glycosylation with p-methylthiophenol to form the protected glucoside.
- Deprotection and benzylation to secure hydroxyl groups, followed by oxidative desulfurization to expose the anomeric hydroxyl.
- Oxidation to gluconolactone, LDA-mediated addition, ring-opening, and intramolecular aldol condensation to construct the carbocyclic core.
- Radical dechlorination, stereoselective reductive amination with 2-amino-1,3-propanediol, and final catalytic hydrogenolysis to yield voglibose.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the core concerns of procurement managers and supply chain heads regarding cost stability and supply continuity. The substitution of expensive chiral precursors with commodity chemicals like glucose monohydrate creates a buffer against raw material price volatility, ensuring a more predictable cost structure for long-term contracts. Additionally, the elimination of column chromatography in the early stages of the synthesis drastically reduces solvent consumption and waste disposal costs, contributing to a greener manufacturing footprint that aligns with increasingly stringent environmental regulations. The robustness of the reaction conditions, particularly the insensitivity to moisture in the oxidation steps, reduces the risk of batch failures due to environmental factors, thereby enhancing overall supply chain reliability.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive, globally available starting materials and the minimization of unit operations. By replacing complex purification techniques with simple recrystallization and filtration steps, the method significantly lowers the consumption of silica gel and organic solvents, which are major cost drivers in fine chemical production. The high yields observed in the initial five steps, cumulatively reaching over 85%, ensure that material throughput is maximized, reducing the effective cost per kilogram of the intermediate. Furthermore, the use of catalytic amounts of reagents where possible, such as the Lewis acid in step 2, contributes to a leaner bill of materials.
- Enhanced Supply Chain Reliability: The reliance on stable, non-perishable reagents like glucose and sodium acetate mitigates the risk of supply disruptions often associated with specialized fine chemicals. The process tolerance to ambient conditions in several key steps reduces the dependency on highly specialized, climate-controlled infrastructure, allowing for more flexible manufacturing locations. This flexibility is crucial for building a resilient supply network capable of withstanding geopolitical or logistical shocks. The simplified post-treatment protocols also shorten the cycle time per batch, enabling faster turnaround times and improved responsiveness to market demand fluctuations.
- Scalability and Environmental Compliance: Designed with industrial application in mind, this synthesis avoids the use of hazardous reagents that would complicate regulatory approval or waste management. The switch to a methanol-formic acid system for the final hydrogenolysis step exemplifies a commitment to safer chemistry, as it avoids the handling of pyrophoric palladium black. The high atom economy and reduced solvent load make this process inherently more scalable, allowing manufacturers to transition from pilot plant to multi-ton commercial production with minimal process re-engineering. This scalability ensures that the supply of high-purity voglibose can be expanded rapidly to meet growing global demand for anti-diabetic medications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield expectations, purity profiles, and operational feasibility. Understanding these nuances is essential for technical teams evaluating the adoption of this methodology for their own manufacturing pipelines.
Q: What are the primary advantages of this voglibose synthesis route over conventional methods?
A: This route utilizes cheap and readily available glucose monohydrate as the starting material, avoiding expensive chiral pool precursors. It features mild reaction conditions, particularly in the oxidation steps, and simplifies purification by enabling recrystallization instead of column chromatography for key intermediates, significantly lowering production costs.
Q: How does the process ensure high stereoselectivity for the alpha-configuration?
A: The process achieves high stereoselectivity during the reductive amination step (Step 10) using sodium cyanoborohydride. This specific reducing agent favors the formation of the required unique alpha-configuration product with a yield of 73.1%, effectively avoiding complex separation processes typically required for epimeric mixtures.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for industrial scalability. It employs robust reagents like DMSO-Acetic Anhydride for oxidation which are insensitive to air and moisture, and utilizes a methanol-formic acid solvent system in the final step that improves solubility and yield. The total yield for the first five steps reaches 85.1%, demonstrating high efficiency suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Voglibose Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this optimized synthetic route for the global diabetes care market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical efficiencies of this patent are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of voglibose intermediate or API meets the highest international standards. We are committed to leveraging our technical expertise to deliver consistent quality and supply security for our partners.
We invite pharmaceutical companies and generic manufacturers to collaborate with us to optimize their supply chains using this advanced technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out to request specific COA data and route feasibility assessments to determine how this innovative synthesis method can enhance your product portfolio and reduce time-to-market for your anti-diabetic formulations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
