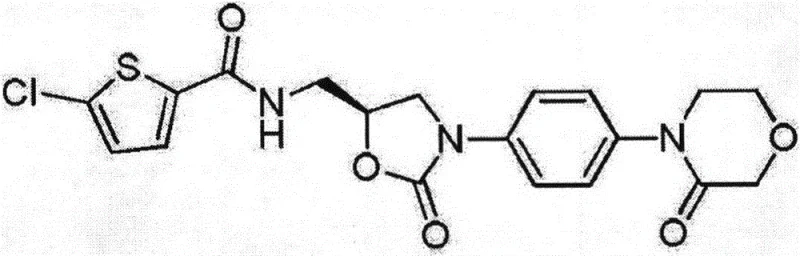
Advanced Manufacturing of Rivaroxaban Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical landscape for anticoagulants continues to evolve, with Rivaroxaban standing as a cornerstone therapy for preventing thrombosis. The efficiency of its supply chain relies heavily on the robustness of its intermediate synthesis. Patent CN110938069A introduces a refined preparation method for a critical Rivaroxaban intermediate, addressing key bottlenecks in yield and purity that have historically plagued large-scale manufacturing. This technical insight report analyzes the patented route, highlighting its potential to serve as a reliable pharmaceutical intermediates supplier solution for global enterprises seeking to optimize their API production lines. The method leverages a specific sequence of condensation and cyclization reactions that not only streamline the workflow but also ensure the structural integrity required for high-purity rivaroxaban. By focusing on solvent systems that are both effective and economically viable, this approach offers a compelling alternative to older, more cumbersome synthetic pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Rivaroxaban and its precursors has been documented in various patents, including US7157456 and CN1900074, yet many of these established routes suffer from significant operational inefficiencies. Conventional methods often rely on harsh reaction conditions or expensive catalysts that complicate the purification process, leading to lower overall yields and higher production costs. The presence of difficult-to-remove impurities in earlier synthetic stages can propagate through the workflow, necessitating extensive and costly downstream processing to meet the stringent purity specifications required for pharmaceutical applications. Furthermore, some traditional routes utilize solvents that are environmentally hazardous or difficult to recover, creating additional burdens for waste management and regulatory compliance. These factors collectively contribute to extended lead times and reduced supply chain flexibility, making it challenging for manufacturers to respond swiftly to market demands. The reliance on multi-step protections and deprotections in older methodologies further exacerbates the complexity, increasing the risk of batch-to-batch variability.
The Novel Approach
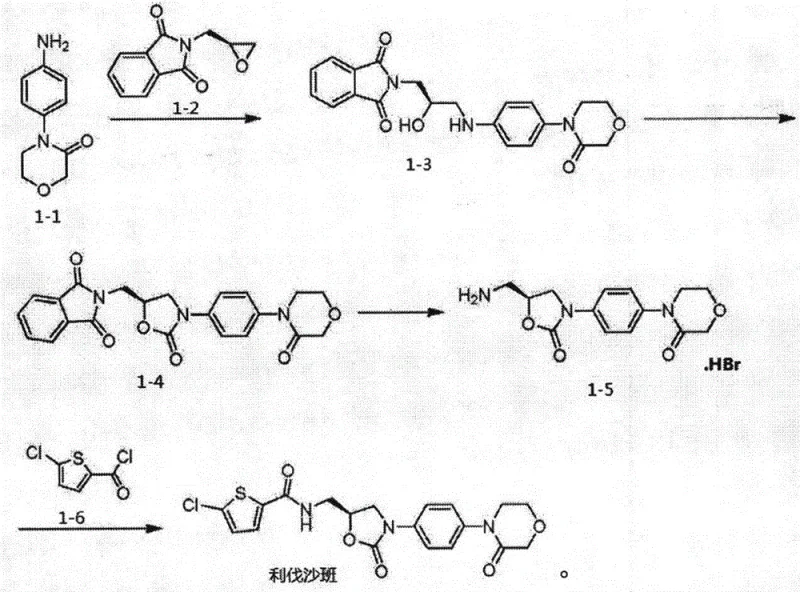
In contrast, the method disclosed in CN110938069A presents a streamlined strategy that significantly simplifies the synthetic pathway while enhancing product quality. This novel approach utilizes a carefully optimized solvent system comprising ethanol and water for the initial condensation step, which not only facilitates the reaction at a moderate temperature of 65°C but also aids in the crystallization of the intermediate. The strategic use of carbonyldiimidazole and 4-dimethylaminopyridine in the subsequent cyclization step ensures high conversion rates without the need for toxic heavy metals. By integrating a specific recrystallization protocol using N,N-dimethylformamide and ethanol-water mixtures, the process effectively removes trace impurities, resulting in a final product with exceptional purity. This methodology directly addresses the need for cost reduction in API manufacturing by minimizing solvent consumption and reducing the number of purification cycles. The robustness of this route makes it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates, offering a stable and reproducible platform for industrial production.
Mechanistic Insights into Ethanol-Water Mediated Condensation and Cyclization
The core of this synthetic innovation lies in the precise control of reaction kinetics during the formation of the oxazolidinone ring system. The initial reaction between compound 1-1 and compound 1-2 in an ethanol-water mixture at 65°C is critical for establishing the correct stereochemistry and preventing side reactions. The specific mass ratio of 1.1:1 between the reactants ensures that the limiting reagent is fully consumed, driving the equilibrium towards the desired intermediate 1-3. The presence of water in the organic solvent system plays a dual role, acting as a co-solvent to dissolve polar intermediates while also facilitating the removal of byproducts through phase separation or crystallization. This careful balance of solvent polarity and temperature is essential for maintaining the integrity of the sensitive functional groups present in the molecule. Subsequent cyclization using carbonyldiimidazole activates the hydroxyl group for intramolecular nucleophilic attack, forming the five-membered ring with high fidelity. The use of 4-dimethylaminopyridine as a nucleophilic catalyst accelerates this acylation process, ensuring that the reaction proceeds to completion within a short timeframe at 60°C.
Impurity control is further enhanced in the later stages through the formation of the hydrobromide salt of intermediate 1-5. The conversion of the free amine to its salt form not only stabilizes the molecule against oxidation but also allows for efficient purification via filtration. The final coupling reaction with compound 1-6 is conducted under mild basic conditions using triethylamine, which prevents the racemization of the chiral center. The dual recrystallization strategy employed in the final step is particularly effective; the first crystallization from DMF and ethanol-water removes bulk impurities, while the second pulping in ethanol-water refines the crystal lattice to achieve the target 99.9% purity. This multi-layered approach to purification ensures that the impurity profile remains well within the limits required for regulatory submission. For R&D directors, understanding these mechanistic nuances is vital for troubleshooting potential scale-up issues and ensuring consistent quality across large batches.
How to Synthesize Rivaroxaban Intermediate Efficiently
The implementation of this synthesis route requires strict adherence to the specified reaction parameters to maximize yield and purity. The process begins with the condensation of the aniline derivative and the epoxide precursor, followed by a cyclization step that constructs the core heterocyclic scaffold. Subsequent ring opening and salt formation prepare the molecule for the final amidation coupling. Each step is designed to be operationally simple, relying on standard unit operations such as heating, filtration, and crystallization that are easily transferable to pilot and production scales. The detailed standardized synthesis steps see the guide below for specific stoichiometric ratios and temperature profiles.
- Condense compound 1-1 with 1-2 in ethanol and water at 65°C to form intermediate 1-3.
- React intermediate 1-3 with carbonyldiimidazole and DMAP in THF at 60°C to yield compound 1-4.
- Treat compound 1-4 with ethanolamine followed by hydrobromic acid to generate hydrobromide salt 1-5.
- Couple salt 1-5 with compound 1-6 using triethylamine, followed by dual recrystallization to obtain Rivaroxaban.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this patented method offers substantial benefits by leveraging widely available raw materials and solvents. The reliance on ethanol, water, and ethyl acetate eliminates the need for specialized or controlled substances that often face supply constraints or regulatory hurdles. This accessibility translates directly into enhanced supply chain reliability, as manufacturers are less vulnerable to disruptions in the availability of exotic reagents. The simplified workflow reduces the overall processing time, allowing for faster turnover of batches and more responsive inventory management. For supply chain heads, the predictability of this process means that production schedules can be optimized with greater confidence, reducing the need for excessive safety stock. The elimination of complex transition metal catalysts also removes the requirement for expensive metal scavenging steps, further streamlining the production timeline.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven by the high efficiency of the reaction steps and the minimization of waste. By achieving high conversion rates in the initial condensation and cyclization steps, the process reduces the amount of unreacted starting material that needs to be recovered or disposed of. The use of cost-effective solvents like ethanol and water significantly lowers the raw material expenditure compared to routes relying on chlorinated or aromatic solvents. Furthermore, the high purity of the intermediates reduces the burden on downstream purification, saving both time and resources. The avoidance of expensive noble metal catalysts removes a significant cost center from the bill of materials. These factors combine to deliver significant cost savings without compromising on the quality of the final API.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures a consistent supply of high-quality intermediates, which is critical for maintaining continuous API production. The use of stable intermediates, such as the hydrobromide salt, allows for safer storage and transportation, reducing the risk of degradation during logistics. The scalability of the process means that suppliers can easily ramp up production volumes to meet surges in demand without requiring major capital investment in new equipment. This flexibility is essential for mitigating risks associated with market volatility. By establishing a reliable pharmaceutical intermediates supplier relationship based on this technology, companies can secure a stable source of critical materials for their anticoagulant portfolios.
- Scalability and Environmental Compliance: The process is inherently designed for industrial production, with reaction conditions that are easily managed in large-scale reactors. The moderate temperatures and atmospheric pressure operations reduce energy consumption and safety risks associated with high-pressure or cryogenic processes. The solvent system is amenable to recovery and recycling, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing site. The high purity of the final product minimizes the generation of hazardous waste streams associated with extensive purification. This alignment with environmental compliance standards facilitates smoother regulatory approvals and enhances the sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the patent data to provide accurate guidance for stakeholders evaluating this technology. Understanding these details is crucial for making informed decisions about process adoption and supplier selection.
Q: What is the purity profile of the Rivaroxaban intermediate produced via this method?
A: The patented process demonstrates exceptional purity control, with intermediate 1-3 achieving 98.7% HPLC purity and the final Rivaroxaban product reaching 99.9% purity after recrystallization, significantly minimizing impurity risks for downstream API manufacturing.
Q: How does this synthesis route improve supply chain stability compared to conventional methods?
A: By utilizing commercially available solvents like ethanol and ethyl acetate and avoiding complex transition metal catalysts, this method reduces dependency on specialized reagents, thereby enhancing supply chain reliability and reducing lead time for high-purity pharmaceutical intermediates.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent explicitly states the method features simple operation and high yield, making it highly suitable for industrial production and commercial scale-up of complex pharmaceutical intermediates without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rivaroxaban Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing the technical expertise to translate complex patent methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Rivaroxaban intermediate meets the highest global standards. Our commitment to quality and consistency makes us a trusted partner for pharmaceutical companies seeking to secure their supply chains against market fluctuations.
We invite you to collaborate with us to optimize your production costs and enhance your supply chain resilience. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments for your upcoming projects. By leveraging our manufacturing capabilities, you can ensure a steady supply of high-purity materials for your critical drug formulations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
