Advanced Phosphino-Oxygen Modified Bipyridyl-Phenazinyl Red TADF Materials for High-Efficiency OLED Manufacturing
The landscape of organic light-emitting diode (OLED) technology is undergoing a transformative shift with the advent of advanced Thermally Activated Delayed Fluorescence (TADF) materials, as detailed in the groundbreaking patent CN110283209B. This intellectual property introduces a novel class of phosphino-oxygen group modified bipyridyl-phenazinyl materials specifically engineered for red and near-infrared emission applications. Traditional fluorescent materials have long been constrained by a theoretical internal quantum efficiency (IQE) limit of 25%, while phosphorescent alternatives, though efficient, rely on scarce and costly heavy metals. The innovation described in this patent addresses these critical bottlenecks by utilizing a unique donor-acceptor molecular architecture that facilitates reverse intersystem crossing (RISC), theoretically enabling 100% IQE without the burden of precious metal catalysts. 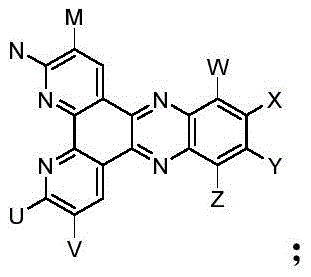
Furthermore, the specific incorporation of the phosphino-oxygen moiety serves a dual purpose: it acts as a secondary electron acceptor to fine-tune photoelectric properties and provides substantial steric bulk to prevent molecular aggregation. This structural ingenuity directly tackles the pervasive issue of concentration quenching often observed in red-emitting TADF systems, where high polarity leads to rapid efficiency attenuation. For R&D directors and procurement specialists in the display industry, this represents a pivotal opportunity to access high-performance emissive layers that promise both superior device longevity and a more sustainable, cost-effective supply chain compared to legacy phosphorescent technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional red and near-infrared OLED emitters have historically struggled with a trade-off between efficiency and stability, primarily due to the inherent physical limitations of first-generation fluorescent materials and the economic constraints of second-generation phosphorescent systems. Fluorescent emitters are fundamentally restricted by spin statistics, capable of harvesting only singlet excitons while wasting 75% of triplet excitons as heat, which severely limits the overall power efficiency of the display panel. On the other hand, while phosphorescent materials utilizing iridium or platinum complexes can harvest both singlet and triplet excitons, they introduce significant supply chain vulnerabilities due to the geopolitical scarcity and price volatility of these heavy metals. Moreover, the high molecular polarity often associated with efficient red emitters tends to induce strong intermolecular interactions in the solid state, leading to severe concentration quenching effects that degrade device performance over time and necessitate complex doping strategies to mitigate.
The Novel Approach
The novel approach delineated in patent CN110283209B circumvents these historical challenges through a sophisticated molecular design that integrates a bipyridyl-phenazine acceptor core with triphenylamine donors and, crucially, diphenyl phosphine-oxygen modifiers. This architecture creates a small energy gap between the lowest singlet and triplet excited states (ΔEST < 0.5 eV), facilitating efficient thermal up-conversion of triplet excitons to singlet states via reverse intersystem crossing. The strategic placement of the phosphine-oxygen group not only enhances electron injection and transport capabilities due to its strong polarization effect but also introduces significant steric hindrance. This steric bulk physically separates the luminous cores within the thin film, effectively suppressing non-radiative decay pathways caused by molecular stacking and ensuring that the high theoretical efficiency of the TADF mechanism is maintained even at practical operating concentrations.
Mechanistic Insights into Phosphine-Oxide Mediated Steric Protection
The mechanistic superiority of this material class lies in the synergistic interaction between the electron-deficient bipyridyl-phenazine core and the electron-rich triphenylamine units, which establishes a robust intramolecular charge transfer (ICT) state essential for TADF activity. Upon electrical excitation, the formation of excitons occurs in the standard 1:3 singlet-to-triplet ratio; however, the minimized ΔEST allows the triplet population to thermally activate back to the singlet manifold, from which radiative decay occurs, effectively recycling the otherwise lost triplet energy. The introduction of the phosphine-oxygen (P=O) group further modulates the electronic landscape by acting as an auxiliary electron-withdrawing group, which stabilizes the lowest unoccupied molecular orbital (LUMO) and improves the balance of charge carrier injection. This electronic tuning is critical for reducing the driving voltage of the OLED device, thereby lowering power consumption and heat generation during operation.
From an impurity control and purity perspective, the synthetic route leverages well-defined palladium-catalyzed coupling reactions that offer high selectivity, minimizing the formation of regio-isomers that could act as quenching sites. The steric protection afforded by the bulky diphenylphosphine oxide groups also plays a vital role in the solid-state morphology of the emitted layer. By preventing close π-π stacking interactions between adjacent molecules, the material maintains its amorphous stability and prevents crystallization during device operation, which is a common failure mode in organic thin films. This morphological stability ensures consistent color purity and brightness over the operational lifetime of the display, addressing a key reliability metric for commercial adoption in high-end consumer electronics and lighting applications.
How to Synthesize Phosphino-Oxygen Modified Bipyridyl-Phenazinyl Efficiently
The synthesis of these high-performance TADF materials follows a streamlined two-step protocol that is highly amenable to scale-up, beginning with the condensation of a dibromo-phenanthroline-dione precursor with a functionalized diamine. This initial step constructs the rigid bipyridyl-phenazine core, establishing the fundamental acceptor unit of the molecule under relatively mild thermal conditions in an ethanol solvent system. The subsequent step involves a palladium-catalyzed cross-coupling reaction with diphenylphosphine, followed by an in-situ oxidation to generate the stable phosphine-oxide functionality. This sequence avoids the use of unstable phosphine intermediates and ensures the final product possesses the necessary thermal and chemical robustness for vacuum deposition processes. 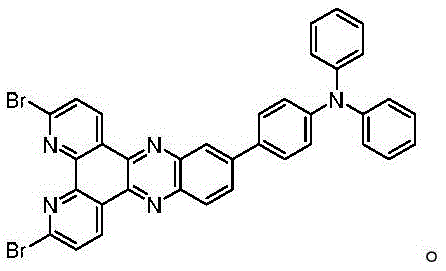
- Condense 2,9-dibromo-phenanthroline-5,6-dione with a specific diamine precursor in ethanol at 80°C for 48 hours to form the brominated intermediate.
- Perform a palladium-catalyzed coupling reaction between the intermediate and diphenylphosphine in DMF at 150°C for 48 hours.
- Oxidize the resulting phosphine species using hydrogen peroxide, followed by extraction and column chromatography purification to isolate the final phosphino-oxygen modified material.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this phosphino-oxygen modified TADF technology offers profound strategic advantages centered around cost rationalization and supply security. The most immediate impact is the elimination of expensive heavy metal precursors such as iridium or platinum, which are subject to extreme market volatility and concentrated geographic supply risks. By shifting to a purely organic synthesis pathway, manufacturers can decouple their production costs from the fluctuations of the precious metals market, leading to a more predictable and stable cost of goods sold (COGS). Furthermore, the synthetic route utilizes commodity chemicals and standard catalytic systems, which simplifies the sourcing strategy and reduces the dependency on specialized, single-source suppliers often associated with complex organometallic reagents.
- Cost Reduction in Manufacturing: The absence of heavy metals not only lowers raw material expenses but also drastically simplifies the downstream purification and waste management processes. Traditional phosphorescent manufacturing requires rigorous and costly steps to remove trace metal residues to meet environmental and performance standards, whereas this organic TADF route eliminates the need for expensive metal scavenging resins and specialized hazardous waste disposal protocols. Additionally, the high thermal stability of the final compounds, evidenced by decomposition temperatures exceeding 370°C, reduces material loss during the high-vacuum thermal evaporation process, improving the overall material utilization rate and yield in the fabrication line.
- Enhanced Supply Chain Reliability: The synthetic precursors, such as dibromo-phenanthroline-dione and various triphenylamine derivatives, are based on widely available petrochemical feedstocks, ensuring a robust and diversified supply base. This contrasts sharply with the supply chain for iridium-based emitters, which is often constrained by mining output and refining capacity. The ability to source starting materials from multiple global vendors mitigates the risk of production stoppages due to supplier shortages, providing procurement teams with greater leverage in negotiations and ensuring continuous availability of critical emissive materials for mass production schedules.
- Scalability and Environmental Compliance: The two-step synthesis described in the patent is inherently scalable, utilizing standard reaction vessels and workup procedures that translate easily from laboratory to pilot and commercial scales. The reaction conditions, while requiring elevated temperatures, do not demand extreme pressures or cryogenic environments, reducing the capital expenditure required for specialized reactor infrastructure. From an environmental compliance standpoint, the organic nature of the waste stream simplifies treatment compared to heavy metal-laden effluents, aligning with increasingly stringent global regulations on hazardous substances in electronics manufacturing and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of phosphino-oxygen modified TADF materials in OLED production lines. These insights are derived directly from the experimental data and performance metrics disclosed in the patent documentation, providing a factual basis for evaluating the technology's readiness for integration. Understanding these specifics is crucial for technical teams assessing compatibility with existing device architectures and for business leaders calculating the total cost of ownership.
Q: What is the primary advantage of the phosphino-oxygen group modification in this TADF material?
A: The introduction of the diphenyl phosphine-oxygen group provides significant steric hindrance, which effectively inhibits intermolecular interactions and reduces concentration quenching, thereby enhancing the efficiency stability of the electroluminescent device.
Q: How does this material compare to traditional phosphorescent OLED emitters regarding cost?
A: Unlike traditional phosphorescent materials that rely on expensive heavy metal dopants like iridium or platinum, this TADF material utilizes purely organic components, significantly reducing raw material costs and eliminating the need for complex heavy metal removal processes.
Q: What is the thermal stability profile of the synthesized compounds?
A: Thermogravimetric analysis indicates high thermal stability, with decomposition temperatures ranging from approximately 370°C to over 500°C depending on the specific derivative, ensuring robustness during the vacuum evaporation processes used in OLED fabrication.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphino-Oxygen Modified TADF Material Supplier
As the demand for high-efficiency, heavy-metal-free display materials accelerates, NINGBO INNO PHARMCHEM stands ready to support your transition to next-generation TADF technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the complex multi-step synthesis of these bipyridyl-phenazinyl derivatives can be executed with the highest standards of quality and consistency. We understand that the purity specifications for OLED emitters are exceptionally stringent, and our rigorous QC labs are equipped to verify trace impurity profiles and ensure batch-to-batch reproducibility that meets the exacting demands of panel manufacturers.
We invite you to engage with our technical procurement team to discuss how this innovative material class can optimize your product roadmap. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching from phosphorescent to TADF emitters in your specific application. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, securing a competitive advantage in the rapidly evolving landscape of organic optoelectronics.
