Scaling High-Purity Rupestonic Acid Glycolipid Derivatives for Next-Generation Antiviral Therapeutics
Scaling High-Purity Rupestonic Acid Glycolipid Derivatives for Next-Generation Antiviral Therapeutics
The pharmaceutical landscape is constantly evolving towards more potent and selective antiviral agents, and patent CN101857613A represents a significant stride in this direction by detailing the synthesis of novel rupestonic acid glycolipid derivatives. This intellectual property outlines a robust methodology for modifying rupestonic acid, a bioactive sesquiterpenoid isolated from Artemisia rupestris, into a series of sugar ester analogs designed to enhance therapeutic efficacy against influenza viruses. For R&D directors and procurement specialists alike, this technology offers a compelling value proposition: it transforms a naturally occurring scaffold into a library of high-value pharmaceutical intermediates through a chemically elegant and operationally simple phase transfer catalysis (PTC) process. The strategic incorporation of fully acetylated glycosyl segments not only modifies the physicochemical properties of the parent molecule but also unlocks new biological activities, specifically targeting Influenza A (H3N2, H1N1) and Influenza B strains.
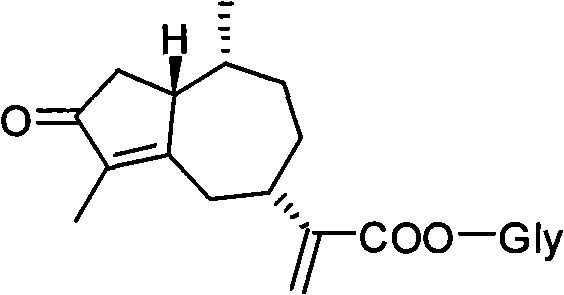
From a supply chain perspective, the ability to synthesize these derivatives reliably positions manufacturers as a reliable pharmaceutical intermediate supplier capable of meeting the rigorous demands of modern drug discovery. The patent emphasizes that the method employs mild reaction conditions and straightforward experimental steps, which are critical factors for reducing lead time for high-purity pharmaceutical intermediates in a commercial setting. By leveraging this technology, companies can bypass the limitations of direct extraction from plant sources, which often suffer from seasonal variability and low yields, and instead opt for a controlled semi-synthetic route that ensures consistent quality and supply continuity for downstream API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, obtaining bioactive terpenoid derivatives from natural sources like Artemisia rupestris has been fraught with challenges that hinder large-scale commercialization. Conventional isolation techniques often require extensive solvent extraction and repeated chromatographic separations to obtain the parent rupestonic acid in sufficient purity, a process that is both time-consuming and resource-intensive. Furthermore, direct modification of the parent compound using classical esterification methods frequently necessitates harsh conditions, such as strong acidic catalysts or elevated temperatures, which can lead to the degradation of the sensitive sesquiterpene skeleton or the sugar moieties. These aggressive conditions often result in complex impurity profiles, including dehydration products or rearranged isomers, making the subsequent purification steps difficult and costly. For procurement managers, these inefficiencies translate into higher costs and unpredictable timelines, creating bottlenecks in the development of antiviral candidates.
The Novel Approach
In stark contrast, the methodology described in CN101857613A introduces a refined synthetic strategy that overcomes these historical hurdles through the application of phase transfer catalysis. By utilizing tetraethylbenzyl ammonium chloride (TEBAc) as a catalyst in a biphasic system of 1,2-dichloroethane and aqueous sodium hydroxide, the reaction proceeds efficiently at moderate temperatures ranging from 50°C to 60°C. This approach facilitates the nucleophilic attack of the rupestonic acid carboxylate anion on the fully acetylated bromo-sugar electrophile without compromising the structural integrity of either component. The result is a cleaner reaction profile with higher selectivity for the desired ester linkage. As illustrated in the reaction scheme below, this method allows for the modular introduction of various sugar units, enabling the rapid generation of diverse analogs for structure-activity relationship (SAR) studies.
This novel approach significantly streamlines the workflow for cost reduction in pharmaceutical intermediate manufacturing by eliminating the need for expensive coupling reagents or extreme reaction parameters. The simplicity of the workup procedure, which involves basic liquid-liquid extraction followed by standard column chromatography, ensures that the process is readily adaptable for commercial scale-up of complex pharmaceutical intermediates. Moreover, the formation of a specific solvent-derived byproduct (Compound 3), which interestingly retains antiviral activity, suggests that the process is robust enough to tolerate minor variations while still yielding biologically relevant material, thereby reducing the risk of batch failure in a production environment.
Mechanistic Insights into Phase Transfer Catalyzed Esterification
The core of this synthetic innovation lies in the mechanistic efficiency of the phase transfer catalyst, TEBAc, which acts as a molecular shuttle to transport the rupestonic acid anion from the aqueous phase into the organic phase where the bromo-sugar resides. In the absence of the catalyst, the electrostatic repulsion and solubility differences between the ionic carboxylate salt and the lipophilic sugar halide would severely limit the reaction rate. TEBAc forms a lipophilic ion pair with the carboxylate anion, solubilizing it in the 1,2-dichloroethane layer and dramatically increasing its local concentration near the electrophilic sugar substrate. This intimate contact promotes an SN2-type substitution mechanism where the carboxylate displaces the bromide leaving group, forming the ester bond with inversion of configuration at the anomeric center of the sugar, although the specific stereochemical outcome depends on the neighboring group participation of the acetyl groups.
Regarding impurity control, the mechanism also explains the formation of the observed byproducts and how they can be managed. The reaction conditions are tuned to favor the intermolecular esterification over potential intramolecular side reactions or hydrolysis of the acetyl protecting groups. However, the patent notes the formation of Compound 3, a product resulting from the reaction with the solvent 1,2-dichloroethane, likely via an elimination-addition sequence or direct alkylation under the basic conditions. Understanding this mechanistic pathway is crucial for R&D teams aiming to optimize the process, as it highlights the importance of controlling the stoichiometry of the base and the reaction time to minimize solvent incorporation. The ability to separate these components via gradient elution (petroleum ether:ethyl acetate) demonstrates that despite the formation of byproducts, the polarity differences are sufficient for effective purification, ensuring the delivery of high-purity OLED material or pharmaceutical grade intermediates depending on the final application.
How to Synthesize Rupestonic Acid Glycolipid Derivatives Efficiently
The synthesis of these valuable antiviral intermediates follows a logical three-step sequence that balances reaction efficiency with ease of purification, making it an ideal candidate for technology transfer. The process begins with the activation of rupestonic acid in a biphasic mixture, followed by the controlled addition of the glycosyl donor, and concludes with a straightforward isolation protocol. This standardized approach minimizes operator error and ensures reproducibility across different batches, which is essential for maintaining regulatory compliance in GMP environments. The detailed standardized synthesis steps see the guide below for specific operational parameters.
- Dissolve rupestonic acid in 1,2-dichloroethane and add 5% sodium hydroxide solution along with the phase transfer catalyst TEBAc under stirring at room temperature.
- Introduce the fully acetylated bromo-sugar dissolved in 1,2-dichloroethane to the mixture and stir at 50-60°C until TLC indicates reaction completion.
- Separate the organic phase, wash with sodium hydroxide and water, dry over anhydrous Na2SO4, and purify the crude syrup via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this PTC-based synthesis route offers tangible strategic advantages that go beyond mere chemical novelty. The primary benefit lies in the substantial cost savings achieved through the simplification of the reaction infrastructure; the use of common solvents like 1,2-dichloroethane and inexpensive inorganic bases like sodium hydroxide eliminates the dependency on specialized, high-cost reagents often required for peptide or glycosidic couplings. Furthermore, the mild thermal requirements (50-60°C) reduce energy consumption and lower the safety risks associated with high-pressure or high-temperature reactors, directly contributing to cost reduction in API manufacturing. The robustness of the phase transfer system also means that the process is less sensitive to minor fluctuations in raw material quality, enhancing overall supply chain reliability.
- Cost Reduction in Manufacturing: The elimination of expensive coupling agents and the use of catalytic amounts of TEBAc significantly lower the raw material cost per kilogram of the final product. Additionally, the simplified workup procedure reduces solvent usage and waste disposal costs, as the aqueous washes effectively remove inorganic salts without requiring complex quenching steps. This lean manufacturing approach ensures that the production of these intermediates remains economically viable even at smaller scales, allowing for flexible batch sizing based on market demand.
- Enhanced Supply Chain Reliability: The starting materials, including rupestonic acid and various peracetylated bromo-sugars, are either isolable from abundant natural sources or commercially available from established chemical suppliers. This diversity in sourcing options mitigates the risk of single-supplier dependency, ensuring reducing lead time for high-purity pharmaceutical intermediates. The stability of the intermediates and the final products under standard storage conditions further simplifies logistics, allowing for the maintenance of strategic stockpiles without significant degradation concerns.
- Scalability and Environmental Compliance: The process is inherently scalable, as phase transfer catalysis is a well-established technology in the fine chemical industry with known parameters for scale-up from grams to tons. The use of 1,2-dichloroethane, while requiring proper handling, allows for efficient recycling and recovery systems, aligning with modern environmental compliance standards. The absence of heavy metal catalysts removes the need for stringent metal scavenging steps, simplifying the purification train and reducing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of rupestonic acid derivatives, based on the specific data and embodiments provided in the patent literature. These insights are designed to clarify the feasibility of implementing this technology in a commercial setting and to highlight the specific advantages of the described synthetic route over alternative methods. Understanding these details is crucial for stakeholders evaluating the potential integration of these intermediates into their existing pipelines.
Q: What is the primary advantage of using Phase Transfer Catalysis in this synthesis?
A: The use of tetraethylbenzyl ammonium chloride (TEBAc) allows the reaction to proceed under mild conditions (50-60°C) in a biphasic system, significantly simplifying the operational complexity compared to traditional high-temperature esterification methods.
Q: Which specific derivatives showed the highest antiviral activity in the patent data?
A: Experimental results indicated that compounds 2d (xylosyl derivative) and 2g (lactose derivative) exhibited superior inhibitory effects against Influenza A (H3N2) virus, while the solvent-derived byproduct 3 showed broad-spectrum activity against both Type A and Type B influenza viruses.
Q: How does this method address impurity control during scale-up?
A: The process utilizes standard liquid-liquid extraction and gradient elution column chromatography (petroleum ether:ethyl acetate), which are highly scalable techniques that effectively separate the target esters from unreacted starting materials and side products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rupestonic Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in CN101857613A and are fully equipped to bring these advanced antiviral intermediates from the laboratory bench to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We operate state-of-the-art rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of rupestonic acid glycolipid derivative meets the exacting standards required for preclinical and clinical development. Our commitment to quality assurance means that we can navigate the complexities of PTC chemistry to deliver material that is free from critical impurities.
We invite you to collaborate with us to explore the full commercial potential of this technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can accelerate your drug development programs while optimizing your overall budget. Let us be your partner in turning this promising scientific innovation into a market-leading therapeutic reality.
