Scaling High-Purity Iloperidone Production with Advanced Aqueous Emulsion Catalysis
Scaling High-Purity Iloperidone Production with Advanced Aqueous Emulsion Catalysis
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more sustainable manufacturing processes, particularly for complex antipsychotic agents. Patent CN102516236A introduces a groundbreaking preparation method for Iloperidone, a potent atypical antipsychotic drug, which fundamentally alters the synthetic landscape by replacing hazardous organic solvents with an advanced aqueous surfactant system. This innovation addresses critical pain points in the production of high-purity pharmaceutical intermediates, offering a route that is not only environmentally benign but also economically superior due to simplified downstream processing. By leveraging the unique properties of non-ionic surfactants to create nano-emulsions, this technology ensures exceptional mass transfer efficiency, resulting in crude product purities exceeding 98.8% and overall yields surpassing 96%. For R&D directors and supply chain managers seeking a reliable API intermediate supplier, this methodology represents a significant leap forward in process robustness and scalability.
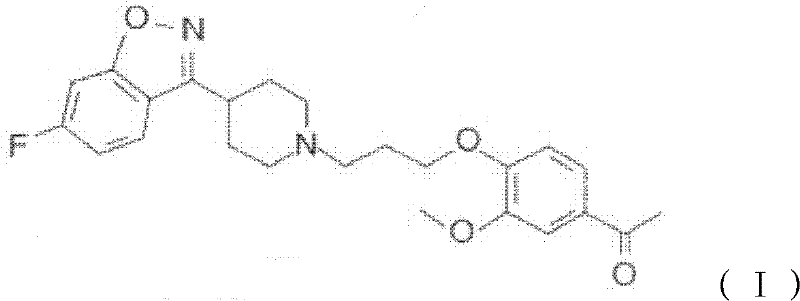
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Iloperidone has been plagued by significant safety and efficiency challenges associated with solvent selection and reaction heterogeneity. The original process described in U.S. Pat 5776963 relied heavily on N,N-dimethylformamide (DMF) as the reaction medium, necessitating high temperatures of 90°C and extended reaction times of up to 16 hours. This approach suffered from a dismal crude yield of merely 58%, largely due to the formation of stubborn by-products and the difficulty in isolating the product from the high-boiling, toxic solvent. Furthermore, DMF poses severe occupational health risks and generates hazardous waste streams that require costly and complex treatment protocols. Even subsequent improvements, such as those found in Chinese Patent CN101768154 which attempted to switch to water, failed to fully resolve the engineering bottlenecks; these water-based systems often resulted in severe agglomeration and caking of the product, leading to poor heat transfer, entrapped impurities, and difficult filtration operations that hampered commercial scale-up.
The Novel Approach
In stark contrast to these legacy methods, the novel process detailed in CN102516236A utilizes a sophisticated "oil-in-water" emulsion system driven by non-ionic surfactants like Tween 80. This approach effectively solubilizes the hydrophobic organic reactants within microscopic micelles dispersed in an aqueous continuous phase, dramatically increasing the effective contact surface area between the reacting species. As illustrated in the reaction scheme below, the coupling of the piperidine benzisoxazole fragment with the chloropropoxy phenyl ketone proceeds with remarkable homogeneity and efficiency. This physical state prevents the notorious caking and agglomeration observed in previous water-based attempts, ensuring that the crystalline product forms as fine, free-flowing particles that are easily filtered and washed. The result is a streamlined operation that reduces reaction times to just 3-5 hours while boosting crude yields to over 96%, fundamentally transforming the economic and operational viability of Iloperidone manufacturing.
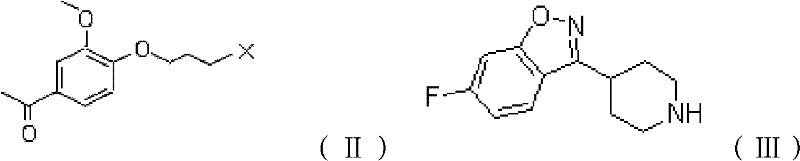
Mechanistic Insights into Surfactant-Mediated Nucleophilic Substitution
The core of this technological breakthrough lies in the mechanistic role of the non-ionic surfactant and the potassium iodide catalyst within the aqueous medium. When reagents such as 6-fluoro-3-(4-piperidyl)-1,2-benzoisoxazole hydrochloride and 1-[4-(3-chloropropoxy)-3-methoxyphenyl] ethanone are introduced into the Tween 80 solution, they are encapsulated within the hydrophobic cores of the surfactant micelles. This nano-confinement effect creates a localized environment with a high concentration of reactants, effectively mimicking the conditions of an organic solvent while maintaining the safety profile of water. The addition of potassium iodide serves as a potent nucleophilic catalyst, facilitating the SN2 displacement of the chloride atom on the propoxy chain by the piperidine nitrogen. This catalytic cycle lowers the activation energy barrier, allowing the reaction to proceed rapidly at moderate temperatures of 70-85°C, thereby minimizing thermal degradation and the formation of colored impurities that typically necessitate activated carbon treatment.
Furthermore, the control of crystal growth dynamics in this emulsion system is critical for achieving the reported high purity specifications. In conventional heterogeneous reactions, rapid precipitation often leads to the occlusion of unreacted starting materials and solvent molecules within the crystal lattice, creating persistent impurities that are difficult to remove. However, the controlled supersaturation achieved in the surfactant-mediated system promotes the formation of uniform, well-defined crystals with minimal defect sites. This structural integrity ensures that impurities remain in the mother liquor rather than being trapped within the solid product. Consequently, the crude Iloperidone obtained directly from filtration exhibits a purity of greater than 98.8% by HPLC normalization, a level of quality that traditionally would require multiple recrystallization steps. This inherent purity simplifies the refining process to a single methanol recrystallization, yielding a final product with purity exceeding 99.6% and single maximum impurity levels below 0.1%.
How to Synthesize Iloperidone Efficiently
The implementation of this synthesis route requires precise control over stoichiometry and temperature to maximize the benefits of the emulsion catalysis. The process begins with the preparation of the aqueous surfactant solution, followed by the sequential addition of the base, catalyst, and organic substrates. Maintaining the reaction temperature within the optimal window of 70-85°C is crucial; temperatures that are too low result in incomplete conversion, while excessive heat may degrade the surfactant or promote side reactions. Upon completion, the product precipitates naturally upon cooling, allowing for straightforward isolation via suction filtration. The following guide outlines the standardized operational parameters derived from the patent embodiments to ensure reproducible high-yield production suitable for industrial application.
- Prepare the reaction medium by dissolving a non-ionic surfactant, such as Tween 80, in water to form an emulsion base.
- Combine 6-fluoro-3-(4-piperidyl)-1,2-benzoisoxazole hydrochloride and 1-[4-(3-chloropropoxy)-3-methoxyphenyl] ethanone with an inorganic base like potassium carbonate and a potassium iodide catalyst.
- Heat the mixture to 70-85°C for 3-5 hours, then cool, filter the precipitate, and recrystallize from methanol to achieve >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this aqueous surfactant-based methodology offers profound strategic advantages beyond mere technical performance. The elimination of DMF and toluene from the process workflow removes the burden of handling, storing, and disposing of Class 2 and Class 3 solvents, which are subject to increasingly stringent environmental regulations and volatile pricing markets. By substituting these hazardous chemicals with water and commodity-grade surfactants, manufacturers can significantly reduce their raw material costs and mitigate the risk of supply disruptions associated with specialty solvent availability. Moreover, the simplified workup procedure, which avoids the use of activated carbon and complex extraction sequences, translates directly into reduced labor hours and lower utility consumption, driving down the overall cost of goods sold (COGS) for this high-value antipsychotic intermediate.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic simplification of the downstream processing train. Traditional methods often require multiple solvent exchanges, extensive washing cycles, and the use of expensive adsorbents like activated carbon to meet color and purity specifications. In this novel route, the high selectivity of the surfactant-mediated reaction minimizes by-product formation, effectively eliminating the need for carbon decolorization. Additionally, the use of water as the primary reaction medium removes the capital and operational expenditures associated with solvent recovery distillation columns and explosion-proof infrastructure, leading to substantial long-term savings in both CAPEX and OPEX for production facilities.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the reliance on widely available, non-hazardous reagents enhances the resilience of the supply chain. Potassium carbonate, potassium iodide, and Tween series surfactants are bulk commodities with stable global supply networks, unlike specialized organic solvents which can face logistical bottlenecks. The robustness of the reaction against minor variations in mixing or temperature further ensures consistent batch-to-batch quality, reducing the rate of rejected batches and ensuring a steady flow of material to downstream formulation teams. This reliability is critical for maintaining uninterrupted production schedules for finished dosage forms in the competitive CNS therapeutic market.
- Scalability and Environmental Compliance: The physical characteristics of the reaction mixture, specifically the lack of agglomeration and the formation of free-flowing crystals, make this process exceptionally scalable from pilot plant to multi-ton commercial production. The absence of viscous sludge or hard cakes ensures that standard filtration and drying equipment can be utilized without modification, facilitating rapid technology transfer. Environmentally, the process aligns perfectly with Green Chemistry principles by generating an aqueous waste stream that is far easier to treat than DMF-laden effluent. This compliance reduces the environmental tax burden and simplifies the permitting process for new manufacturing lines, positioning the producer as a leader in sustainable pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous emulsion technology for Iloperidone synthesis. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on how this method outperforms legacy techniques in terms of yield, purity, and operational safety. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this greener synthetic route for their own supply chains.
Q: What are the primary advantages of using a non-ionic surfactant in Iloperidone synthesis?
A: The use of non-ionic surfactants like Tween 80 creates a stable oil-in-water nano-emulsion. This significantly increases the interfacial surface area between hydrophobic reactants, leading to faster reaction kinetics, higher yields (>96%), and preventing the agglomeration issues seen in previous water-based methods.
Q: How does this method compare to traditional DMF-based processes in terms of purity?
A: Traditional DMF processes often yield crude products around 58% purity requiring extensive workup. This aqueous method produces crude Iloperidone with >98.8% purity directly from the reactor, which can be easily refined to >99.6% via simple methanol recrystallization without needing activated carbon decolorization.
Q: Is the potassium iodide catalyst essential for this reaction?
A: Yes, potassium iodide acts as a crucial nucleophilic catalyst in this substitution reaction. It facilitates the displacement of the chloride leaving group on the propoxy chain, significantly lowering the activation energy and allowing the reaction to proceed efficiently at moderate temperatures (70-85°C) rather than extreme conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iloperidone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex antipsychotic intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this aqueous surfactant technology are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced HPLC and GC-MS instrumentation to verify that every batch of Iloperidone meets the exacting standards required by global regulatory authorities. Our facility is equipped to handle the specific thermal and mixing requirements of emulsion chemistry, guaranteeing a consistent supply of high-quality material for your drug development programs.
We invite you to collaborate with us to optimize your supply chain for this critical psychiatric medication. By leveraging our technical capabilities, you can access a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this green manufacturing route for your organization. We encourage potential partners to contact our technical procurement team to request specific COA data from our recent pilot batches and to discuss detailed route feasibility assessments tailored to your volume requirements. Let us help you secure a sustainable, cost-effective, and high-quality source of Iloperidone intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
