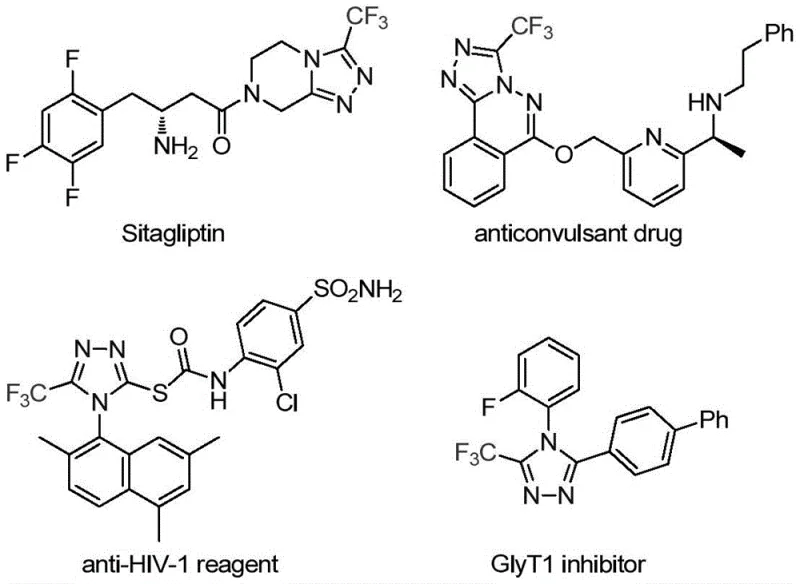
Advanced Catalytic Route for Commercial-Scale Trifluoromethyl Triazole Intermediates in Pharmaceutical Manufacturing
The recently granted Chinese patent CN113307778A introduces a transformative methodology for synthesizing structurally complex trifluoromethyl-substituted triazole intermediates that serve as critical building blocks in modern pharmaceutical development. This innovation addresses longstanding synthetic challenges by establishing a mild-condition catalytic pathway that overcomes the limitations of conventional high-energy cyclization techniques previously required for such heterocyclic systems. The patented process specifically targets the production of pharmacologically relevant compounds like those found in Sitagliptin and other therapeutic agents where precise trifluoromethyl positioning enhances metabolic stability and bioavailability profiles. By leveraging dual molybdenum-copper catalysis under optimized thermal parameters, this approach delivers unprecedented control over regioselectivity while maintaining operational simplicity suitable for industrial implementation. The methodology represents a significant advancement in heterocyclic chemistry that directly responds to the pharmaceutical industry's growing demand for reliable access to fluorinated building blocks with stringent purity requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for trifluoromethyl triazoles have been severely constrained by harsh reaction conditions that necessitate elevated temperatures exceeding 150°C or highly corrosive reagents like strong acids and bases during cyclization steps. These methods frequently produce complex impurity profiles due to uncontrolled side reactions at elevated temperatures, particularly when attempting to synthesize the valuable but challenging-to-access disubstituted variants required for advanced pharmaceutical applications. The existing literature approaches suffer from narrow substrate scope limitations that prevent structural diversification without complete route redesign, forcing medicinal chemists to compromise on molecular design during lead optimization phases. Furthermore, conventional techniques often require multiple protection/deprotection steps that significantly increase both processing time and waste generation while reducing overall atom economy. The inherent instability of certain intermediates under traditional reaction conditions also leads to inconsistent yields that cannot be reliably scaled beyond laboratory quantities without substantial process re-engineering efforts.
The Novel Approach
The patented methodology fundamentally reimagines triazole synthesis through a dual molybdenum-copper catalytic system that operates under remarkably mild thermal conditions between 70°C and 90°C while maintaining exceptional reaction efficiency across diverse substrates. This innovative approach eliminates the need for hazardous reagents by utilizing commercially available starting materials including molybdenum hexacarbonyl and cuprous acetate as catalysts that work synergistically to facilitate the key cycloaddition step. The process demonstrates remarkable substrate flexibility where variations in aryl substituents can be readily accommodated without modifying core reaction parameters, enabling rapid generation of structural analogs through simple precursor substitution. Crucially, the methodology achieves high regioselectivity for the desired disubstituted products that were previously difficult to access through conventional routes. The simplified workup procedure involving standard filtration and chromatography makes this approach particularly suitable for industrial implementation while maintaining excellent yield consistency across different molecular variants.
Mechanistic Insights into Mo/Cu-Catalyzed Triazole Formation
The reaction mechanism initiates with molybdenum hexacarbonyl activating the functionalized isonitrile through coordination chemistry that lowers the energy barrier for subsequent cycloaddition steps while simultaneously preventing undesired oligomerization pathways common with free isonitriles. This activation step creates a nucleophilic species that undergoes copper-promoted [3+2] cycloaddition with trifluoroethylimidoyl chloride through a concerted transition state that ensures precise regiochemical control during ring formation. The dual catalyst system operates through a sophisticated relay mechanism where copper facilitates initial bond formation while molybdenum stabilizes key intermediates throughout the cyclization process. Molecular sieve addition plays a critical role in maintaining anhydrous conditions that prevent hydrolysis of sensitive intermediates while also absorbing byproducts that could otherwise inhibit catalyst activity. The thermal profile between 70°C and 90°C represents an optimal balance where sufficient energy exists for bond rearrangement without triggering decomposition pathways that would compromise product integrity.
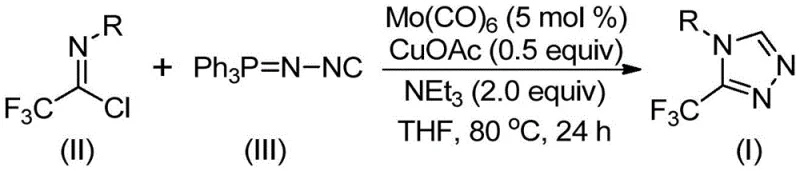
Impurity control is achieved through multiple synergistic mechanisms inherent in this catalytic system where precise temperature regulation prevents thermal degradation pathways while the dual catalyst combination minimizes side reactions through selective activation of specific functional groups. The use of triethylamine as a mild base facilitates proton transfer steps without promoting unwanted elimination reactions that could generate impurities common in stronger basic conditions. The solvent system selection (THF being optimal) provides ideal polarity characteristics that stabilize key intermediates while allowing efficient separation during workup procedures. Molecular sieve incorporation actively removes trace water that could hydrolyze sensitive chloroimine intermediates before cyclization occurs. This multi-faceted approach to impurity management results in consistently high purity profiles exceeding industry standards without requiring additional purification steps beyond standard chromatography.
How to Synthesize Trifluoromethyl Triazole Intermediate Efficiently
This innovative route provides a streamlined pathway for producing high-purity intermediates essential for advanced pharmaceutical development while maintaining operational simplicity suitable for manufacturing environments. The methodology eliminates complex multi-step sequences previously required by leveraging direct cycloaddition chemistry under carefully optimized catalytic conditions that ensure consistent product quality across batches. Detailed standardized synthesis steps are provided below.
- Combine molybdenum hexacarbonyl (5 mol%), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieve, trifluoroethylimidoyl chloride, and functionalized isonitrile in THF solvent under inert atmosphere.
- Heat the reaction mixture to 80°C and maintain stirring for 24 hours while monitoring reaction progression through standard analytical techniques.
- After completion, filter through silica gel and purify by column chromatography using standard protocols to obtain high-purity crystalline product.
Commercial Advantages for Procurement and Supply Chain Teams
The patented process directly addresses critical pain points in pharmaceutical intermediate supply chains by delivering a robust manufacturing solution that enhances both cost efficiency and supply reliability through fundamental process improvements rather than incremental optimizations. This approach transforms what was previously considered a challenging synthetic target into a commercially viable intermediate through strategic catalyst selection and reaction design that aligns with modern manufacturing requirements.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and specialized reaction equipment significantly reduces capital expenditure requirements while simplifying facility qualification processes. The use of commercially available starting materials at optimal stoichiometric ratios minimizes raw material costs without compromising yield or purity profiles. Streamlined purification procedures eliminate multiple processing steps required by conventional methods, substantially reducing both labor costs and solvent consumption while improving overall process efficiency through higher atom economy.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through reliance on globally available commodity chemicals rather than specialized or restricted reagents that create single-point failure risks in traditional supply chains. The robust nature of the reaction tolerates minor variations in raw material quality without affecting final product specifications, providing significant buffer against supplier inconsistencies. Shorter production cycles combined with simplified logistics requirements enable more responsive inventory management strategies that reduce lead time variability while maintaining consistent delivery schedules even during market fluctuations.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability characteristics due to its compatibility with standard manufacturing equipment across all production scales from laboratory validation through commercial manufacturing without requiring specialized engineering modifications. Waste generation is minimized through high atom efficiency and elimination of hazardous byproducts common in alternative synthetic routes, significantly reducing environmental impact while lowering waste treatment costs. The aqueous-compatible workup procedure simplifies effluent management compared to traditional methods requiring specialized solvent recovery systems, enhancing overall sustainability metrics without compromising production throughput.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations derived from extensive analysis of the patented methodology's implementation potential across diverse pharmaceutical manufacturing environments.
Q: How does this method overcome limitations in conventional synthesis of disubstituted triazoles?
A: The patented Mo/Cu dual-catalyst system enables mild reaction conditions (80°C) that prevent decomposition pathways common in traditional high-temperature cyclization methods. This specifically addresses the historical challenge of synthesizing 3,4-disubstituted variants by providing precise control over regioselectivity through substrate design flexibility.
Q: What scalability advantages does the catalytic system offer for commercial production?
A: The process demonstrates exceptional scalability due to its tolerance for standard laboratory equipment and straightforward transition from gram-scale validation to multi-kilogram batches. The absence of specialized pressure reactors or cryogenic conditions eliminates major engineering barriers during plant-scale implementation while maintaining consistent yield profiles across scales.
Q: How does substrate flexibility impact commercial viability for pharmaceutical applications?
A: The broad functional group tolerance allows rapid generation of diverse analogs by simply varying the aryl substituent on the isonitrile component. This enables pharmaceutical developers to efficiently explore structure-activity relationships without redeveloping synthetic routes, significantly accelerating lead optimization timelines while leveraging existing regulatory pathways.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazole Intermediate Supplier
Our company leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities specifically calibrated for fluorinated heterocyclic compounds. This patented methodology represents just one example of our commitment to developing innovative solutions that address complex synthetic challenges while ensuring reliable supply chain performance through vertically integrated manufacturing capabilities across multiple global facilities. Our technical team possesses deep expertise in optimizing catalytic processes like this Mo/Cu system for industrial implementation while maintaining consistent quality standards required by regulatory authorities worldwide.
Request a Customized Cost-Saving Analysis from our technical procurement team to obtain specific COA data and route feasibility assessments tailored to your manufacturing requirements and quality specifications.
