Advanced Triazole Synthesis: Commercially Scalable Route for Fluorinated Pharmaceutical Intermediates
Patent CN114920707B introduces a novel synthetic route for 3-trifluoromethyl substituted 1,2,4-triazole compounds that fundamentally reimagines heterocyclic synthesis through strategic utilization of dimethylformamide (DMF) as both solvent and carbon source. This breakthrough methodology operates effectively under ambient air conditions without requiring anhydrous or anaerobic environments typically mandated by conventional approaches. The process achieves robust yields through molecular iodine-catalyzed tandem cyclization at moderate temperatures (110–130°C) over practical reaction durations (10–15 hours), demonstrating exceptional functional group tolerance across diverse aromatic substrates. By eliminating transition metal catalysts entirely, this innovation addresses critical pain points in pharmaceutical manufacturing including metal contamination risks and complex purification requirements. The strategic design enables direct scalability from laboratory development to commercial production while maintaining stringent purity specifications essential for active pharmaceutical ingredient intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional syntheses of trifluoromethyl-substituted triazoles frequently require stringent anhydrous and anaerobic conditions due to the sensitivity of transition metal catalysts commonly employed in such transformations. These methods typically involve multi-step sequences with low atom economy that generate significant metal-containing waste streams requiring specialized treatment before disposal. The reliance on expensive palladium or copper catalysts not only elevates raw material costs but also necessitates rigorous post-reaction metal removal processes that add complexity to quality control procedures and increase production timelines. Furthermore, conventional approaches often exhibit narrow substrate scope with poor tolerance for functional groups commonly found in drug molecules, limiting their applicability in late-stage diversification strategies essential for modern pharmaceutical development pipelines.
The Novel Approach
The patented methodology overcomes these challenges through an innovative iodine-catalyzed cyclization that operates effectively under ambient air conditions without any moisture-sensitive reagents or specialized handling requirements. By utilizing DMF as both reaction medium and carbon source, the process eliminates the need for external carbon donors while maintaining excellent functional group compatibility across diverse aromatic substrates including those with electron-donating or electron-withdrawing substituents. The reaction proceeds efficiently at moderate temperatures with short processing times demonstrating remarkable robustness even with complex molecular architectures. Crucially, the absence of transition metals removes costly metal scavenging steps while simplifying quality control procedures for final product release without compromising purity standards required for pharmaceutical applications.
Mechanistic Insights into Iodine-Catalyzed Triazole Formation
The reaction mechanism operates through two distinct pathways depending on which component of DMF serves as the carbon source during cyclization. When the formyl group participates in the transformation, it undergoes condensation with trifluoroethyliminohydrazide to form a hydrazone intermediate followed by intramolecular cyclization and elimination of dimethylamine to yield the triazole product. Alternatively, when the N-methyl group acts as carbon donor, DMF first reacts with iodine to generate an amine salt that eliminates hydrogen iodide before nucleophilic addition to the hydrazide precursor. This sequence produces an azadiene intermediate that undergoes intramolecular cyclization and oxidative aromatization to form the final triazole structure under aerobic conditions without requiring additional oxidants.
Impurity control is achieved through the inherent selectivity of the iodine-mediated cyclization process which minimizes side reactions commonly observed in traditional metal-catalyzed systems. The absence of transition metals eliminates potential metal contamination pathways while the moderate reaction temperature prevents thermal decomposition of sensitive functional groups present in complex pharmaceutical intermediates. The use of air as the reaction atmosphere actually facilitates the oxidative aromatization step without requiring additional oxidants that could introduce new impurities or complicate waste stream management. Post-reaction purification via standard column chromatography effectively removes any minor byproducts formed during the tandem cyclization process consistently delivering products with pharmaceutical-grade purity suitable for direct incorporation into drug synthesis sequences without additional refinement steps.
How to Synthesize 3-Trifluoromethyl-1,2,4-triazole Efficiently
This patented synthetic route represents a significant advancement in triazole chemistry by enabling efficient construction of fluorinated heterocycles under operationally simple conditions that leverage commercially available starting materials and standard laboratory equipment while eliminating specialized handling procedures required by conventional approaches. The methodology demonstrates exceptional robustness across diverse substrate classes making it particularly valuable for pharmaceutical manufacturers seeking reliable routes to complex intermediates with stringent purity requirements.
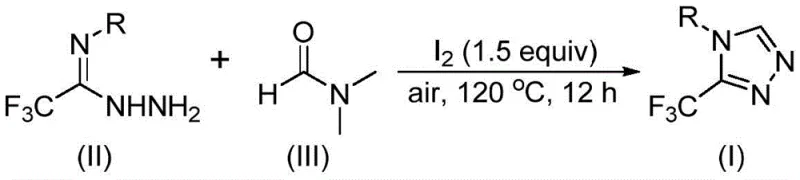
- Combine trifluoroethyliminohydrazide (0.5 mmol) with molecular iodine (1.5 equivalents) in DMF solvent (5–8 mL per mmol) under ambient air conditions.
- Heat the reaction mixture to 120°C and maintain at this temperature for 12 hours with continuous stirring.
- Perform standard workup including filtration through silica gel followed by column chromatography purification to isolate the triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate supply chains by offering a robust solution that enhances both cost efficiency and operational reliability through fundamental process improvements rather than incremental optimizations. The elimination of transition metal catalysts removes significant cost drivers associated with catalyst procurement and post-reaction purification while improving environmental sustainability metrics essential for modern manufacturing operations.
- Cost Reduction in Manufacturing: The strategic elimination of expensive transition metal catalysts significantly reduces raw material costs while avoiding downstream metal removal processes that require specialized equipment and generate hazardous waste streams requiring costly disposal protocols. The use of DMF as both solvent and reactant streamlines material procurement by consolidating multiple inputs into a single commercially available reagent thereby reducing supply chain complexity and associated logistics expenses without compromising product quality.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials including molecular iodine and DMF ensures consistent supply availability even during market fluctuations while eliminating moisture-sensitive reagents reduces vulnerability to humidity-related batch failures during transportation or handling operations across global supply networks.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability from laboratory to commercial production without requiring parameter reoptimization or specialized equipment modifications while substantially reducing hazardous waste generation through elimination of metal catalysts and avoidance of compressed gas dependencies that can create supply chain bottlenecks during scale-up operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented synthesis methodology in pharmaceutical manufacturing operations based on specific patent disclosures regarding process advantages and limitations.
Q: How does this method eliminate transition metal contamination risks in pharmaceutical intermediates?
A: The iodine-catalyzed process operates without transition metals entirely, removing potential contamination pathways that require costly scavenging steps and complex purification protocols in traditional syntheses.
Q: What makes this synthesis particularly suitable for commercial scale-up in existing facilities?
A: The use of air-stable reagents under standard atmospheric conditions eliminates requirements for specialized inert gas equipment while maintaining consistent yields across diverse substrates.
Q: How does DMF's dual role as solvent and carbon source improve process economics?
A: DMF consolidation reduces raw material procurement complexity by serving two functions simultaneously while avoiding additional carbon donors that increase cost and waste streams.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-triazole Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs dedicated to pharmaceutical intermediate manufacturing. This patented methodology exemplifies our commitment to developing innovative solutions that address real-world manufacturing challenges in fluorinated heterocycle synthesis through fundamental process improvements rather than incremental optimizations. As a trusted CDMO partner with deep expertise in complex molecule production we offer comprehensive technical support from route design through commercial-scale manufacturing operations ensuring seamless technology transfer and consistent product quality.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this methodology can optimize your specific production requirements while meeting all regulatory compliance standards. Contact us today to obtain specific COA data and route feasibility assessments tailored to your pharmaceutical intermediate needs through our dedicated technical support channels.
