Revolutionizing Quinclorac Production: Advanced Liquid-Phase Catalytic Oxidation for Commercial Scale-Up
Revolutionizing Quinclorac Production: Advanced Liquid-Phase Catalytic Oxidation for Commercial Scale-Up
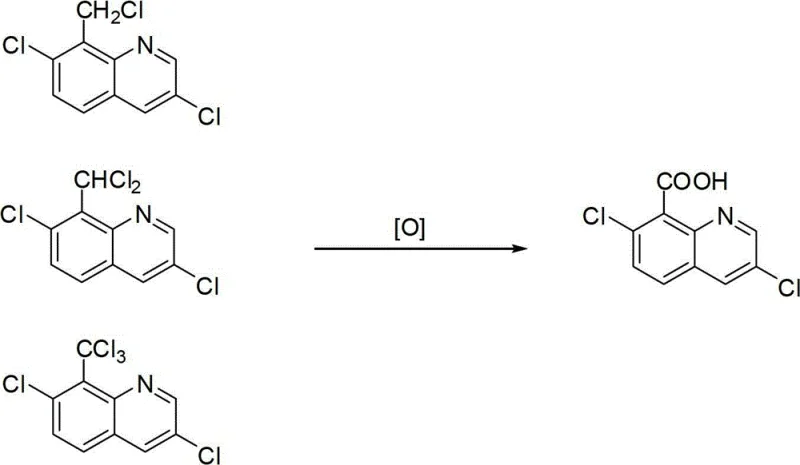
The global demand for high-efficacy herbicides continues to drive innovation in agrochemical intermediate synthesis, particularly for established active ingredients like quinclorac. A pivotal advancement in this domain is documented in Chinese patent CN103420909A, which discloses a novel liquid-phase catalytic oxidation method for preparing 3,7-dichloro-8-quinolinecarboxylic acid. This technology represents a significant departure from traditional stoichiometric oxidation routes, introducing a cobalt-manganese-bromine (Co-Mn-Br) ternary composite catalyst system that operates under moderate pressure and temperature conditions. For R&D directors and process engineers, this patent offers a compelling alternative to legacy methods that rely heavily on corrosive mineral acids. The core innovation lies in the ability to utilize molecular oxygen as the terminal oxidant within an aliphatic carboxylic acid solvent, fundamentally altering the economic and environmental profile of quinclorac manufacturing. By shifting from waste-intensive batch processes to a more catalytic, recyclable workflow, this approach addresses critical pain points regarding waste acid disposal and product purity that have long plagued the industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the carboxyl group in quinclorac has been fraught with significant chemical engineering challenges and environmental liabilities. Prior art, such as US4632696 and German patent DE3108873, typically relies on the oxidation of 3,7-dichloro-8-chloromethylquinoline using concentrated nitric acid in the presence of concentrated sulfuric acid. This conventional pathway suffers from severe drawbacks, primarily the generation of massive quantities of spent acid waste that is difficult and costly to treat. Furthermore, the use of nitric acid as a strong oxidant often leads to the formation of undesirable nitro-substituted byproducts, which compromise the quality of the final agrochemical intermediate and necessitate complex purification steps. Alternative methods involving hydroxylamine hydrochloride followed by hydrolysis also introduce additional unit operations and reagent costs. These legacy processes are not only environmentally unsustainable due to high E-factors but also pose safety risks associated with handling large volumes of hot, concentrated corrosive acids. Consequently, manufacturers face escalating compliance costs and supply chain vulnerabilities linked to the disposal of hazardous chemical waste.
The Novel Approach
In stark contrast, the methodology outlined in CN103420909A introduces a streamlined liquid-phase catalytic oxidation protocol that effectively circumvents the limitations of mineral acid oxidation. By employing a Co-Mn-Br ternary catalyst system, the reaction utilizes molecular oxygen (or air) to oxidize the chloromethyl side chain directly to the carboxylic acid functionality. This transition from stoichiometric oxidants to catalytic aerobic oxidation drastically reduces the chemical load on the downstream wastewater treatment facilities. The process operates in an aliphatic carboxylic acid solvent, preferably acetic acid, which serves as both the reaction medium and a stabilizer for the catalytic species. Crucially, the patent demonstrates that the mother liquor remaining after product isolation can be distilled, dehydrated, and recycled back into the reactor. This closed-loop capability significantly enhances atom economy and reduces raw material consumption. For procurement managers, this shift implies a more robust and cost-effective supply chain, as the reliance on volatile commodity acids is minimized in favor of reusable solvents and regenerable catalyst systems.
Mechanistic Insights into Co-Mn-Br Catalyzed Aerobic Oxidation
The heart of this technological breakthrough is the Co-Mn-Br ternary catalytic cycle, often referred to in industrial chemistry as MC (Mid-Century) oxidation chemistry. In this mechanism, the cobalt and manganese salts act as redox mediators that facilitate the generation of free radicals from the organic substrate. The bromide component plays a critical role as a co-catalyst, assisting in the hydrogen abstraction from the benzylic position of the 3,7-dichloro-8-chloromethylquinoline. Under the specified conditions of 100~255°C and 0.5~3 MPa pressure, the catalyst system activates molecular oxygen to form peroxy radicals, which propagate the oxidation chain reaction. The synergy between cobalt and manganese allows for lower activation energies compared to single-metal systems, ensuring high conversion rates while maintaining selectivity for the carboxylic acid over potential over-oxidation products. The patent specifies optimal molar ratios, such as Co/Mn ranging from 0.1 to 10:1, which fine-tunes the redox potential of the solution. This precise control over the radical environment is what enables the high selectivity observed in the experimental data, preventing the degradation of the sensitive quinoline ring structure.
From an impurity control perspective, this catalytic mechanism offers distinct advantages over nitric acid oxidation. Since the oxidant is molecular oxygen rather than a nitrogen-based species, there is zero risk of nitrating the electron-rich aromatic rings of the quinoline scaffold. Nitro-impurities are notoriously difficult to remove and can be toxic, making their elimination a priority for regulatory compliance in agrochemical registration. Furthermore, the mild acidity of the acetic acid solvent compared to sulfuric acid minimizes acid-catalyzed side reactions such as polymerization or ring-opening. The patent data indicates that the crude product obtained from this reaction already possesses high purity, which simplifies the downstream purification process. Typically, a single recrystallization step from ethanol is sufficient to elevate the purity to ≥97%, as demonstrated in the provided examples where purities of 97.9% were achieved. This mechanistic cleanliness translates directly into operational efficiency, reducing the need for extensive chromatography or multiple recrystallizations that would otherwise erode overall yield.
How to Synthesize Quinclorac Efficiently
Implementing this liquid-phase catalytic oxidation route requires careful attention to reactor design and parameter control to ensure safety and reproducibility. The process begins with the preparation of the substrate, 3,7-dichloro-8-chloromethylquinoline, which is dissolved in glacial acetic acid along with the cobalt, manganese, and bromide catalyst sources. The reaction is conducted in a pressurized vessel, such as a titanium autoclave, capable of withstanding the oxygen pressure and corrosive environment. Detailed standard operating procedures regarding catalyst activation, oxygen feed rates, and temperature ramping profiles are critical for maximizing the turnover number of the catalyst. While the patent provides specific exemplary ranges, scaling this chemistry requires rigorous hazard analysis due to the presence of oxygen and organic solvents at elevated temperatures. The following guide outlines the fundamental steps derived from the patent claims to assist process chemists in replicating this high-efficiency synthesis.
- Prepare the reaction system by dissolving 3,7-dichloro-8-chloromethylquinoline in an aliphatic carboxylic acid solvent, preferably acetic acid.
- Add the Co-Mn-Br ternary composite catalyst system, ensuring the molar ratio of Co/Mn is between 0.1-10: 1 and Br/(Co+Mn) is 0.01-10:1.
- Introduce oxygen-containing gas at 100-255°C and 0.5-3 MPa pressure, then isolate and recrystallize the solid product to achieve ≥97% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the adoption of the Co-Mn-Br catalytic oxidation process offers substantial strategic benefits. The primary value driver is the drastic simplification of the waste management infrastructure. By eliminating the generation of spent sulfuric and nitric acid mixtures, manufacturers can avoid the high costs associated with neutralization, disposal, and environmental permitting. This reduction in hazardous waste volume directly correlates to lower operational expenditures and a smaller environmental footprint, aligning with modern green chemistry mandates. Additionally, the ability to recycle the acetic acid solvent and the catalyst components creates a more circular production model. This reduces the exposure to raw material price volatility, as the consumption of fresh solvent per kilogram of product is significantly diminished. For supply chain heads, this means a more predictable cost structure and reduced dependency on external waste treatment vendors.
- Cost Reduction in Manufacturing: The transition to a catalytic aerobic oxidation system fundamentally alters the cost basis of quinclorac production. Unlike stoichiometric oxidants which are consumed in large excess and generate equivalent amounts of waste, the Co-Mn-Br catalyst system operates at low loading levels and facilitates the use of cheap molecular oxygen. The elimination of expensive and hazardous mineral acids removes a major cost center from the P&L statement. Furthermore, the simplified workup procedure, which avoids complex extraction or neutralization steps required for acid waste, reduces labor and utility consumption. The high selectivity of the reaction ensures that raw material inputs are converted efficiently into the desired product rather than lost to byproducts, thereby improving the effective yield and lowering the cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Relying on commodity gases like oxygen or air and recyclable solvents enhances the resilience of the supply chain. Traditional methods dependent on concentrated nitric acid are subject to logistical constraints and regulatory scrutiny regarding the transport of hazardous corrosives. By shifting to a process that generates less hazardous waste and utilizes safer reagents, manufacturers can mitigate the risk of production stoppages due to supply disruptions or regulatory crackdowns. The robustness of the catalyst system also allows for longer campaign runs without frequent catalyst replenishment, ensuring consistent output volumes. This reliability is crucial for meeting the demands of downstream formulators who require steady supplies of high-purity agrochemical intermediates to maintain their own production schedules.
- Scalability and Environmental Compliance: The liquid-phase nature of this reaction makes it highly amenable to scale-up in standard stirred-tank reactors, facilitating the transition from pilot plant to commercial production. The absence of solid oxidants or highly viscous acid mixtures improves heat and mass transfer characteristics, allowing for better temperature control and safety at larger scales. From a compliance standpoint, the reduction in waste acid discharge simplifies the permitting process for new manufacturing facilities or the retrofitting of existing ones. The process aligns with increasingly stringent global environmental regulations regarding effluent quality and carbon emissions. By adopting this cleaner technology, companies can future-proof their operations against tightening environmental standards, avoiding potential fines and reputational damage associated with pollution.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic oxidation technology. These insights are derived directly from the experimental data and claims presented in patent CN103420909A, providing a factual basis for decision-making. Understanding the nuances of catalyst recovery, solvent selection, and impurity profiles is essential for evaluating the feasibility of this route for your specific manufacturing context. We encourage technical teams to review these details closely when assessing process upgrades or new vendor qualifications.
Q: What are the advantages of the Co-Mn-Br catalytic system over traditional nitric acid oxidation?
A: The Co-Mn-Br system eliminates the use of concentrated nitric and sulfuric acids, thereby avoiding the formation of nitro-impurities and significantly reducing the volume of hazardous waste acid requiring disposal.
Q: Can the solvent and mother liquor be recycled in this process?
A: Yes, the patent explicitly states that the separated mother liquor can be distilled and dehydrated to recover the acetic acid solvent, which can then be reused in subsequent batches, enhancing process sustainability.
Q: What purity levels can be achieved with this oxidation method?
A: Through simple ethanol recrystallization of the crude product obtained from this catalytic oxidation, purity levels exceeding 97% (specifically up to 97.9% in examples) can be consistently achieved.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinclorac Supplier
The technological potential of the Co-Mn-Br catalytic oxidation route for quinclorac synthesis is immense, offering a pathway to cleaner and more economical herbicide production. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such advanced patent methodologies into commercial reality. As a seasoned CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully transferred to industrial manufacturing. Our facilities are equipped with state-of-the-art high-pressure reactors and rigorous QC labs capable of meeting stringent purity specifications required by global agrochemical markets. We understand the critical importance of consistency and quality in the supply of active pharmaceutical and agrochemical ingredients.
We invite you to collaborate with us to optimize your supply chain for quinclorac and related heterocyclic intermediates. Our team is ready to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. By leveraging our process development capabilities, we can help you realize the economic benefits of this catalytic technology. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to drive efficiency and sustainability in your agrochemical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
