Advanced Manufacturing of 3-Chloro-2-Methylaniline for High-Efficiency Herbicide Synthesis
The global demand for high-efficiency herbicides, particularly quinolinecarboxylic acid derivatives like quinclorac, has placed significant scrutiny on the supply chain quality of their key precursors. Patent CN100427458C introduces a transformative production process for 3-chloro-2-methylaniline, a critical agrochemical intermediate, addressing long-standing challenges in purity and environmental compliance. This innovative methodology utilizes a sulfur-mediated reduction system in a polar aprotic solvent environment, offering a robust alternative to traditional heavy metal-catalyzed pathways. By leveraging elemental sulfur and sodium bicarbonate at moderate temperatures ranging from 110°C to 140°C, the process achieves exceptional chemoselectivity, ensuring the nitro group is reduced to an amine without compromising the sensitive aryl-chloride bond. For R&D directors and procurement strategists, this technology represents a pivotal shift towards safer, more sustainable, and economically viable manufacturing protocols for complex aromatic amines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-chloro-2-methylaniline has been plagued by significant technical and operational hurdles that compromise both yield and supply chain stability. Traditional methods often rely on catalytic hydrogenation using Raney Nickel, which necessitates rigorous safety protocols due to the pyrophoric nature of the catalyst and carries a high risk of hydrodechlorination, leading to unwanted dehalogenated byproducts that are difficult to separate. Alternatively, the iron powder-hydrochloric acid reduction method, while chemically effective, generates voluminous quantities of toxic iron sludge and acidic wastewater, creating severe environmental liabilities and escalating waste treatment costs for manufacturers. Furthermore, direct chlorination routes of 2-methylaniline typically result in complex isomeric mixtures, such as 3-chloro and 5-chloro isomers in unfavorable ratios, requiring energy-intensive and yield-loss-prone separation processes that undermine overall process efficiency.
The Novel Approach
The novel approach detailed in the patent data circumvents these legacy issues by employing a heterogeneous reduction system utilizing inexpensive elemental sulfur and sodium bicarbonate in solvents like N,N-dimethylformamide (DMF) or N,N-dimethylacetamide (DMAC). This method operates under atmospheric pressure and moderate thermal conditions, eliminating the need for specialized high-pressure hydrogenation reactors and the associated safety risks of handling compressed hydrogen gas. The use of sodium bicarbonate serves a dual purpose: it acts as a mild base to neutralize acidic byproducts generated during the redox process and helps drive the reaction equilibrium towards the desired amine product. This results in a streamlined workflow where the reaction mixture can be simply filtered to remove solid sulfur residues, followed by direct vacuum distillation to isolate the target molecule with high purity, significantly reducing the complexity of downstream processing.
Mechanistic Insights into Sulfur-Mediated Nitro Reduction
The core of this technological advancement lies in the unique redox mechanism facilitated by elemental sulfur in a basic polar medium. In this system, sulfur acts as the primary reducing agent, undergoing oxidation while transferring electrons to the nitro group of the 2-chloro-6-nitrotoluene substrate. The reaction likely proceeds through a series of intermediate nitrogen-oxygen species, eventually culminating in the formation of the primary amine functionality. The presence of sodium bicarbonate is crucial as it buffers the reaction environment, preventing the accumulation of acidic species that could otherwise protonate the amine product or degrade the solvent system. This buffering capacity ensures that the reaction kinetics remain favorable throughout the extended reaction time of 10 to 24 hours, allowing for complete conversion of the starting material even at the relatively mild temperature range of 110°C to 140°C.
From an impurity control perspective, this mechanism offers superior chemoselectivity compared to transition metal catalysis. The absence of active metal surfaces prevents the oxidative addition of the aryl-chloride bond, a common failure mode in nickel or palladium-catalyzed hydrogenations that leads to dechlorinated impurities. Consequently, the impurity profile of the crude product is significantly cleaner, dominated primarily by unreacted starting material or minor sulfur-containing organics that are easily removed during the fractional distillation step. This high level of selectivity is paramount for pharmaceutical and agrochemical applications, where trace impurities can adversely affect the biological activity and regulatory approval of the final herbicide product.
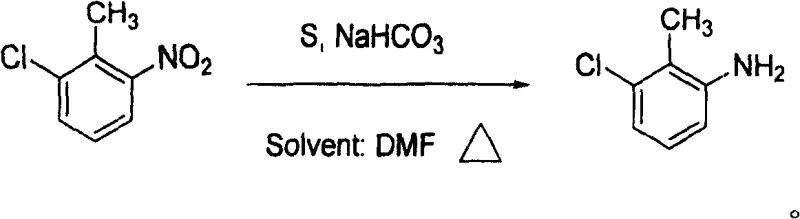
How to Synthesize 3-Chloro-2-Methylaniline Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and thermal profiles to maximize yield and minimize side reactions. The process begins with the careful charging of the reactor with the polar aprotic solvent, followed by the sequential addition of the nitro-aromatic substrate, elemental sulfur, and the bicarbonate base. Maintaining the molar equivalent ratio of 2-chloro-6-nitrotoluene to sulfur between 1:3 and 1:5 is critical to ensure sufficient reducing power without excessive reagent waste. Detailed standardized operating procedures regarding heating rates, agitation speeds, and distillation cut points are essential for reproducibility and are outlined in the technical guidelines below.
- Charge the reactor with DMF or DMAC solvent, 2-chloro-6-nitrotoluene, elemental sulfur (3-5 equivalents), and sodium bicarbonate (3-5 equivalents).
- Heat the reaction mixture to 110-140°C and stir continuously for 10-24 hours to ensure complete reduction of the nitro group.
- Filter the hot reaction mixture to remove solid byproducts, then subject the filtrate to vacuum fractional distillation to isolate the pure amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this sulfur-based reduction technology offers profound strategic advantages that extend beyond simple chemical transformation. The substitution of expensive and hazardous catalysts with commodity-grade sulfur and bicarbonate drastically alters the cost structure of the manufacturing process, removing the volatility associated with precious metal markets and specialized catalyst regeneration services. Furthermore, the simplification of the workup procedure, which replaces complex extractions and sludge handling with a straightforward filtration and distillation sequence, significantly reduces labor hours and utility consumption per batch. This operational efficiency translates directly into improved margin potential and a more resilient supply chain capable of responding rapidly to market fluctuations in herbicide demand.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts such as Raney Nickel removes the substantial costs associated with catalyst purchase, storage, and the rigorous removal of trace metal residues from the final product to meet regulatory specifications. Additionally, the use of cheap, non-toxic sulfur as the reducing agent replaces costly hydrogen gas or iron powder, leading to a fundamental decrease in raw material expenditure. The simplified post-treatment process also reduces the consumption of solvents and water required for washing and purification steps, further driving down the variable costs of production without compromising the high purity standards required for agrochemical intermediates.
- Enhanced Supply Chain Reliability: By relying on widely available and stable raw materials like sulfur and sodium bicarbonate, manufacturers can mitigate the risks of supply disruptions often associated with specialized catalysts or high-pressure gases. The robustness of the reaction conditions, which do not require high-pressure vessels or cryogenic cooling, allows for production in a broader range of facilities, thereby diversifying the potential manufacturing base and reducing geopolitical or logistical bottlenecks. This flexibility ensures a consistent and reliable flow of 3-chloro-2-methylaniline to downstream formulators, safeguarding the production schedules of finished herbicide products against upstream volatility.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard stirred-tank reactors and atmospheric pressure operations, facilitating a seamless transition from pilot plant to commercial-scale production without the need for capital-intensive equipment upgrades. From an environmental standpoint, the drastic reduction in heavy metal waste and acidic effluents aligns with increasingly stringent global environmental regulations, minimizing the liability and cost of waste disposal. This green chemistry profile not only enhances the corporate sustainability image but also future-proofs the manufacturing asset against tightening regulatory frameworks regarding industrial emissions and hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfur-mediated reduction process. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, aiming to clarify the operational benefits and chemical rationale for adopting this methodology. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing production lines or for procurement specialists assessing the long-term value proposition of suppliers utilizing this technology.
Q: Why is sulfur reduction preferred over catalytic hydrogenation for 3-chloro-2-methylaniline?
A: Sulfur reduction avoids the risk of hydrodechlorination, a common side reaction in catalytic hydrogenation where the chlorine atom is inadvertently removed. Additionally, it eliminates the need for expensive pyrophoric catalysts like Raney Nickel and high-pressure equipment.
Q: What is the typical purity achievable with this sulfur-based process?
A: According to patent data, this process consistently achieves a GC purity of 99%, which is critical for the subsequent synthesis of quinclorac herbicides where impurities can significantly affect efficacy.
Q: How does this method impact waste treatment compared to iron powder reduction?
A: Unlike the traditional iron powder-hydrochloric acid method which generates massive amounts of iron sludge and acidic wastewater, the sulfur-bicarbonate method produces solid sulfur residues that are easily filtered, drastically simplifying three-waste treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Chloro-2-Methylaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficiency of your herbicide synthesis depends heavily on the quality and consistency of your intermediate supply. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated sulfur-reduction chemistry described in patent CN100427458C is executed with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, delivering 3-chloro-2-methylaniline that meets the exacting standards required for high-performance agrochemical formulations.
We invite you to collaborate with our technical team to explore how this optimized manufacturing route can enhance your supply chain resilience and cost efficiency. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced production capabilities can support your strategic goals in the global agrochemical market.
