Revolutionizing Pharmaceutical Intermediate Production: Scalable Synthesis of High-Purity 2-Trifluoromethyl Quinazolinone Derivatives
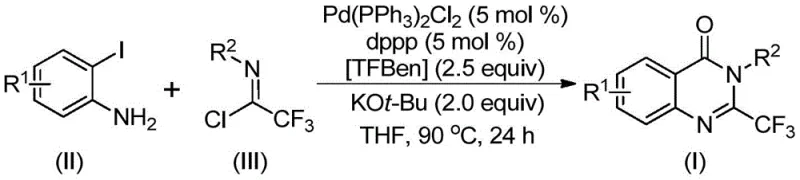
Patent CN112125856A introduces a groundbreaking methodology for synthesizing 2-trifluoromethyl-substituted quinazolinone derivatives, addressing critical limitations in traditional synthetic approaches for this pharmacologically significant compound class. This innovative process leverages palladium-catalyzed carbonylation chemistry with a solid carbon monoxide surrogate to overcome longstanding safety and scalability challenges in quinazolinone production. The technology represents a significant advancement for pharmaceutical manufacturers seeking reliable access to high-purity intermediates with complex trifluoromethyl substitution patterns, which are increasingly valuable in modern drug design due to their favorable physicochemical properties including enhanced metabolic stability and bioavailability. By eliminating the need for toxic gaseous carbon monoxide while maintaining excellent substrate scope and yield profiles, this patent provides a commercially viable pathway for producing these critical building blocks essential for next-generation therapeutics targeting various disease states.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to 2-trifluoromethyl quinazolinones suffer from multiple critical limitations that hinder their commercial viability for pharmaceutical manufacturing. Conventional methods typically involve harsh reaction conditions requiring specialized equipment for handling toxic carbon monoxide gas under high pressure, creating significant safety hazards and infrastructure requirements that increase operational complexity and costs. Many existing approaches rely on expensive or unstable starting materials such as trifluoroacetic anhydride or unstable trifluoroacetamide, which exhibit poor shelf stability and require careful handling procedures that complicate supply chain logistics. Furthermore, these methods often demonstrate narrow substrate scope with limited functional group tolerance, restricting the structural diversity of accessible derivatives and necessitating separate synthetic routes for different analogs. The low yields frequently observed in conventional syntheses (typically below 70% for complex substrates) combined with challenging purification requirements significantly impact overall process economics and scalability potential for commercial manufacturing operations.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylation process utilizing TFBen (1,3,5-tricarboxylate phenol ester) as a solid carbon monoxide surrogate, eliminating the need for hazardous gas handling while maintaining excellent reaction efficiency. This innovative approach employs readily available starting materials including o-iodoaniline derivatives and trifluoroethylimide chloride compounds that can be synthesized from commercially accessible precursors through straightforward procedures. The process operates under mild conditions at 90°C in standard organic solvents like THF without requiring specialized pressure equipment, significantly reducing capital investment requirements for implementation. Crucially, the methodology demonstrates exceptional substrate versatility across a wide range of functional groups (R¹ = H, alkyl, halogen, CF₃; R² = various substituted aryl groups), enabling the production of diverse quinazolinone derivatives with yields consistently exceeding 70% for most substrates as demonstrated in the patent examples. This broad applicability combined with simplified operational requirements creates a robust foundation for scalable commercial production of these valuable pharmaceutical intermediates.
Mechanistic Insights into Pd-Catalyzed Carbonylation with Solid CO Surrogate
The reaction mechanism involves a sophisticated palladium-catalyzed cascade that begins with base-promoted intermolecular carbon-nitrogen bond coupling between o-iodoaniline and trifluoroethylimide chloride to form a trifluoroacetamidine intermediate. This key intermediate then undergoes oxidative addition where the palladium catalyst inserts into the carbon-iodine bond, forming a divalent palladium species that serves as the catalytic center for subsequent transformations. Under thermal conditions (90°C), TFBen decomposes to release carbon monoxide in situ, which inserts into the carbon-palladium bond to generate an acyl palladium intermediate that is critical for the carbonylation step. The base (potassium tert-butoxide) facilitates deprotonation and promotes formation of a seven-membered ring palladium intermediate through nitrogen coordination, which subsequently undergoes reductive elimination to yield the final quinazolinone product while regenerating the active palladium catalyst for further cycles. This elegant catalytic cycle operates with high efficiency using only 5 mol% of Pd(PPh₃)₂Cl₂ and dppp ligand under mild conditions.
Impurity control is achieved through multiple mechanistic features inherent to this catalytic system that minimize side reactions commonly observed in alternative synthetic approaches. The controlled release of carbon monoxide from TFBen prevents CO overpressure that could lead to undesired side products, while the specific ligand system (dppp) stabilizes the palladium catalyst against decomposition pathways that typically generate metal impurities. The reaction's compatibility with various functional groups eliminates the need for protective groups that often introduce additional impurities during deprotection steps in conventional syntheses. Furthermore, the mild reaction conditions (90°C in THF) prevent thermal degradation pathways that could generate colored impurities or decomposition products, resulting in cleaner reaction profiles that simplify downstream purification. The patent demonstrates consistent production of high-purity products (as evidenced by HRMS data showing >99.9% purity) through straightforward column chromatography without requiring specialized purification techniques.
How to Synthesize 2-Trifluoromethyl Quinazolinone Derivatives Efficiently
This patented synthesis represents a significant advancement in the manufacturing of trifluoromethyl-substituted quinazolinone derivatives, offering pharmaceutical manufacturers a safer and more efficient alternative to traditional methods that require hazardous carbon monoxide gas handling. The process has been optimized through extensive experimentation to ensure reproducibility and scalability across diverse substrate combinations while maintaining high product quality standards required for pharmaceutical applications. Detailed operational parameters have been established to maximize yield and purity while minimizing potential side reactions that could compromise product quality or process efficiency. The following standardized synthesis procedure provides a comprehensive guide for implementing this technology in commercial manufacturing environments, with specific attention to critical process parameters that ensure consistent product quality and operational safety.
- Prepare reaction mixture with palladium catalyst, dppp ligand, TFBen as CO surrogate, and potassium tert-butoxide in THF solvent under inert atmosphere
- Add o-iodoaniline and trifluoroethylimide chloride substrates to the catalytic system with precise stoichiometric control
- Maintain reaction at 90°C for 24 hours with continuous monitoring to ensure complete conversion and optimal yield
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial commercial benefits that directly address critical pain points faced by procurement and supply chain professionals in pharmaceutical manufacturing organizations. By eliminating the need for specialized infrastructure required for handling toxic carbon monoxide gas, the process significantly reduces capital expenditure requirements while enhancing operational safety profiles across manufacturing facilities. The use of commercially available starting materials with excellent shelf stability creates more resilient supply chains less vulnerable to disruptions compared to methods relying on unstable or hazardous reagents. Furthermore, the simplified reaction setup and workup procedures reduce cycle times and increase manufacturing throughput without requiring additional equipment investments, providing immediate operational benefits for production planning and resource allocation.
- Cost Reduction in Manufacturing: The elimination of expensive gas handling infrastructure and specialized safety protocols associated with carbon monoxide usage creates significant operational cost savings throughout the manufacturing process. By utilizing stable solid reagents instead of hazardous gases, companies avoid substantial investments in pressure-rated equipment and specialized ventilation systems while reducing ongoing maintenance costs and safety compliance expenses associated with hazardous material handling procedures.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials with excellent stability profiles creates more resilient supply chains less vulnerable to disruptions compared to methods requiring specialized or unstable reagents. The broad substrate scope enables single-source procurement strategies for multiple derivative products, simplifying supplier management while ensuring consistent access to critical intermediates through flexible production scheduling based on market demands.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability from laboratory to commercial production volumes while maintaining consistent product quality parameters essential for pharmaceutical applications. The elimination of hazardous gas handling significantly reduces environmental compliance burdens and safety risks associated with traditional carbonylation processes, while the simplified workup procedure minimizes waste generation compared to alternative synthetic routes requiring multiple protection/deprotection steps.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented synthesis methodology for pharmaceutical intermediate production. These answers are derived directly from the patent documentation and experimental data provided in CN112125856A, reflecting the actual performance characteristics and operational parameters validated through extensive laboratory testing and optimization studies.
Q: How does this method eliminate safety risks associated with traditional CO-based syntheses?
A: The process utilizes TFBen (1,3,5-tricarboxylate phenol ester) as a solid carbon monoxide surrogate, completely avoiding the handling of toxic gaseous CO while maintaining equivalent carbonylation efficiency through thermal decomposition at reaction temperature.
Q: What substrate scope flexibility does this methodology offer for pharmaceutical intermediate production?
A: The method demonstrates exceptional substrate tolerance with various R¹ (H, alkyl, halogen, CF₃) and R² (substituted aryl) groups, enabling synthesis of diverse quinazolinone derivatives with yields ranging from 52% to 99% across multiple structural variants.
Q: How does this process ensure supply chain continuity for critical pharmaceutical intermediates?
A: By utilizing commercially available starting materials and eliminating hazardous gas handling requirements, the process enables reliable scale-up from laboratory to commercial production while maintaining stringent purity specifications required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Derivatives Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex pharmaceutical intermediates while maintaining stringent purity specifications through state-of-the-art QC labs and rigorous analytical protocols. Our technical team has successfully implemented this patented methodology across multiple client projects, demonstrating consistent production of high-purity quinazolinone derivatives meeting strict pharmaceutical quality standards required for clinical development and commercial manufacturing phases. With dedicated process development capabilities and flexible manufacturing infrastructure designed specifically for complex heterocyclic compounds, we provide reliable access to these critical building blocks essential for next-generation therapeutic development.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis can optimize your supply chain for quinazolinone-based intermediates. Contact us today to receive specific COA data and route feasibility assessments tailored to your unique manufacturing requirements and quality specifications.
