Advanced Manufacturing of High-Purity Finasteride Intermediates via Optimized Steroid Functionalization
Advanced Manufacturing of High-Purity Finasteride Intermediates via Optimized Steroid Functionalization
The pharmaceutical industry constantly seeks robust synthetic routes for critical active pharmaceutical ingredient (API) precursors, and patent CN101863954A presents a significant advancement in the preparation of N-tert-butyl-4-aza-5 alpha-androstane-3-ketone-17 beta-formamide. This compound serves as a pivotal intermediate in the synthesis of Finasteride, a widely prescribed medication for benign prostatic hyperplasia and androgenic alopecia. The disclosed methodology outlines a comprehensive six-step transformation starting from readily available 3 beta-hydroxyl-androstane-5-alkene-17 beta-carboxylic acid. By integrating esterification, Oppenauer oxidation, oxidative cleavage, cyclization, catalytic hydrogenation, and a specialized amidation step, this process achieves a total recovery rate of 44-51% with exceptional purity profiles exceeding 99% HPLC content. For R&D directors and procurement specialists, this route represents a strategic opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials with optimized cost structures and enhanced safety protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-aza-androstane derivatives has been plagued by inefficiencies that hinder large-scale commercial viability. Prior art, such as the methods disclosed in Chinese patent CN101531698A, often relied on starting materials like 3-carbonyl-4-androstene-17 beta-carboxylate methyl ester and suffered from cumulative yield losses, reporting a total recovery of merely 52% over four critical steps. Furthermore, traditional amidation strategies frequently necessitated the use of sodium hydride, a pyrophoric and highly hazardous reagent that imposes severe safety constraints and requires specialized handling equipment, thereby inflating operational expenditures. The reliance on expensive solvents like tert-butanol in oxidative cleavage steps further exacerbated production costs, while impurity profiles often required extensive downstream purification to meet stringent regulatory standards for hormonal therapies. These legacy processes created bottlenecks in the supply chain, limiting the ability to scale production rapidly in response to market demand fluctuations.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN101863954A introduces a rationalized synthetic pathway that systematically addresses these historical pain points through chemical innovation and process optimization. By initiating the sequence from 3 beta-hydroxyl-androstane-5-alkene-17 beta-carboxylic acid, the method leverages a highly efficient esterification step yielding 95-97%, setting a strong foundation for subsequent transformations. The strategic replacement of tert-butanol with acetone in the oxidative cleavage stage not only drastically reduces solvent costs but also enhances the reaction yield by approximately 10% in that specific unit operation. Moreover, the substitution of dangerous sodium hydride with a tert-butylamine-based magnesium halide reagent in the final amidation step eliminates significant safety risks while maintaining high conversion rates. This holistic redesign ensures that intermediates remain stable and high-purity throughout the sequence, facilitating a smoother transition from laboratory bench to industrial reactor.
Mechanistic Insights into Oppenauer Oxidation and Oxidative Cleavage
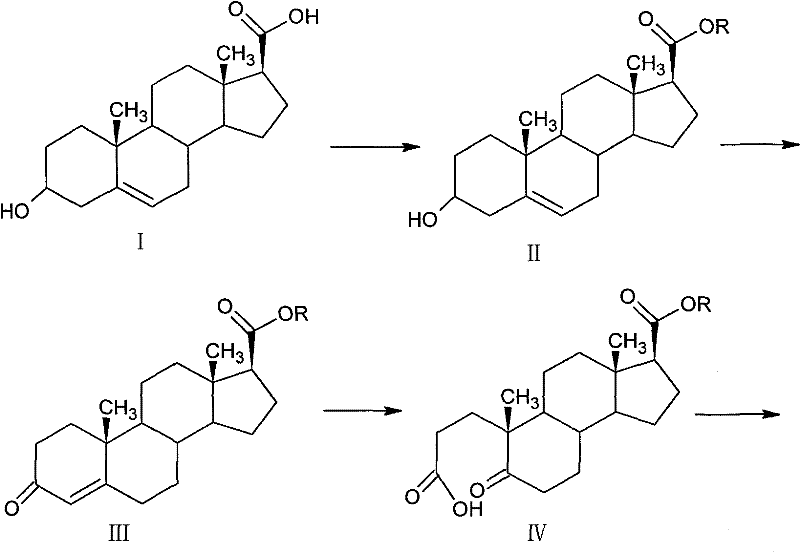
The core of this synthetic strategy relies on the precise execution of the Oppenauer oxidation, a reversible reaction that selectively oxidizes secondary alcohols to ketones using aluminum isopropoxide and a ketone acceptor, typically cyclohexanone. In this specific application, the conversion of the 3-beta-hydroxyl group in Compound II to the 3-ketone in Compound III is driven by the removal of water and the use of excess cyclohexanone, pushing the equilibrium towards the desired enone product with yields reaching 78-80%. This step is critical for establishing the conjugated system necessary for the subsequent oxidative ring opening. Following this, the process employs a rigorous oxidative cleavage using sodium periodate and potassium permanganate in an acetone-sodium bicarbonate aqueous system. This dual-oxidant system effectively cleaves the carbon-carbon double bond of the steroid nucleus, generating the ring-opened dicarbonyl intermediate (Compound IV) with remarkable selectivity and a yield of 90-92%, minimizing the formation of over-oxidized byproducts that often complicate steroid chemistry.
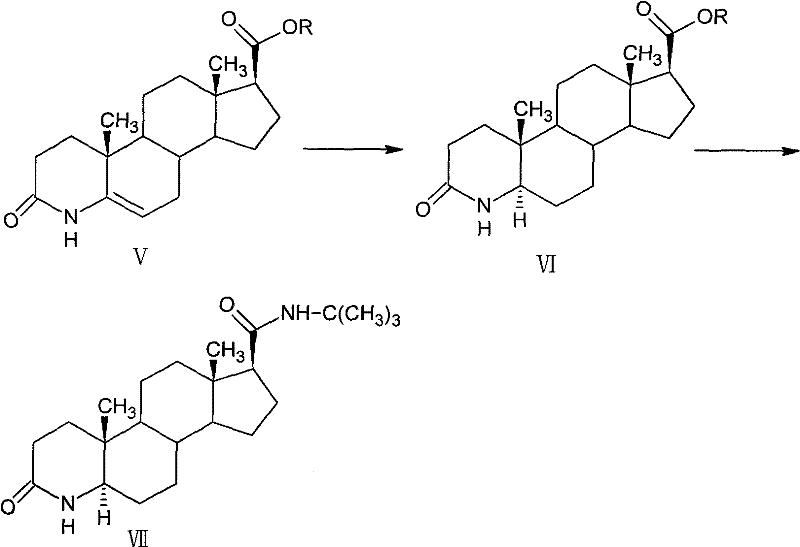
Following the ring opening, the mechanism shifts to a cyclization event where the ring-opened product reacts with ammonium acetate in glacial acetic acid to form the 4-aza-androst-5-ene structure (Compound V). This intramolecular condensation is highly efficient, achieving yields of 89-91% and establishing the nitrogen atom at the 4-position which is characteristic of 5-alpha-reductase inhibitors. The subsequent catalytic hydrogenation using palladium on carbon (Pd/C) under controlled pressure (normal to 1.5MPa) stereoselectively reduces the 5,6-double bond to yield the 5-alpha-isomer (Compound VI) with a content greater than 96% and minimal 5-beta-isomer contamination (≤3%). Finally, the nucleophilic attack of the tert-butylamine magnesium halide reagent on the ester carbonyl of Compound VI completes the synthesis. This Grignard-type amidation proceeds under mild conditions in inert solvents like THF, avoiding the harsh conditions of traditional methods and ensuring the final product (Compound VII) maintains an HPLC purity of over 99%.
How to Synthesize N-tert-butyl-4-aza-5 alpha-androstane-3-ketone-17 beta-formamide Efficiently
Executing this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and safety. The process begins with the acid-catalyzed esterification of the starting carboxylic acid, followed by the moisture-sensitive Oppenauer oxidation which demands anhydrous conditions and careful temperature control during reflux. The oxidative cleavage step utilizes a biphasic system that requires precise pH adjustment post-reaction to isolate the ring-opened acid efficiently. Subsequent cyclization and hydrogenation steps are robust but require monitoring of isomer ratios to ensure the correct 5-alpha stereochemistry is maintained. The final amidation is the most critical step for safety and yield, requiring the in-situ generation of the magnesium amide reagent before addition to the steroid ester. For a detailed breakdown of the specific reagent quantities, reaction times, and workup procedures for each of these six stages, please refer to the standardized synthesis guide below.
- Esterify 3 beta-hydroxyl-androstane-5-alkene-17 beta-carboxylic acid with methanol or ethanol under acid catalysis to form the corresponding ester.
- Perform Oppenauer oxidation using aluminum isopropoxide and cyclohexanone to convert the 3-hydroxyl group to a ketone.
- Execute oxidative cleavage with sodium periodate and potassium permanganate, followed by cyclization with ammonium acetate, hydrogenation, and final amidation with tert-butylamine magnesium halide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible operational improvements and risk mitigation strategies. The elimination of sodium hydride from the process flow removes a major hazard classification from the manufacturing site, significantly lowering insurance premiums and safety compliance costs associated with storing and handling pyrophoric materials. Furthermore, the switch to acetone as a primary solvent for the oxidative cleavage step leverages a commodity chemical with a stable global supply chain, insulating production from the price volatility often seen with specialized solvents like tert-butanol. The high purity of intermediates at each stage reduces the need for extensive recrystallization or chromatographic purification, thereby shortening cycle times and increasing overall plant throughput. These factors combine to create a manufacturing process that is not only economically superior but also more resilient to external supply shocks.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by both raw material optimization and yield enhancement. By replacing expensive solvents with cost-effective alternatives like acetone and eliminating the need for hazardous reagents that require special disposal protocols, the direct cost of goods sold is substantially reduced. Additionally, the improved yields in the oxidative cleavage and amidation steps mean that less starting material is wasted, maximizing the output per batch. The high selectivity of the hydrogenation step minimizes the loss of valuable steroid backbone to unwanted isomers, ensuring that the majority of the input mass is converted into saleable high-purity intermediate. This efficiency allows for competitive pricing models without sacrificing margin, making it an attractive option for cost-sensitive generic drug manufacturers.
- Enhanced Supply Chain Reliability: Stability is a key concern for long-term supply contracts, and this process excels by generating intermediates that are chemically stable and easy to store. The robustness of the reaction conditions, which utilize standard industrial equipment like reflux condensers and hydrogenation autoclaves, means that production can be easily replicated across different manufacturing sites without requiring bespoke infrastructure. The avoidance of air-sensitive or moisture-sensitive reagents in the later stages simplifies logistics and storage requirements, reducing the risk of batch failures due to environmental exposure. This reliability ensures consistent delivery schedules, allowing downstream API manufacturers to plan their production runs with greater confidence and reduced safety stock requirements.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns well with green chemistry principles by reducing the use of hazardous substances and improving atom economy. The aqueous workup procedures and the ability to recover and recycle solvents like acetone and toluene minimize the volume of organic waste generated per kilogram of product. The scalability is proven by the use of standard unit operations that are well-understood in the fine chemical industry, allowing for seamless scale-up from pilot plant to multi-ton commercial production. This ease of scaling, combined with a cleaner waste profile, facilitates faster regulatory approvals and reduces the environmental footprint of the manufacturing facility, aligning with the sustainability goals of modern pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived directly from the comparative data and experimental examples provided in the patent documentation to clarify the specific advantages over legacy methods. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or for procurement teams negotiating supply agreements based on quality specifications.
Q: What is the total yield of the new synthesis method compared to prior art?
A: The patented method achieves a total recovery of 44-51% over six steps. Specifically, the four-step sequence from the ring-opening product achieves a recovery of 60-65%, which is significantly higher than previous methods that reported around 52%.
Q: How does this process improve safety regarding hazardous chemicals?
A: The process avoids the use of highly hazardous sodium hydride (NaH) often used in traditional amidation steps. Instead, it utilizes a tert-butylamine-based magnesium halide reagent, which offers a safer operational profile for industrial scaling.
Q: What solvents are used to reduce production costs?
A: The oxidative cleavage step replaces expensive tert-butanol with acetone as the primary solvent. This substitution not only lowers raw material costs but also improves the reaction yield by approximately 10% in that specific step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-tert-butyl-4-aza-5 alpha-androstane-3-ketone-17 beta-formamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of life-saving medications. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including the >99% HPLC purity required for this specific Finasteride intermediate. We are committed to delivering materials that not only meet but exceed the structural and impurity profile requirements defined in patents like CN101863954A, providing you with a secure foundation for your API synthesis.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to be your trusted partner in the complex landscape of steroid intermediate manufacturing.
