Advanced Manufacturing of N-tert-butyl-4-aza-androstane Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical industry constantly seeks robust, scalable, and cost-efficient pathways for synthesizing complex steroid intermediates, particularly those serving as precursors to blockbuster drugs like Finasteride. Patent CN101863954B introduces a refined preparation method for N-tert-butyl-4-aza-5 alpha-androstane-3-ketone-17 beta-formamide, a critical intermediate in the production of 5-alpha-reductase inhibitors. This technical disclosure outlines a comprehensive six-step synthetic strategy that begins with readily available 3 beta-hydroxy-androst-5-ene-17 beta-carboxylic acid. By integrating established reactions such as Oppenauer oxidation and oxidative cleavage with a novel amidation technique using tert-butylamine-based magnesium halide reagents, the process achieves exceptional purity levels exceeding 99% HPLC content. For R&D directors and procurement specialists, this methodology represents a significant advancement in process chemistry, offering a viable alternative to traditional routes that often suffer from lower yields or the use of hazardous reagents. The strategic optimization of reaction conditions and solvent systems underscores a commitment to both chemical efficiency and operational safety.
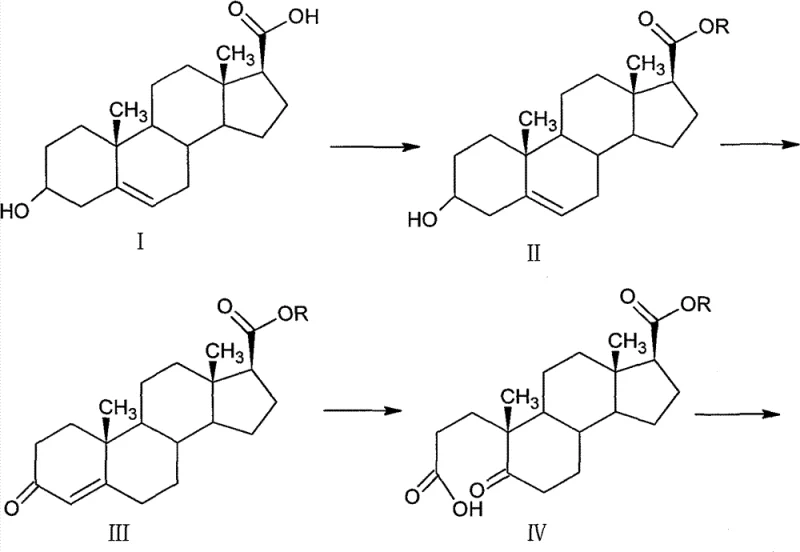
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-aza-androstane derivatives has relied on pathways that present significant challenges regarding overall yield and operational safety. Prior art, such as the method disclosed in CN101531698A, typically initiates from 3-carbonyl-4-androstene-17 beta-carboxylate methyl ester and proceeds through oxidation, ring closure, hydrogenation, and high-pressure aminolysis. While functional, these conventional routes often report a cumulative yield of approximately 52% over the critical four-step sequence, which leaves substantial room for improvement in terms of material throughput and cost efficiency. Furthermore, certain legacy processes necessitate the use of highly hazardous chemicals, such as sodium amide, which poses severe safety risks during handling and requires specialized containment infrastructure. The reliance on expensive or difficult-to-remove solvents in oxidative cleavage steps further complicates the downstream processing, leading to increased waste generation and higher purification costs. These factors collectively contribute to a less favorable economic profile for manufacturers aiming to produce high-purity pharmaceutical intermediates at a commercial scale.
The Novel Approach
In contrast, the methodology described in patent CN101863954B introduces a series of strategic modifications that effectively address the shortcomings of earlier techniques. By initiating the synthesis from 3 beta-hydroxy-androst-5-ene-17 beta-carboxylic acid, the process leverages a stable and accessible starting material to drive the reaction sequence forward with enhanced control. A key innovation lies in the substitution of tert-butanol with acetone as the solvent for the oxidative cleavage step, which not only reduces raw material costs but also reportedly improves the reaction yield by approximately 10%. Additionally, the replacement of dangerous sodium amide with a tert-butylamine-based magnesium halide reagent for the final amidation step significantly enhances operational safety while maintaining high conversion rates. The optimized route demonstrates a superior four-step total recovery rate of 60-65% when calculated from the ketone ester intermediate, surpassing the performance of prior art. This holistic improvement in yield, safety, and cost structure makes the novel approach particularly attractive for industrial applications where margin optimization and risk mitigation are paramount.
Mechanistic Insights into Oppenauer Oxidation and Grignard-Type Amidation
The core of this synthetic strategy relies on precise mechanistic control to ensure stereochemical integrity and functional group compatibility. The transformation of the 3-hydroxyl group to a ketone is achieved via Oppenauer oxidation, utilizing aluminum isopropoxide as a catalyst and cyclohexanone as the hydrogen acceptor. This reversible reaction is driven to completion by the removal of water and the use of excess cyclohexanone, ensuring high conversion to the enone intermediate (Compound III) with purity reaching 99%. Following this, the oxidative cleavage of the double bond employs a sodium periodate and potassium permanganate system in an acetone-bicarbonate aqueous medium. This specific combination allows for the selective rupture of the carbon-carbon double bond to generate the dicarboxylic acid derivative (Compound IV) without over-oxidizing sensitive functionalities elsewhere in the steroid skeleton. The subsequent cyclization with ammonium acetate in glacial acetic acid efficiently constructs the nitrogen-containing A-ring, setting the stage for the final structural elaborations.
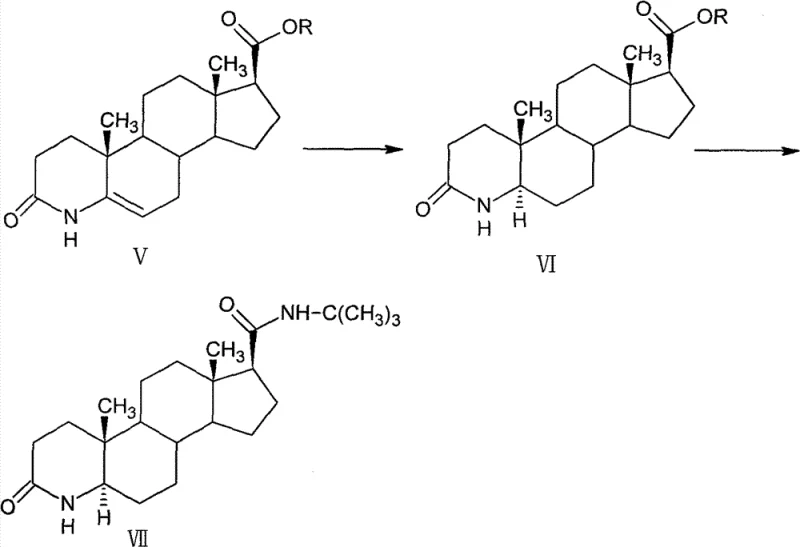
The final stages of the synthesis demonstrate sophisticated control over stereochemistry and amide bond formation. Catalytic hydrogenation using palladium on carbon is employed to reduce the double bond in the 4-aza ring system, selectively producing the thermodynamically stable 5-alpha isomer (Compound VI) with minimal formation of the unwanted 5-beta isomer (≤ 3%). The culmination of the route involves a unique amidation reaction where the C17 ester is converted directly to the tert-butyl amide. Instead of traditional aminolysis which might require harsh conditions, the process utilizes a pre-formed tert-butylamine magnesium halide reagent. This Grignard-type species acts as a potent nucleophile, attacking the ester carbonyl to displace the alkoxide and form the desired amide bond under relatively mild conditions. This mechanistic pathway avoids the degradation of the sensitive 4-aza-3-keto system, thereby preserving the high purity and yield of the final Finasteride intermediate. Such mechanistic elegance ensures that impurities are minimized at the source, reducing the burden on downstream purification units.
How to Synthesize N-tert-butyl-4-aza-androstane Efficiently
Executing this synthesis requires careful attention to reaction parameters and purification protocols to maximize the benefits outlined in the patent. The process is designed to be linear and scalable, moving from esterification to the final amidation with intermediate isolation and refinement steps that ensure quality accumulation. Operators must strictly adhere to the specified molar ratios, particularly for the oxidants and catalysts, to prevent side reactions that could compromise the steroid backbone. The use of standard laboratory equipment such as reflux condensers, autoclaves for hydrogenation, and standard filtration setups makes this route accessible for pilot plant operations. Detailed standardized operating procedures for each of the six steps, including specific temperature ranges, reaction times, and workup methods, are essential for reproducibility. For a comprehensive guide on the exact experimental conditions and stoichiometry required to replicate this high-yield process, please refer to the technical breakdown below.
- Esterification of the starting carboxylic acid using methanol or ethanol under acid catalysis to form the corresponding ester.
- Oppenauer oxidation using aluminum isopropoxide and cyclohexanone to convert the 3-hydroxyl group to a ketone.
- Oxidative cleavage of the double bond using sodium periodate and potassium permanganate in an acetone-bicarbonate system.
- Cyclization with ammonium acetate in glacial acetic acid to form the 4-aza ring structure.
- Catalytic hydrogenation using palladium on carbon to saturate the double bond and establish the 5-alpha configuration.
- Final amidation using a tert-butylamine-based magnesium halide reagent to convert the ester to the target amide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers compelling advantages for procurement managers and supply chain directors focused on cost reduction and reliability. The strategic selection of reagents and solvents directly impacts the bill of materials, driving down the overall cost of goods sold without sacrificing quality. By eliminating the need for exotic or hazardous reagents, the process simplifies logistics and reduces the regulatory burden associated with transporting and storing dangerous chemicals. Furthermore, the improved yields at each stage mean that less raw material is required to produce a kilogram of the final active intermediate, effectively amplifying the purchasing power of the manufacturing facility. These efficiencies translate into a more resilient supply chain capable of meeting fluctuating market demands with greater agility and lower overhead.
- Cost Reduction in Manufacturing: The substitution of tert-butanol with acetone in the oxidative cleavage step represents a direct material cost saving, as acetone is a ubiquitous and inexpensive commodity chemical. Moreover, the reported increase in yield for this specific step reduces the amount of starting material wasted, compounding the financial benefit. The avoidance of high-pressure aminolysis equipment and the associated energy costs further contributes to a leaner manufacturing budget. By streamlining the purification process through high-selectivity reactions, the consumption of chromatography media or excessive recrystallization solvents is minimized, leading to substantial operational expenditure reductions.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials like 3 beta-hydroxy-androst-5-ene-17 beta-carboxylic acid ensures a stable upstream supply, mitigating the risk of production stoppages due to raw material shortages. The use of robust catalysts such as aluminum isopropoxide and palladium on carbon, which are standard in the fine chemical industry, guarantees consistent availability from multiple global vendors. Additionally, the improved stability of the intermediates allows for more flexible inventory management, enabling manufacturers to stockpile key precursors without significant degradation concerns. This robustness fosters a dependable supply continuity that is critical for long-term contracts with major pharmaceutical clients.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial suitability, utilizing reaction conditions that are easily transferable from laboratory flasks to large-scale reactors. The replacement of hazardous sodium amide with safer magnesium-based reagents significantly lowers the environmental, health, and safety (EHS) footprint of the facility. Reduced solvent costs and higher yields inherently lead to less chemical waste generation per unit of product, simplifying effluent treatment and disposal compliance. This alignment with green chemistry principles not only reduces disposal costs but also enhances the corporate sustainability profile, a growing requirement for suppliers in the global pharmaceutical market.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for integration into their supply chains. The following questions address common inquiries regarding the practical implementation, quality control, and comparative benefits of this method. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Whether you are concerned about isomer purity, reaction safety, or scale-up feasibility, the information below provides a clear overview of what this technology offers.
Q: What are the key advantages of this synthesis route compared to prior art?
A: This method offers a significantly improved total yield of 60-65% for the critical four-step sequence compared to 52% in previous methods. It also replaces hazardous reagents like sodium amide with safer tert-butylamine magnesium halide reagents and utilizes cost-effective solvents like acetone instead of tert-butanol.
Q: How is high purity maintained throughout the process?
A: The process ensures high purity through specific recrystallization steps, such as refining with methanol after oxidation and hydrogenation. The final product achieves an HPLC content of greater than 99%, with strict control over isomer content (5-beta-H isomer ≤ 3%).
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the route is reasonable and particularly applicable to industrial production. The use of common solvents like toluene, acetone, and acetic acid, along with standard catalysts like palladium on carbon, facilitates easy scale-up from laboratory to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-tert-butyl-4-aza-androstane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development and production of life-saving medications. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. By leveraging advanced synthetic methodologies like the one described in CN101863954B, we can offer our partners a competitive edge through superior product consistency and reliable supply volumes.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to optimize their supply chains for Finasteride and related steroidal drugs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can support your long-term strategic goals.
