Advanced Synthetic Route for 7-MAC Intermediate: Cost-Effective Manufacturing for Cephalosporins
Advanced Synthetic Route for 7-MAC Intermediate: Cost-Effective Manufacturing for Cephalosporins
The pharmaceutical industry constantly seeks robust and economical pathways for producing critical antibiotic intermediates, particularly for the methoxy cephalosporin class which includes vital drugs like cefmetazole and cefotetan. Patent CN101696213A discloses a groundbreaking synthetic method for 7-alpha-methoxy-7-amino-3-methyl-4-thia-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid benzhydryl ester, commonly abbreviated as 7-MAC. This innovation addresses significant bottlenecks in traditional manufacturing by shifting the starting material from the scarce Cephamycin C to the widely available 7-aminocephalosporanic acid (7-ACA). Furthermore, the patent introduces critical reagent substitutions, such as replacing methyl sulfur chloride with the more reactive methyl sulfur bromide, and utilizing activated manganese dioxide for diazomethane generation instead of chloramine-T. These modifications collectively result in a process that is not only more cost-effective but also operationally simpler and higher yielding, making it highly attractive for industrial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of the 7-MAC parent nucleus has been constrained by two primary synthetic routes, both of which present substantial logistical and economic challenges for large-scale manufacturers. The first conventional method relies on Cephamycin C as the raw material; however, this starting material is notoriously difficult to source in sufficient quantities and commands a high market price due to complex fermentation requirements. The second existing method, while utilizing 7-ACA, suffers from inefficient chemical transformations, specifically during the imidization step where methyl sulfur chloride is employed. Methyl sulfur chloride possesses relatively low reactivity, often leading to incomplete reactions that necessitate rigorous purification steps and result in suboptimal yields. Additionally, the traditional generation of diphenyl diazomethane involves the use of chloramine-T, an oxidant that is not only expensive but also introduces complexities in waste treatment and operational safety. These cumulative inefficiencies create a high barrier to entry for cost-sensitive production environments.
The Novel Approach
The novel approach detailed in the patent fundamentally re-engineers the synthesis pathway to maximize efficiency and minimize input costs through strategic chemical substitutions. By anchoring the synthesis on 7-ACA, a commodity chemical with a stable and abundant global supply, the method immediately secures a lower baseline cost for raw materials. The core innovation lies in the imidization step, where methyl sulfur bromide replaces methyl sulfur chloride; the carbon-bromine bond in this reagent is weaker than the carbon-chlorine bond, acting as a superior leaving group that significantly accelerates reaction kinetics and drives the equilibrium toward the desired product. Furthermore, the protocol adopts activated manganese dioxide for the oxidation of benzophenonehydrazone to generate the necessary diazomethane species in situ. This switch eliminates the reliance on costly chloramine-T, streamlining the workflow and reducing the burden on downstream processing. This holistic optimization ensures a smoother, more predictable manufacturing process suitable for high-volume output.
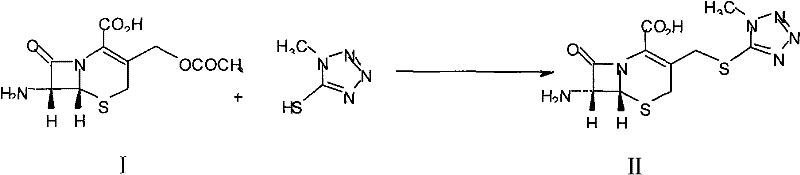
Mechanistic Insights into Methyl Sulfur Bromide Mediated Imidization
The mechanistic superiority of this synthetic route is most evident in the imidization and subsequent diazomethane coupling steps, where precise control over reactivity determines the final purity and yield of the intermediate. In the conversion of 7-TMCA to the protected intermediate, the use of methyl sulfur bromide facilitates a more nucleophilic attack on the sulfur center, effectively forming the sulfonium species required for the subsequent elimination and rearrangement. The enhanced leaving group ability of the bromide ion compared to chloride ensures that the activation energy for this step is lowered, allowing the reaction to proceed more rapidly and at potentially milder temperatures, which preserves the integrity of the sensitive beta-lactam ring. Concurrently, the protection of the carboxylic acid moiety using silylating agents such as BSA, HMDS, or TMCS prevents unwanted side reactions at the acid site, ensuring that the electrophilic attack occurs selectively at the intended position. This level of chemoselectivity is crucial for maintaining the stereochemical integrity of the cephalosporin nucleus.
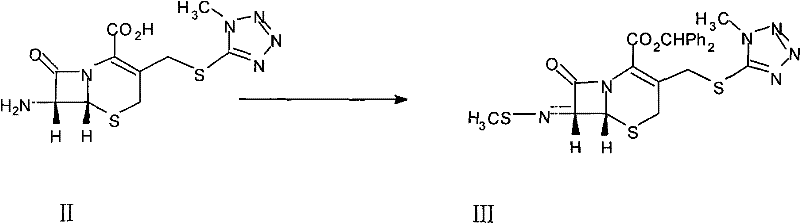
Following the imidization, the introduction of the diazo group via the manganese dioxide-mediated oxidation of benzophenonehydrazone represents a significant refinement in green chemistry principles applied to pharmaceutical synthesis. Activated manganese dioxide acts as a heterogeneous oxidant that cleanly converts the hydrazone to the diazo compound without generating the excessive saline waste associated with chloramine-T oxidation. The resulting diphenyl diazomethane then reacts with the activated sulfonium intermediate to install the necessary carbon framework. Finally, the methoxylation step utilizes an aluminum salt complex prepared from aluminum trichloride and methanol, which serves as a mild yet effective source of the methoxy nucleophile. This reagent system allows for the stereoselective introduction of the 7-alpha-methoxy group, a critical structural feature that confers beta-lactamase resistance to the final antibiotic. The entire sequence is designed to minimize impurity formation, with the patent reporting HPLC purities exceeding 98% for the final 7-MAC product.
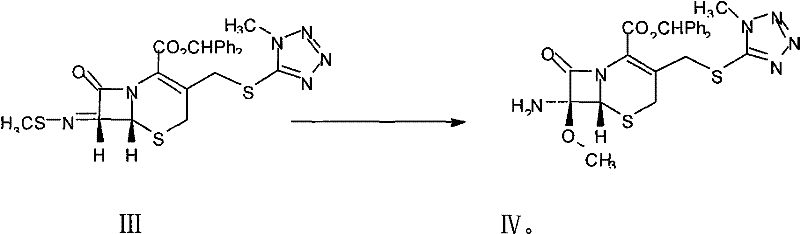
How to Synthesize 7-MAC Efficiently
The synthesis of 7-MAC via this patented route involves a carefully orchestrated three-step sequence that balances reaction kinetics with operational simplicity to achieve high purity outcomes. The process begins with the condensation of 7-ACA and MMTZ under acidic conditions to form 7-TMCA, followed by the critical imidization and diazo-coupling sequence, and concludes with the stereoselective methoxylation. Each stage requires precise temperature control, typically maintaining cryogenic conditions between 0°C and 5°C during reagent addition to prevent thermal degradation of the beta-lactam core. The detailed standardized synthesis steps, including specific molar ratios, solvent choices like acetonitrile and methylene chloride, and workup procedures involving pH adjustments and crystallization, are outlined below to guide process engineers in replicating this high-efficiency protocol.
- React 7-ACA with 1-methyl-5-mercaptotetrazole (MMTZ) using sulfuric acid catalyst to synthesize 7-TMCA.
- Perform imidization of 7-TMCA with methyl sulfur bromide, followed by reaction with diphenyl diazomethane generated from benzophenonehydrazone and activated manganese dioxide.
- Conduct methoxylation of the intermediate using an aluminum salt methoxylation reagent to obtain the final 7-MAC product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology offers tangible strategic benefits that extend beyond simple unit cost savings to encompass broader supply security and operational resilience. The shift away from Cephamycin C to 7-ACA mitigates the risk of raw material shortages, as 7-ACA is produced on a massive global scale with a mature and competitive supplier base. This transition ensures that production schedules are not held hostage by the volatility of niche fermentation products. Moreover, the replacement of expensive and hazardous reagents like chloramine-T with activated manganese dioxide simplifies the procurement of consumables and reduces the regulatory burden associated with handling strong oxidants. The overall simplification of the workflow, characterized by fewer purification hurdles and higher crude yields, translates directly into reduced manufacturing cycle times and lower utility consumption per kilogram of product.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of high-cost inputs with commoditized alternatives without sacrificing quality. By utilizing 7-ACA, which is significantly cheaper than Cephamycin C, the direct material cost is drastically lowered. Additionally, the use of methyl sulfur bromide improves reaction efficiency, meaning less raw material is wasted in unreacted streams or lost during extensive purification. The replacement of chloramine-T with activated manganese dioxide further contributes to cost savings, as manganese dioxide is a bulk chemical with a favorable price point. These cumulative changes result in a substantially reduced cost of goods sold (COGS), enhancing the margin potential for the final API.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on widely available industrial chemicals rather than specialized fermentation derivatives. 7-ACA is a standard building block in the cephalosporin value chain, ensuring that multiple qualified suppliers exist to mitigate single-source risks. The robustness of the chemical steps, particularly the high-yielding imidization using methyl sulfur bromide, reduces the likelihood of batch failures that can disrupt delivery schedules. Furthermore, the simplified operational requirements mean that the process can be easily transferred between manufacturing sites or scaled up without requiring exotic equipment or highly specialized operator training, thereby securing long-term supply stability.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is designed to be inherently greener and easier to manage at multi-ton scales. The elimination of chloramine-T reduces the generation of nitrogenous saline waste, simplifying wastewater treatment protocols and lowering disposal costs. The use of activated manganese dioxide, a solid reagent that can be filtered and potentially regenerated or disposed of safely, aligns better with modern environmental standards. The high purity of the intermediate (greater than 98% by HPLC) minimizes the need for resource-intensive recrystallization steps, reducing solvent consumption and energy usage. This makes the process not only scalable to hundreds of tons but also compliant with increasingly stringent environmental regulations governing pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 7-MAC synthesis route, derived directly from the patent specifications and process data. Understanding these nuances is essential for R&D teams evaluating the technology transfer and for procurement officers assessing the total cost of ownership. The answers provided reflect the specific advantages of the methyl sulfur bromide and manganese dioxide methodologies over legacy processes, highlighting the practical implications for industrial production.
Q: Why is 7-ACA preferred over Cephamycin C for 7-MAC synthesis?
A: 7-ACA is significantly more abundant and inexpensive compared to Cephamycin C, which is difficult to source. Using 7-ACA as the starting material drastically reduces raw material costs and ensures a stable supply chain for large-scale production.
Q: What are the advantages of using methyl sulfur bromide over methyl sulfur chloride?
A: Methyl sulfur bromide exhibits higher reactivity than methyl sulfur chloride during the imidization step. This enhancement leads to more complete reactions, higher yields, and simplified operational procedures, ultimately lowering the overall production cost.
Q: How does the new method improve safety and cost regarding diazomethane generation?
A: The process utilizes activated manganese dioxide to react with benzophenonehydrazone, replacing the traditional and expensive chloramine-T. This substitution not only reduces reagent costs but also simplifies the operation and improves the safety profile of the diazomethane generation step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-MAC Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates like 7-MAC play in the global supply of life-saving cephalosporin antibiotics. Our technical team has extensively analyzed this patented route and possesses the expertise to implement these advanced synthetic strategies at a commercial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of material. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 7-MAC meets the exacting standards required for downstream API synthesis.
We invite pharmaceutical partners to collaborate with us to leverage these cost-saving technological advancements for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can enhance your project's profitability and timeline.
