Advanced Synthesis of 2-Amino-5-chloro-N,3-dimethylbenzamide for Scalable Agrochemical Manufacturing
Introduction to Patent CN110845341B
The global demand for next-generation anthranilic diamide insecticides, specifically Chlorantraniliprole, has necessitated the development of more efficient and environmentally benign synthetic routes for its key precursors. Patent CN110845341B, published in October 2022, discloses a groundbreaking preparation method for 2-amino-5-chloro-N,3-dimethylbenzamide, a critical intermediate in this value chain. Unlike traditional methodologies that rely on harsh nitration conditions and toxic cyclization agents, this invention utilizes a strategic sequence of electrophilic chlorination and selective nucleophilic substitution starting from the abundant feedstock 2-methylaniline. The technical breakthrough lies in the precise control of regioselectivity during the chlorination and cyanation steps, leveraging thermodynamic stability differences to maximize the yield of the desired isomer while minimizing byproduct formation. This report analyzes the technical feasibility and commercial implications of this patented process for international procurement and R&D teams seeking reliable agrochemical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-amino-5-chloro-N,3-dimethylbenzamide has been plagued by significant safety and efficiency challenges inherent to nitro-chemistry pathways. The most common prior art involves the nitration of 3-methylbenzoic acid, a reaction notorious for its poor atom economy and the generation of substantial acidic waste streams. Furthermore, subsequent steps often require the use of highly toxic reagents such as phosgene or diphosgene to effect cyclization, posing severe occupational health risks and requiring specialized, capital-intensive containment infrastructure. The reduction of nitro groups typically relies on heavy metal catalysts like palladium on carbon or stoichiometric iron powder, which introduces complex purification burdens to remove trace metal impurities that can poison downstream biological assays. Additionally, the overall yield of these multi-step nitration routes is often compromised, with literature citing nitration yields as low as 45-50%, leading to inflated production costs and inconsistent supply continuity for large-scale agrochemical manufacturing.
The Novel Approach
The methodology outlined in CN110845341B represents a paradigm shift by bypassing nitration entirely in favor of a direct functionalization strategy starting from 2-methylaniline. This novel approach initiates with an oxidative chlorination using hydrochloric acid and hydrogen peroxide, a greener oxidant system that avoids the hazards associated with mixed acid nitration. The process then employs a copper-mediated cyanation to selectively replace a chlorine atom, exploiting the thermodynamic instability of the ortho-chlorine relative to the para-position to achieve high regioselectivity. This strategic routing not only simplifies the operational workflow by utilizing common, non-proprietary reagents but also drastically improves the mass balance of the synthesis. By eliminating the need for phosgene and heavy metal reductions, the new process offers a cleaner impurity profile and a more straightforward path to commercial scale-up, addressing the critical pain points of both environmental compliance and cost reduction in agrochemical intermediate manufacturing.
Mechanistic Insights into Regioselective Chlorination and Cyanation
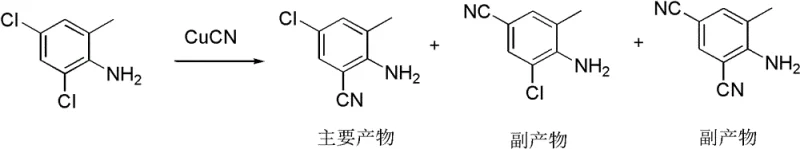
The core technical innovation of this patent resides in the meticulous control of electrophilic aromatic substitution and nucleophilic aromatic substitution mechanisms. In the initial chlorination step, the amino group of 2-methylaniline acts as a strong activating group, directing incoming chlorine species to the ortho and para positions. However, the reaction conditions—specifically the molar ratios of HCl to H2O2 and the temperature control between 10-75°C—are tuned to favor the formation of the 2,4-dichloro-6-methylaniline isomer (Formula II) over other potential polychlorinated byproducts. The patent data indicates that optimizing the hydrogen peroxide concentration to 20-45% and maintaining specific stoichiometric ratios allows for a yield of 85-90% in this critical first step, demonstrating a high degree of kinetic control that prevents over-chlorination or ring degradation.
Following chlorination, the selective substitution of the chlorine atom with a cyano group is achieved through a copper-catalyzed mechanism, likely proceeding via a radical or organometallic intermediate involving CuCN and a copper halide co-catalyst. The patent highlights a fascinating aspect of chemical reactivity where the chlorine atom at the ortho-position relative to the amino group exhibits different thermodynamic stability compared to the para-chlorine, allowing for selective displacement. This selectivity is crucial because it ensures that the cyano group is installed at the correct position (Formula III) without requiring extensive chromatographic separation of isomers. The subsequent hydrolysis of the nitrile to the carboxylic acid, followed by esterification and amidation, proceeds with high efficiency, leveraging standard organic transformations that are well-understood and easily scalable in a GMP-compliant environment, ensuring the final product meets stringent purity specifications required for pharmaceutical and agrochemical applications.
How to Synthesize 2-Amino-5-chloro-N,3-dimethylbenzamide Efficiently
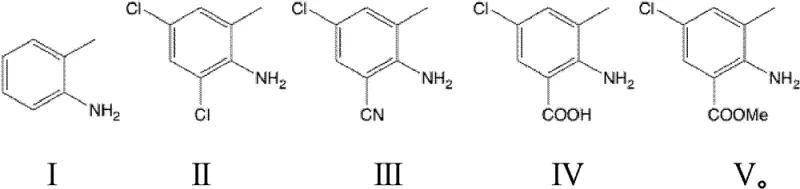
The synthesis protocol described in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing precise temperature control and stoichiometric management to maximize yield. The process begins with the careful addition of hydrochloric acid to 2-methylaniline under cooling, followed by the controlled dosing of hydrogen peroxide to manage the exothermic chlorination reaction. Subsequent steps involve heating the dichloro-intermediate with cuprous cyanide in a polar aprotic solvent like DMF, followed by a rigorous workup involving ammonia water treatment to remove copper residues. The final stages convert the nitrile functionality into the target amide through base-catalyzed hydrolysis, acid-catalyzed esterification, and finally aminolysis with monomethylamine. For detailed operational parameters, including exact reflux times, solvent volumes, and crystallization conditions, please refer to the standardized synthesis guide below.
- Perform regioselective chlorination of 2-methylaniline using HCl and H2O2 to obtain 2,4-dichloro-6-methylaniline.
- Execute selective cyanation using CuCN and copper halide to substitute the ortho-chlorine, forming 2-amino-5-chloro-3-methylbenzonitrile.
- Hydrolyze the nitrile group to carboxylic acid, followed by esterification and final amidation with monomethylamine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this synthetic route offers compelling advantages that directly address the volatility and cost pressures currently facing the fine chemical sector. By shifting the starting material from specialized nitro-compounds to commodity anilines, manufacturers can decouple their supply chains from the fluctuations of the nitration market, which is often constrained by environmental regulations on acidic waste disposal. The elimination of phosgene and diphosgene not only reduces the regulatory burden and insurance costs associated with handling Schedule 1 chemicals but also simplifies the facility requirements, allowing for production in a wider range of chemical plants without the need for ultra-high containment systems. This flexibility enhances supply chain resilience, ensuring that production schedules are not disrupted by regulatory audits or safety incidents related to toxic reagent storage.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the significant improvement in overall yield and the reduction of waste treatment costs. By achieving a total yield of 58-68% through a shorter, more direct pathway, the consumption of raw materials per kilogram of final product is substantially lowered compared to the 31-41% yields typical of older nitration-based routes. Furthermore, the replacement of expensive noble metal catalysts (like Palladium) and toxic reagents with inexpensive copper salts and hydrogen peroxide results in a drastic reduction in direct material costs. The simplified purification steps, relying largely on precipitation and recrystallization rather than complex distillation or column chromatography, further decrease energy consumption and labor hours, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: The reliance on 2-methylaniline as the primary feedstock provides a significant logistical advantage, as this chemical is produced on a massive scale globally for various dye and polymer applications, ensuring a stable and competitive supply. Unlike niche nitro-intermediates that may have limited suppliers and long lead times, 2-methylaniline is readily available from multiple sources, mitigating the risk of single-supplier dependency. The robustness of the reaction conditions, which tolerate a range of temperatures and concentrations without catastrophic failure, also implies a higher success rate in batch production, reducing the frequency of failed batches that can disrupt delivery timelines and strain inventory buffers for downstream agrochemical formulators.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing unit operations such as filtration, phase separation, and reflux that are easily translated from liter-scale reactors to multi-ton vessels. The absence of heavy metal waste streams and the use of aqueous workups significantly lower the E-factor (mass of waste per mass of product), aligning with modern green chemistry principles and corporate sustainability goals. This environmental compatibility facilitates smoother permitting processes for capacity expansion and reduces the long-term liability associated with hazardous waste disposal, making it an attractive option for companies aiming to future-proof their manufacturing assets against tightening global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived from the specific advantages and operational details disclosed in the patent documentation. These insights are intended to clarify the feasibility of adopting this route for commercial production and to highlight the quality standards achievable through this methodology. Understanding these nuances is essential for technical directors and procurement specialists evaluating the transition from legacy processes to this improved manufacturing platform.
Q: How does this new process improve safety compared to traditional nitration routes?
A: The novel route eliminates the use of hazardous nitrating agents and toxic phosgene/diphosgene reagents required in conventional methods, significantly reducing environmental impact and operator risk.
Q: What is the expected purity profile for the final agrochemical intermediate?
A: Through optimized recrystallization and selective substitution steps, the process consistently achieves purity levels exceeding 99%, minimizing downstream purification costs.
Q: Is the starting material 2-methylaniline readily available for large-scale production?
A: Yes, 2-methylaniline is a commodity chemical with a robust global supply chain, ensuring stable raw material availability and preventing bottlenecks in commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-5-chloro-N,3-dimethylbenzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven execution capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN110845341B are fully realized in practical, large-volume manufacturing. Our state-of-the-art facilities are equipped to handle the specific thermal and catalytic requirements of this chlorination-cyanation sequence, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 2-amino-5-chloro-N,3-dimethylbenzamide meets the exacting standards required for high-performance agrochemical formulations.
We invite forward-thinking organizations to collaborate with us to leverage this cost-effective and environmentally superior technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, comparing the total landed cost of this new route against your current sourcing model. We encourage you to contact us today to obtain specific COA data from our recent pilot runs and to discuss route feasibility assessments that can accelerate your time-to-market for next-generation crop protection solutions.
