Revolutionizing Chlorantraniliprole Intermediate Production with Green Iron Catalysis and Commercial Scalability
Revolutionizing Chlorantraniliprole Intermediate Production with Green Iron Catalysis and Commercial Scalability
The global demand for high-efficiency, low-toxicity insecticides like chlorantraniliprole has necessitated a paradigm shift in the manufacturing of its critical precursors. Patent CN115772090A introduces a groundbreaking synthesis method for 2-amino-5-chloro-N,3-dimethylbenzamide, a pivotal intermediate in the agrochemical sector. This technology leverages a novel supported nano iron catalyst system combined with tert-butyl hydroperoxide (TBHP) to facilitate a direct amidation reaction between 2-amino-3-methyl-5-chlorobenzoic acid and methylamine aqueous solution. By operating under mild conditions of normal temperature and pressure, specifically around 40°C, this process circumvents the severe limitations of traditional dehydration-based amidation which often fails in the presence of water. The strategic implementation of this green catalytic pathway not only addresses the urgent industry need for cleaner production but also establishes a robust framework for cost reduction in agrochemical manufacturing by eliminating hazardous reagents and simplifying downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzamide derivatives such as 2-amino-5-chloro-N,3-dimethylbenzamide has been plagued by significant chemical and environmental inefficiencies. Conventional routes typically rely on the activation of the carboxylic acid group through conversion to an acyl chloride using aggressive chlorinating agents like phosphorus trichloride, phosphorus pentachloride, or thionyl chloride. These reagents are not only hazardous to handle on a large scale but inevitably generate substantial quantities of waste acid, creating a heavy burden on environmental compliance and waste treatment infrastructure. Alternatively, esterification followed by ammonolysis has been employed, yet this multi-step approach suffers from low atom economy, high energy consumption due to extended reaction times and heating requirements, and complicated operational procedures that increase the risk of yield loss. Furthermore, direct dehydration methods using carbodiimides are often incompatible with aqueous methylamine solutions, leading to poor reaction selectivity and the formation of difficult-to-remove byproducts that compromise the final purity of the agrochemical intermediate.
The Novel Approach
In stark contrast to these legacy methodologies, the technology disclosed in CN115772090A presents a streamlined, single-pot solution that directly couples the carboxylic acid and amine substrates. By utilizing a specialized supported nano iron catalyst in conjunction with TBHP as a radical initiator, the process successfully activates the benzoic acid derivative for nucleophilic attack by methylamine without the need for pre-activation into acid chlorides or esters. This radical-mediated amidation proceeds efficiently in benign solvents such as water or ethanol, tolerating the aqueous nature of the methylamine feedstock which was previously a major hurdle. The reaction achieves high conversion rates within a short timeframe of 2 to 8 hours at moderate temperatures, demonstrating exceptional selectivity with virtually no byproduct generation. This fundamental shift in synthetic strategy transforms a complex, waste-intensive operation into a green, atom-economical process that is inherently safer and more suitable for continuous industrial production.
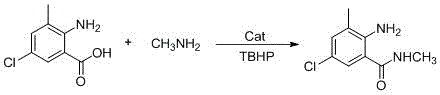
Mechanistic Insights into Supported Nano Iron-Catalyzed Radical Amidation
The core innovation of this synthesis lies in the unique electronic and structural properties of the supported nano iron catalyst, which facilitates a radical chain mechanism distinct from classical ionic amidation pathways. The catalyst, synthesized via the coordination of 2-(2-hydroxymethyl)pyridine ligands with ferric species on an alumina support, acts as a potent redox mediator. Upon interaction with TBHP, the iron centers undergo reversible oxidation state changes that generate tert-butoxyl and hydroxyl radicals. These highly reactive species abstract hydrogen atoms or activate the carboxyl group, creating a transient acyl radical or a highly electrophilic intermediate that is susceptible to nucleophilic attack by the amine. This radical propagation cycle ensures that the reaction proceeds rapidly even at lower temperatures, bypassing the high activation energy barriers associated with thermal dehydration. The heterogeneous nature of the supported catalyst further enhances the reaction kinetics by providing a high surface area for substrate adsorption, thereby maximizing the collision frequency between the reactants and the active catalytic sites.
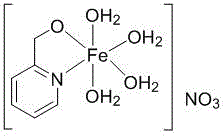
Beyond mere acceleration, the specific design of the catalyst plays a critical role in impurity control and product quality assurance. The steric environment created by the pyridine-based ligands on the iron surface imposes a degree of regioselectivity, ensuring that the amidation occurs exclusively at the carboxyl group without affecting the sensitive amino or chloro substituents on the aromatic ring. This chemoselectivity is paramount for pharmaceutical and agrochemical applications where trace impurities can lead to regulatory rejection or reduced biological efficacy. Moreover, the stability of the iron-ligand complex prevents metal leaching into the reaction medium, which simplifies the purification process and ensures that the final product meets stringent heavy metal specifications. The ability to recover and regenerate the catalyst using simple methanol washing protocols without significant loss of activity underscores the robustness of this mechanistic design, allowing for consistent batch-to-batch reproducibility essential for commercial scale-up of complex agrochemical intermediates.
How to Synthesize 2-amino-5-chloro-N,3-dimethylbenzamide Efficiently
The practical implementation of this synthesis route requires precise control over reaction parameters to maximize yield and purity while maintaining operational safety. The process begins with the preparation of the active catalyst, followed by the careful dosing of reactants in a controlled atmosphere to prevent premature decomposition of the peroxide additive. Operators must maintain the reaction temperature within the optimal window of 40°C to balance reaction rate and selectivity, avoiding thermal runaways that could degrade the product. Following the reaction period, the separation of the heterogeneous catalyst allows for immediate recycling, while the crude product is isolated through a straightforward alkalization and filtration sequence. For a comprehensive understanding of the specific molar ratios, solvent volumes, and step-by-step operational protocols required to replicate these high-yield results, please refer to the detailed standardized synthesis guide provided below.
- Prepare the supported nano iron catalyst by reacting 2-pyridinecarboxaldehyde with sodium hydroxide and alumina, followed by impregnation with ferric nitrate.
- Combine 2-amino-3-methyl-5-chlorobenzoic acid, methylamine aqueous solution, the supported nano iron catalyst, and TBHP in ethanol or water solvent.
- Heat the mixture to 40°C for 6-12 hours, recover the catalyst, adjust pH to 6.0-6.5 with sodium bicarbonate, and isolate the product via filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed technology represents a strategic opportunity to optimize the total cost of ownership for chlorantraniliprole intermediates. By transitioning away from corrosive chlorinating agents and multi-step esterification processes, manufacturers can drastically simplify their supply chains, reducing reliance on volatile raw material markets for reagents like thionyl chloride. The elimination of hazardous waste streams translates directly into lower disposal costs and reduced regulatory overhead, enhancing the overall economic viability of the production line. Furthermore, the compatibility of the process with aqueous methylamine solutions removes the need for expensive anhydrous reagents, leveraging cheaper and more readily available bulk chemicals to drive down input costs significantly.
- Cost Reduction in Manufacturing: The most profound economic benefit stems from the drastic simplification of the synthetic route, which eliminates the need for expensive activating agents and the associated waste treatment infrastructure. By removing the acyl chlorination step, facilities avoid the capital and operational expenditures related to handling corrosive gases and neutralizing large volumes of acidic waste. Additionally, the high atom economy of the direct amidation ensures that a greater proportion of the raw material mass is converted into the final valuable product, minimizing material loss. The reusability of the nano iron catalyst for multiple cycles further amortizes the cost of the catalytic system, contributing to substantial long-term savings in catalyst expenditure compared to single-use homogeneous catalysts.
- Enhanced Supply Chain Reliability: The reliance on stable, commodity-grade raw materials such as benzoic acid derivatives and aqueous amines significantly de-risks the supply chain against disruptions common with specialty reagents. Traditional methods often depend on just-in-time delivery of hazardous chlorinating agents which are subject to strict transportation regulations and potential supply bottlenecks. In contrast, the feedstocks for this green process are widely available from multiple global suppliers, ensuring continuity of supply even during market fluctuations. The robustness of the catalyst, which maintains activity over repeated uses, also reduces the frequency of catalyst replenishment orders, simplifying inventory management and reducing the administrative burden on the procurement team.
- Scalability and Environmental Compliance: From a sustainability perspective, this process aligns perfectly with modern green chemistry principles, making it easier to secure environmental permits and maintain social license to operate. The absence of halogenated waste and the use of benign solvents like water or ethanol minimize the environmental footprint, facilitating smoother regulatory approvals for capacity expansion. The simplicity of the workup procedure, involving basic filtration and drying rather than complex distillations or extractions, makes the process highly scalable from pilot plant to multi-ton commercial production. This scalability ensures that suppliers can rapidly respond to surges in market demand for chlorantraniliprole without compromising on product quality or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing manufacturing facilities.
Q: What are the primary advantages of this iron-catalyzed method over traditional acyl chloride routes?
A: Unlike traditional methods requiring phosphorus trichloride or thionyl chloride which generate hazardous waste acids, this novel process utilizes a supported nano iron catalyst and TBHP to enable direct amidation. This eliminates the need for harsh chlorinating agents, significantly reducing environmental impact and waste treatment costs while maintaining high atom economy.
Q: Can the catalyst be reused effectively in industrial scale production?
A: Yes, the patent data indicates that the supported nano iron catalyst can be recovered, washed with anhydrous methanol, and dried for reuse. Experimental results demonstrate that the catalytic activity does not significantly decrease even after being repeatedly used for five cycles, ensuring consistent process efficiency and reduced raw material consumption.
Q: What purity levels can be achieved with this synthesis route?
A: The optimized process yields 2-amino-5-chloro-N,3-dimethylbenzamide with exceptional purity. Specific examples in the patent report HPLC detection results showing purity levels reaching 99.6%, with reaction yields consistently exceeding 93% under optimized solvent and concentration conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-amino-5-chloro-N,3-dimethylbenzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed synthesis route for the global agrochemical industry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-amino-5-chloro-N,3-dimethylbenzamide delivered meets the highest international standards for insecticide manufacturing. We are committed to leveraging our technical expertise to optimize this green process, delivering a reliable agrochemical intermediate supplier experience that combines quality with sustainability.
We invite forward-thinking partners to collaborate with us to unlock the full value of this technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can enhance your competitive position in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
