Advanced Racemization Recovery for Sitafloxacin Intermediates: Scaling High-Purity Production
Advanced Racemization Recovery for Sitafloxacin Intermediates: Scaling High-Purity Production
The pharmaceutical industry constantly seeks methods to enhance the atom economy and sustainability of active pharmaceutical ingredient (API) synthesis, particularly for complex chiral molecules like sitafloxacin. Patent CN108911972B, published in July 2020, presents a groundbreaking technical solution for the racemization recovery of by-products found in the resolution mother liquor of sitafloxacin intermediates. This innovation specifically targets the efficient recycling of unwanted trans-isomers, namely (1R,2R)-2-fluorocyclopropanecarboxylic acid and (1S,2R)-2-fluorocyclopropanecarboxylic acid, which are typically discarded as waste in conventional production lines. By converting these low-value by-products back into the valuable racemic starting material, the technology offers a robust pathway to improve overall process yield and reduce the environmental footprint of fluoroquinolone antibiotic manufacturing. For R&D directors and process chemists, this represents a critical advancement in impurity management and resource utilization, transforming a linear waste-generating process into a circular, economically superior manufacturing loop.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (1S,2S)-2-fluorocyclopropanecarboxylic acid has been plagued by inefficient routes that generate significant hazardous waste and rely on dangerous reagents. As illustrated in early literature from 1984, classical pathways often involved the addition of dibromofluorocarbene to butadiene, followed by a series of oxidation and esterification steps that culminated in a debromination reaction requiring tributyltin hydride (Bu3SnH). 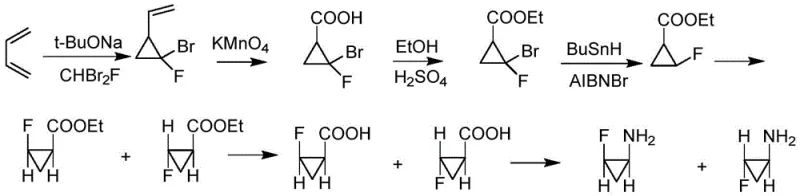 This reliance on organotin compounds poses severe challenges for industrial hygiene and environmental compliance, as removing trace tin residues from the final API is both technically difficult and costly. Furthermore, alternative biological approaches, such as the microbial resolution reported in 1998, while achieving high enantiomeric purity, have failed to gain traction in large-scale manufacturing due to the inherent difficulties in scaling fermentation processes and maintaining consistent sterility. More recent rhodium-catalyzed methods, although stereoselective, still suffer from the fundamental economic flaw of generating substantial quantities of unusable trans-isomer by-products that are difficult to convert, leading to a theoretical maximum yield cap that inflates the cost of goods sold (COGS).
This reliance on organotin compounds poses severe challenges for industrial hygiene and environmental compliance, as removing trace tin residues from the final API is both technically difficult and costly. Furthermore, alternative biological approaches, such as the microbial resolution reported in 1998, while achieving high enantiomeric purity, have failed to gain traction in large-scale manufacturing due to the inherent difficulties in scaling fermentation processes and maintaining consistent sterility. More recent rhodium-catalyzed methods, although stereoselective, still suffer from the fundamental economic flaw of generating substantial quantities of unusable trans-isomer by-products that are difficult to convert, leading to a theoretical maximum yield cap that inflates the cost of goods sold (COGS).
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN108911972B introduces a sophisticated chemical recycling loop that effectively valorizes waste streams. The core innovation lies in the ability to take the specific trans-by-products ((1R,2R) and (1S,2R) isomers) isolated from the resolution mother liquor and chemically revert them to the racemic acid form. 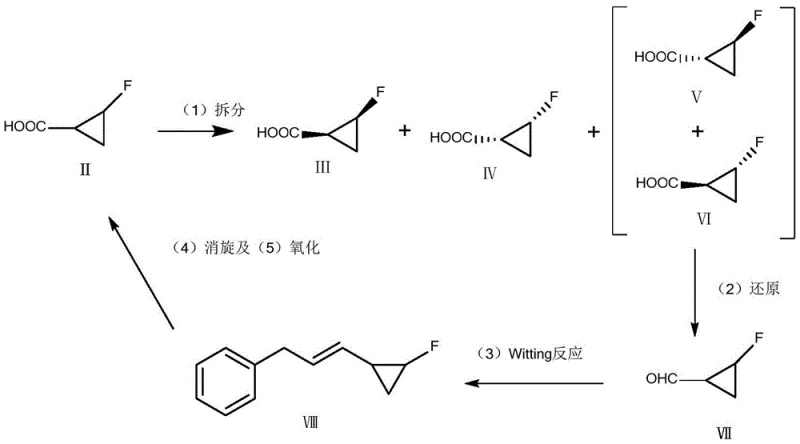 This is achieved through a meticulously designed sequence involving reduction to an aldehyde, followed by a Wittig olefination to create an allyl benzene derivative, which serves as the substrate for a radical-mediated racemization. This approach bypasses the need for toxic tin reagents entirely and avoids the scalability bottlenecks of enzymatic processes. By reintroducing the recycled racemic acid back into the initial resolution step, the process effectively breaks the yield ceiling imposed by traditional 50% theoretical limits of resolution, ensuring that nearly all fluorinated carbon atoms introduced into the process eventually end up in the desired API intermediate, thereby drastically improving the mass balance of the entire production campaign.
This is achieved through a meticulously designed sequence involving reduction to an aldehyde, followed by a Wittig olefination to create an allyl benzene derivative, which serves as the substrate for a radical-mediated racemization. This approach bypasses the need for toxic tin reagents entirely and avoids the scalability bottlenecks of enzymatic processes. By reintroducing the recycled racemic acid back into the initial resolution step, the process effectively breaks the yield ceiling imposed by traditional 50% theoretical limits of resolution, ensuring that nearly all fluorinated carbon atoms introduced into the process eventually end up in the desired API intermediate, thereby drastically improving the mass balance of the entire production campaign.
Mechanistic Insights into Radical-Mediated Racemization and Wittig Olefination
The chemical elegance of this process is rooted in the strategic manipulation of the cyclopropane ring's stereochemistry through a temporary structural modification. The mechanism begins with the selective reduction of the carboxylic acid by-products using lithium tri-tert-butoxyaluminum hydride at cryogenic temperatures (down to -90°C), which converts the acid functionality into a trans-2-fluorocyclopropane formaldehyde without disrupting the sensitive fluorinated cyclopropane ring. This aldehyde is then subjected to a Wittig reaction using a phosphorus ylide generated in situ from 2-bromoethylbenzene and phosphorus oxychloride. This step extends the carbon chain to form (3-(2-fluorocyclopropyl)allyl)benzene, a crucial intermediate that possesses an allylic system susceptible to radical attack. The subsequent racemization step utilizes a free radical initiator, such as azobisisobutyronitrile (AIBN), in the presence of liquid bromine. This generates radical species that abstract the allylic hydrogen, creating a planar radical intermediate that loses its stereochemical memory. Upon recombination or subsequent reaction, the stereochemistry at the cyclopropane ring is scrambled, effectively converting the pure trans-isomers back into a mixture containing cis-isomers upon re-oxidation.
From an impurity control perspective, this mechanism is exceptionally clean because it avoids the formation of heavy metal complexes or persistent organic pollutants. The oxidation step, which closes the loop by converting the racemized allyl benzene back to 2-fluorocyclopropanecarboxylic acid, can be performed using common oxidants like potassium permanganate or hydrogen peroxide. The patent data indicates that the relative content of the four isomers after oxidation and resolution can be tightly controlled, with the desired (1S,2S) isomer being efficiently separated. The use of mild conditions throughout the reduction and alkylation steps ensures that the fluorine atom, which is critical for the biological activity of the final sitafloxacin molecule, remains intact and does not undergo defluorination, a common side reaction in harsh acidic or basic environments. This preservation of the C-F bond integrity is paramount for maintaining the potency and safety profile of the final antibiotic product.
How to Synthesize (1S,2S)-2-fluorocyclopropanecarboxylic acid Efficiently
The implementation of this recycling technology requires precise control over reaction parameters, particularly temperature and stoichiometry, to ensure high fidelity in the stereochemical inversion. The process begins with the resolution of the racemic acid using chiral bases like strychnine or ephedrine, where the mother liquor containing the unwanted isomers is carefully preserved rather than discarded. This standardized workflow allows manufacturing teams to integrate the recycling loop seamlessly into existing production lines without requiring entirely new reactor configurations, facilitating a smooth transition from batch to continuous processing modes if desired.
- Resolution of racemic 2-fluorocyclopropane carboxylic acid to isolate target isomers and collect by-product rich mother liquor.
- Reduction of recovered trans-by-products to trans-2-fluorocyclopropane formaldehyde using lithium tri-tert-butoxyaluminum hydride.
- Conversion of the aldehyde to (3-(2-fluorocyclopropyl)allyl)benzene via Wittig reaction, followed by radical racemization and oxidation to regenerate racemic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this racemization recovery technology translates directly into enhanced operational resilience and significant cost optimization opportunities. By implementing a closed-loop system that recycles internal waste streams, manufacturers can drastically reduce their dependency on external raw material suppliers for the fluorinated cyclopropane building blocks. This internal sourcing capability acts as a buffer against market volatility and supply disruptions, ensuring a more stable and predictable production schedule for sitafloxacin and related fluoroquinolones. Furthermore, the elimination of tributyltin hydride removes a major cost center associated with hazardous waste disposal and specialized scrubbing systems, simplifying the environmental compliance burden and reducing the overall overhead costs of the manufacturing facility.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the conversion of low-value waste into high-value product, which inherently lowers the effective cost per kilogram of the final API intermediate. By recovering the trans-isomers that would otherwise be incinerated or sold at a fraction of the cost, the process maximizes the utility of every mole of expensive fluorinated reagent purchased. Additionally, the patent highlights that the solvents used in the process, such as toluene and THF, can be recovered and reused, further compounding the savings on consumable materials. The avoidance of precious metal catalysts like rhodium also eliminates the need for complex metal scavenging steps, streamlining the downstream purification process and reducing the consumption of expensive chromatography media.
- Enhanced Supply Chain Reliability: Integrating this recycling method diversifies the supply risk profile by reducing the total volume of fresh starting materials required for a given production target. In a global supply chain prone to logistical delays, the ability to stretch existing inventory through chemical recycling provides a critical safety margin. The process relies on commodity chemicals like oxalyl chloride, pyridine, and common oxidants, which are widely available from multiple global suppliers, unlike specialized chiral enzymes or exotic organometallic catalysts that may have single-source supply risks. This commoditization of reagents ensures that production continuity is maintained even if specific niche suppliers face shortages.
- Scalability and Environmental Compliance: The technical simplicity of the reaction steps—primarily standard organic transformations like reduction, Wittig olefination, and oxidation—makes this process highly scalable from pilot plant to multi-ton commercial production. Unlike fermentation processes that require strict sterile conditions and complex bioreactors, this chemical approach can be executed in standard glass-lined or stainless steel reactors found in most fine chemical plants. The reduction in hazardous waste generation, specifically the absence of tin sludge, aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing site against tighter emission standards and reducing the long-term liability associated with hazardous waste storage and treatment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this racemization recovery technology, based on the specific data and embodiments provided in the patent documentation. These insights are derived directly from the experimental examples and comparative data presented in the intellectual property, ensuring that the answers reflect the actual capabilities and limitations of the described chemistry.
Q: How does this patent address the waste issue in sitafloxacin synthesis?
A: The patent introduces a closed-loop recycling system where unwanted trans-isomers ((1R,2R) and (1S,2R)) from the resolution mother liquor are chemically converted back into racemic starting material, significantly reducing material loss.
Q: What are the safety advantages over classical synthesis methods?
A: Unlike classical methods that rely on highly toxic and expensive tributyltin hydride (Bu3SnH) for debromination, this process utilizes milder reagents like lithium tri-tert-butoxyaluminum hydride and avoids heavy metal contamination.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly highlights mild reaction conditions, simple operations, and the ability to recover and reuse solvents, making it highly viable for commercial scale-up compared to difficult-to-scale microbial fermentation methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitafloxacin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced antibiotics like sitafloxacin depends on robust, scalable, and compliant manufacturing processes. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the one described in CN108911972B are translated efficiently from the laboratory to the factory floor. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of (1S,2S)-2-fluorocyclopropanecarboxylic acid meets the exacting standards required for global pharmaceutical registration. We understand the critical nature of chiral purity in fluoroquinolone synthesis and have the analytical expertise to monitor and control isomer ratios throughout the recycling loop.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this innovative recycling technology for their sitafloxacin supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of implementing this waste-recovery strategy in your specific production context. We encourage you to contact us today to discuss your project requirements,索取 specific COA data for our intermediates, and review our detailed route feasibility assessments. Let us help you optimize your manufacturing economics while contributing to a more sustainable pharmaceutical industry through advanced chemical engineering solutions.
