Advanced Green Synthesis of Roflumilast Intermediates: Scaling High-Purity API Production
Advanced Green Synthesis of Roflumilast Intermediates: Scaling High-Purity API Production
The pharmaceutical landscape for respiratory therapeutics is constantly evolving, driven by the need for safer, more efficient manufacturing processes for critical active pharmaceutical ingredients (APIs). A pivotal advancement in this domain is detailed in patent CN102336703B, which outlines a novel, environmentally benign method for synthesizing Roflumilast, a selective phosphodiesterase 4 (PDE4) inhibitor used in the treatment of Chronic Obstructive Pulmonary Disease (COPD). This technology represents a significant departure from legacy synthetic routes, addressing long-standing issues regarding pollution, step count, and overall yield efficiency. By leveraging a bismuth-catalyzed oxidation system followed by a high-efficiency amide coupling strategy, this process offers a robust pathway for producing high-purity pharmaceutical intermediates. For global procurement teams and R&D directors, understanding the nuances of this patent is essential for securing a reliable Roflumilast intermediate supplier capable of meeting stringent regulatory and volume demands.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Roflumilast has been plagued by inefficiencies inherent in multi-step halogenation strategies. Traditional methods, such as those described in earlier patents like WO9501338A1, typically rely on converting the key benzaldehyde precursor into an acid chloride intermediate using thionyl chloride (SOCl2). This approach is fundamentally flawed from both an environmental and economic perspective. The generation of acid chloride releases noxious sulfur dioxide gas and hydrochloric acid fumes, necessitating expensive scrubbing systems and creating significant hazardous waste streams that complicate disposal. Furthermore, the subsequent condensation reaction with 4-amino-3,5-dichloropyridine often requires a substantial excess of the amine reactant—typically 2.0 to 2.2 equivalents—to drive the reaction to completion. Despite this wasteful stoichiometry, the yields remain suboptimal, often hovering around 58%, which drastically inflates the cost of goods sold (COGS) and limits the throughput of manufacturing facilities.
The Novel Approach
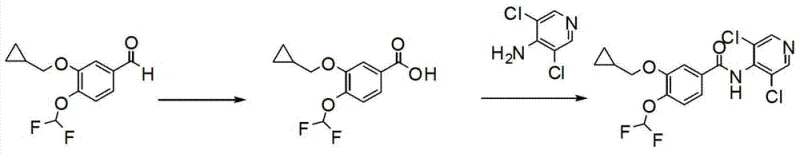
In stark contrast, the methodology disclosed in CN102336703B introduces a streamlined, green chemistry approach that bypasses the problematic acid chloride intermediate entirely. Instead of chlorination, the process employs a direct catalytic oxidation of 3-(cyclopropylmethoxy)-4-(difluoromethoxy)benzaldehyde to its corresponding benzoic acid using a bismuth oxide (Bi2O3) and tert-butyl hydroperoxide (t-BuOOH) system. This oxidation step is remarkably clean, operating under reflux conditions in benign solvents like ethyl acetate, and achieves exceptional conversion rates. The resulting carboxylic acid is then directly coupled with the pyridine amine using modern peptide coupling reagents. This strategic shift not only eliminates the release of toxic gases but also shortens the overall synthetic sequence, thereby reducing the cumulative loss of material at each stage and significantly enhancing the overall process mass intensity (PMI).
Mechanistic Insights into Bi2O3-Catalyzed Oxidation and HATU Coupling
The core innovation of this synthetic route lies in the mechanistic elegance of the bismuth-catalyzed oxidation. Bismuth compounds are increasingly recognized in fine chemical manufacturing for their low toxicity and high Lewis acidity, which facilitates the activation of peroxides. In this specific transformation, Bi2O3 acts as a heterogeneous or semi-heterogeneous catalyst that activates the 70% t-BuOOH oxidant. The mechanism likely involves the coordination of the aldehyde oxygen to the bismuth center, increasing the electrophilicity of the carbonyl carbon and facilitating nucleophilic attack by the hydroperoxide species. This pathway allows for the smooth conversion of the aldehyde to the carboxylic acid under relatively mild thermal conditions (solvent reflux), avoiding the harsh acidic environments that often lead to the degradation of sensitive functional groups like the difluoromethoxy ether. The patent specifies a catalyst loading of merely 0.05 to 0.2 equivalents, demonstrating high turnover efficiency and minimizing heavy metal residue in the final product.
Following oxidation, the condensation step utilizes HATU (2-(7-azabenzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate) as the coupling agent. Unlike traditional carbodiimides which can form stable urea byproducts that are difficult to remove, HATU generates highly reactive O-acylisourea intermediates that rapidly convert to the desired amide bond in the presence of the amine nucleophile. The addition of N-methylmorpholine (NMM) as a base ensures the deprotonation of the amine and neutralizes the generated acid, driving the equilibrium forward. The patent highlights a specific molar ratio of HATU to NMM of 1:2, which is critical for maximizing yield while minimizing side reactions such as racemization or over-activation. This precise control over reaction stoichiometry results in a crude product of high purity, simplifying the final recrystallization step and ensuring the API meets rigorous pharmacopeial standards.
How to Synthesize Roflumilast Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The process begins with the dissolution of the aldehyde starting material in ethyl acetate, followed by the sequential addition of the bismuth catalyst and the oxidant. Careful temperature control during the reflux period is essential to maintain the reaction rate without decomposing the peroxide. Upon completion, the workup involves a simple aqueous extraction and acidification to isolate the benzoic acid intermediate. The second stage involves dissolving the acid, the amine, and the base in a polar aprotic solvent like DMF, followed by the controlled addition of HATU.
- Oxidize 3-(cyclopropylmethoxy)-4-(difluoromethoxy)benzaldehyde using Bi2O3 catalyst and 70% t-BuOOH in ethyl acetate under reflux to obtain the benzoic acid intermediate with 95% yield.
- Activate the resulting benzoic acid using HATU coupling reagent and NMM base in DMF solvent.
- Condense the activated acid with 4-amino-3,5-dichloropyridine to form the final Roflumilast API, followed by recrystallization from ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible operational improvements beyond mere chemical yield. The elimination of the acid chloride step removes a major bottleneck associated with corrosion management and hazardous gas scrubbing, allowing for the use of standard glass-lined or stainless steel reactors without specialized linings. This simplification of the equipment requirements directly lowers capital expenditure (CAPEX) barriers for contract manufacturing organizations (CMOs) and reduces maintenance downtime. Furthermore, the use of widely available, commodity-grade solvents such as ethyl acetate and DMF ensures that raw material sourcing remains stable and cost-effective, insulating the supply chain from volatility associated with specialized reagents.
- Cost Reduction in Manufacturing: The most significant financial advantage stems from the drastic reduction in raw material waste. By achieving a condensation yield of approximately 75% compared to the historical 58%, the process consumes significantly less of the expensive dichloropyridine amine per kilogram of output. Additionally, the removal of the thionyl chloride step eliminates the costs associated with purchasing this hazardous reagent and treating the resulting acidic waste streams. The lower catalyst loading of bismuth oxide further contributes to cost efficiency, as the metal can potentially be recovered or disposed of at a lower cost than precious metal catalysts.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply continuity by reducing the risk of batch failures. The mild reaction conditions minimize the formation of complex, hard-to-remove impurities that often plague scale-up campaigns. This reliability means that production schedules are less likely to be disrupted by the need for re-processing or extensive purification cycles. Moreover, the starting materials, including the fluorinated benzaldehyde and the chloropyridine, are established commodities in the fine chemical sector, ensuring a steady flow of inputs even during periods of market tightness.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this process is vastly superior. The absence of sulfur dioxide emissions simplifies regulatory compliance and reduces the environmental footprint of the manufacturing site. The process is inherently safer to operate at scale, as it avoids the exothermic risks associated with large-scale acid chloride formations. This safety profile facilitates faster regulatory approvals for new manufacturing sites and supports the long-term sustainability goals of modern pharmaceutical companies seeking green chemistry partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Roflumilast synthesis route. These insights are derived directly from the experimental data and claims presented in patent CN102336703B, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer or capacity expansion.
Q: How does the Bi2O3 catalytic system improve impurity profiles compared to traditional chlorination methods?
A: The Bi2O3/t-BuOOH system operates under mild conditions without generating corrosive acidic byproducts like SO2 or HCl, which are common in thionyl chloride oxidations. This results in a cleaner reaction mixture, significantly reducing the burden on downstream purification and ensuring higher purity specifications for the final API.
Q: What are the cost implications of replacing the acid chloride intermediate with direct acid coupling?
A: Eliminating the acid chloride formation step removes the need for hazardous chlorinating agents and the associated waste treatment costs. Furthermore, the direct coupling using HATU achieves higher conversion rates (approx. 75% vs 58% in older methods), reducing raw material consumption per kilogram of finished product.
Q: Is this synthetic route scalable for commercial metric ton production?
A: Yes, the process utilizes common industrial solvents like ethyl acetate and DMF and avoids cryogenic conditions. The use of stable solid catalysts (Bi2O3) and liquid oxidants facilitates safe handling and precise dosing in large-scale reactors, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roflumilast Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, compliant, and scalable manufacturing pathways for respiratory APIs like Roflumilast. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to monitor impurity profiles at every stage of the synthesis. Our facility is equipped to handle the specific solvent systems and coupling reagents required by this green chemistry route, guaranteeing a consistent supply of high-quality intermediates.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced technology for your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this optimized route can reduce your overall COGS. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply chain security.
