Advanced Manufacturing of Azelastine Hydrochloride via Stable Solid Intermediates
Advanced Manufacturing of Azelastine Hydrochloride via Stable Solid Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways that guarantee both high purity and operational stability, particularly for antihistamine agents like Azelastine Hydrochloride. Patent CN113045547B discloses a groundbreaking preparation method that fundamentally alters the production landscape by introducing a stable solid intermediate strategy. Unlike conventional routes that struggle with unstable oily residues or hygroscopic salts, this innovative approach utilizes benzoyl hydrazine and 1-methylhexahydroazepin-4-one as primary raw materials. The process involves a sophisticated sequence of condensation, reduction, and a critical salt-formation step using organic binary weak acids. This technical advancement addresses long-standing challenges in impurity control and storage stability, offering a viable solution for reliable pharmaceutical intermediates supplier networks aiming to optimize their API manufacturing portfolios.
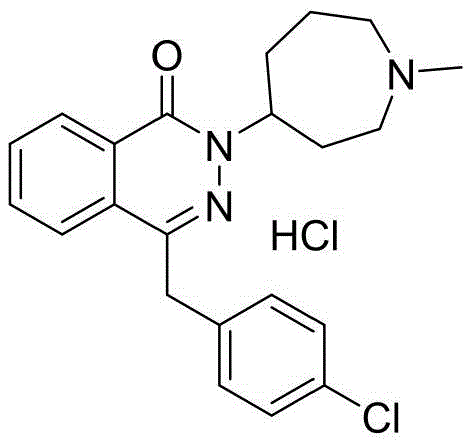
The structural integrity of Azelastine Hydrochloride relies heavily on the precise formation of the phthalazinone ring system coupled with the diazepine moiety. The novelty of this patent lies not just in the final cyclization, but in the stabilization of the precursor chain. By converting the initial reduction product into a crystalline organic acid salt, the process mitigates the degradation pathways common in liquid or amorphous intermediates. This ensures that the subsequent cyclization with 2-(2-(4-chlorophenyl)acetyl)benzoic acid proceeds with maximal efficiency, directly translating to higher yields and reduced waste generation in large-scale reactors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Azelastine has been plagued by significant processing hurdles related to intermediate instability. As documented in European Patent EP2072510A1 and various journal articles, traditional methods often rely on the formation of hydrochloride salts that exist as oils or unstable solids. These intermediates are highly susceptible to hydrolysis due to the strong acidity of the hydrochloric acid radical, which compromises the amido bond integrity. Furthermore, the resulting amino groups are prone to rapid oxidation upon exposure to air, leading to a complex impurity profile that is difficult to purge. Another critical failure point in legacy processes is the tendency of the final product to absorb moisture, forming crystal water that is notoriously difficult to remove via conventional solvent precipitation. This hygroscopic nature complicates drying protocols and often results in bulk drugs that fail to meet strict pharmacopoeial standards for loss on drying.
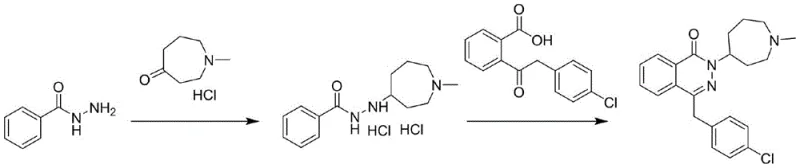
The reliance on hydrochloric acid for intermediate salt formation in prior art creates a corrosive environment that limits equipment lifespan and necessitates specialized lining materials. Additionally, the inability to isolate a stable solid intermediate forces manufacturers to carry forward crude mixtures into the cyclization step, thereby propagating impurities into the final API. The presence of crystal water in the final product not only affects the assay value but can also influence the crystal form, potentially altering bioavailability and dissolution rates. These cumulative defects result in lower overall yields, typically hovering below acceptable commercial thresholds, and require extensive, cost-intensive purification steps such as repeated recrystallization or chromatography to achieve medicinal grade quality.
The Novel Approach
The methodology outlined in CN113045547B introduces a paradigm shift by employing organic binary weak acids, such as succinic acid, fumaric acid, or maleic acid, to form a solid intermediate (Compound 2). This strategic substitution of strong mineral acids with weak organic acids dramatically enhances the thermal and chemical stability of the intermediate. The resulting solid is non-hygroscopic, easy to filter, and can be stored without significant degradation, providing a crucial buffer in the supply chain. This stability allows for rigorous quality control checks before proceeding to the expensive cyclization step, ensuring that only high-purity material enters the final stages of synthesis. The process effectively decouples the reduction phase from the cyclization phase, allowing each step to be optimized independently for maximum conversion efficiency.
Moreover, the final salification step utilizes toluene as a solvent in conjunction with a water separator, facilitating azeotropic distillation to remove water continuously during reflux. This engineering control ensures that the final Azelastine Hydrochloride is obtained in an absolutely anhydrous state, eliminating the persistent issue of crystal water inclusion. The operational simplicity of this water separation technique makes the process highly amenable to scale-up, reducing the batch cycle time and energy consumption associated with prolonged vacuum drying. By achieving a purity of greater than 99 percent and yields exceeding 80 percent, this novel approach sets a new benchmark for cost reduction in pharmaceutical intermediates manufacturing, offering a distinct competitive advantage over legacy synthetic routes.
Mechanistic Insights into Organic Acid-Mediated Stabilization and Cyclization
The core mechanistic advantage of this process begins with the condensation of benzoyl hydrazine and 1-methylhexahydroazepin-4-one, catalyzed by potassium hydroxide in an aqueous medium. Following this, sodium borohydride reduction converts the imine linkage into a stable amine, yielding Compound 1. The critical innovation occurs in the subsequent step where Compound 1 reacts with an organic acid. Unlike hydrochloric acid which forms a labile salt, the organic acid forms a robust crystalline lattice through hydrogen bonding and ionic interactions. This solid-state stabilization protects the reactive amine functionality from oxidative degradation and prevents the hydrolysis of the adjacent amide bond. The choice of a binary weak acid ensures that the protonation is reversible under the specific conditions of the next step, allowing for the controlled release of the free amine necessary for cyclization without generating excessive acidic byproducts.
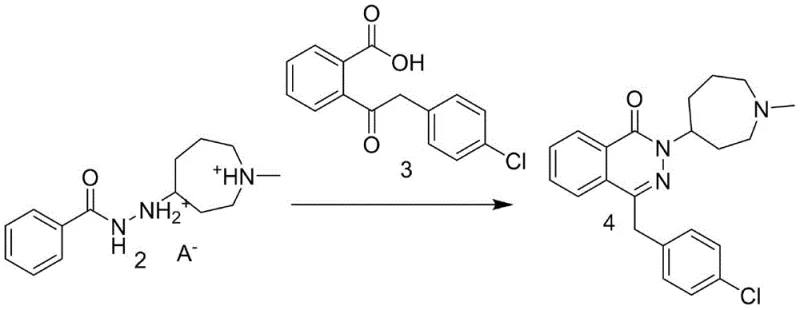
The cyclization mechanism involves the hydrolysis of the stable solid intermediate followed by a condensation reaction with 2-(2-(4-chlorophenyl)acetyl)benzoic acid (Compound 3). Under reflux conditions in hydrochloric acid, the organic acid salt dissociates, and the hydrazine moiety attacks the ketone carbonyl of Compound 3. This nucleophilic attack initiates the ring closure to form the phthalazinone core, releasing a molecule of water. The use of 36 percent hydrochloric acid provides the necessary acidic environment to activate the carbonyl group while maintaining the solubility of the intermediates. The subsequent neutralization and extraction steps are designed to remove the organic acid byproducts and unreacted starting materials, ensuring that the oily Compound 4 obtained is of sufficient purity for the final salification. This precise control over the reaction equilibrium minimizes the formation of regio-isomers and open-chain byproducts, which are common impurities in less controlled cyclization reactions.
How to Synthesize Azelastine Hydrochloride Efficiently
The synthesis protocol detailed in the patent provides a clear, step-by-step framework for producing high-quality Azelastine Hydrochloride suitable for commercial deployment. The process begins with the preparation of the reduced hydrazine derivative, followed by its conversion into a stable organic acid salt. This solid intermediate is then subjected to hydrolysis and cyclization with the chlorophenyl acetyl benzoic acid derivative. The final step involves salt formation in toluene with continuous water removal. For process chemists and plant managers, adhering to the specific molar ratios and temperature controls described—such as maintaining the reduction at 0-10°C and the cyclization under reflux—is critical for replicating the high yields and purity reported. Detailed standardized operating procedures for each unit operation are essential to maintain consistency across batches.
- Perform condensation of benzoyl hydrazine and 1-methylhexahydroazepin-4-one followed by sodium borohydride reduction to obtain Compound 1.
- React Compound 1 with an organic binary weak acid (e.g., succinic acid) to form the stable solid intermediate Compound 2.
- Hydrolyze Compound 2 and cyclize with 2-(2-(4-chlorophenyl)acetyl)benzoic acid (Compound 3) to form the phthalazinone core (Compound 4).
- Salify Compound 4 with hydrochloric acid in toluene using azeotropic water separation to yield the final anhydrous Azelastine Hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the transition to this novel synthetic route offers substantial strategic benefits beyond mere technical superiority. The ability to isolate a stable solid intermediate fundamentally changes the logistics of API manufacturing by allowing for the decoupling of production stages. This means that the intermediate can be produced, quality-tested, and stored for extended periods without degradation, providing a buffer against supply chain disruptions and demand fluctuations. The elimination of hygroscopic materials reduces the need for specialized climate-controlled storage facilities, thereby lowering overhead costs associated with warehousing. Furthermore, the robustness of the process reduces the risk of batch failures, ensuring a more predictable and reliable supply of the final active ingredient for downstream formulation teams.
- Cost Reduction in Manufacturing: The implementation of this process leads to significant cost optimization by eliminating the need for complex purification steps required to remove unstable impurities found in legacy routes. The use of common, inexpensive organic acids like succinic acid instead of specialized reagents lowers raw material costs. Additionally, the high yield exceeding 80 percent means less starting material is wasted per kilogram of final product, directly improving the cost of goods sold (COGS). The simplified workup procedures, such as straightforward filtration of the solid intermediate and azeotropic drying, reduce labor hours and solvent consumption, contributing to a leaner and more economical manufacturing footprint.
- Enhanced Supply Chain Reliability: The stability of the solid intermediate (Compound 2) ensures that production schedules are not dictated by the immediate instability of reaction mixtures. This allows for flexible batch planning and the potential to stockpile intermediates during periods of low raw material cost. The process utilizes widely available commodity chemicals such as toluene, dichloromethane, and potassium hydroxide, reducing the risk of supply bottlenecks associated with exotic catalysts or reagents. By mitigating the risk of oxidation and hydrolysis, the process guarantees a consistent quality profile, reducing the likelihood of rejected shipments and ensuring uninterrupted supply to global pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations like reflux, crystallization, and liquid-liquid extraction that are easily transferable from pilot plant to multi-ton commercial reactors. The use of toluene for water separation is a well-established industrial technique that minimizes energy consumption compared to high-vacuum drying of hydrates. Furthermore, the high purity of the intermediates reduces the load on wastewater treatment systems by minimizing the discharge of complex organic impurities. The ability to recover and recycle solvents like dichloromethane and toluene further enhances the environmental sustainability of the process, aligning with modern green chemistry principles and regulatory compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the operational advantages and quality benchmarks achievable with this method. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term value proposition of this supply source.
Q: Why is the solid intermediate superior to traditional hydrochloride salts?
A: Traditional hydrochloride intermediates are often oily or hygroscopic solids that suffer from poor stability and amide bond hydrolysis. The novel solid intermediate (Compound 2) formed with organic acids is non-hygroscopic, exhibits stability greater than 99 percent purity, and eliminates oxidation risks associated with free amino groups.
Q: How does this process ensure the removal of crystal water?
A: The final salification step utilizes toluene as a solvent connected to a water separator. This allows for azeotropic distillation which effectively removes water during reflux, ensuring the final Azelastine Hydrochloride product is absolutely anhydrous and meets stringent drying weight loss standards.
Q: What are the yield and purity benchmarks for this method?
A: According to patent data, this method achieves a total yield exceeding 80 percent, which is a significant improvement over prior art. Furthermore, the HPLC purity of the final product consistently reaches above 99 percent, minimizing the need for extensive downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azelastine Hydrochloride Supplier
The technical breakthroughs presented in patent CN113045547B highlight the immense potential for optimizing the production of this critical antihistamine agent. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such sophisticated chemistry to the market. Our facility is equipped with rigorous QC labs and stringent purity specifications that ensure every batch of Azelastine Hydrochloride meets the highest international standards. We understand the nuances of handling sensitive intermediates and implementing azeotropic drying technologies, positioning us as an ideal partner for companies seeking to secure a stable and high-quality supply of this API.
We invite you to engage with our technical procurement team to discuss how this optimized route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this solid-intermediate process. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering superior value through innovation and operational excellence in the pharmaceutical intermediates sector.
