Advanced NaDT-Catalyzed Synthesis of Trifluoromethyl Thioesters for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust methods to incorporate fluorine-containing motifs into complex molecular architectures, driven by the profound impact of fluorination on metabolic stability and membrane permeability. Patent CN113860981A introduces a groundbreaking preparation method for trifluoromethyl thioesters, utilizing aldehydes and R1-SCF3 reagents under the catalytic influence of sodium decatungstate. This innovation represents a significant leap forward in fine chemical manufacturing, offering a pathway that circumvents the harsh conditions and toxic reagents often associated with traditional trifluoromethylthiolation strategies. By leveraging a photocatalytic approach, this technology enables the efficient construction of C-SCF3 bonds with remarkable functional group tolerance, addressing a critical bottleneck in the synthesis of high-value pharmaceutical intermediates. The strategic implementation of this patent data allows for the development of scalable processes that align with modern green chemistry principles while maintaining the rigorous purity standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl thioesters has been plagued by significant technical hurdles that impede large-scale commercial adoption and increase overall production costs. Conventional methodologies often rely on the use of unstable or highly toxic reagents, such as mercury-based compounds or stoichiometric amounts of expensive transition metal complexes, which pose severe environmental and safety risks in an industrial setting. Furthermore, many existing protocols require stringent reaction conditions, including extreme temperatures or the use of hazardous solvents, which can lead to the degradation of sensitive functional groups present in complex drug candidates. The reliance on precious metal catalysts not only inflates the raw material costs but also introduces challenges related to the removal of trace metal impurities, a critical quality attribute for active pharmaceutical ingredients. These limitations collectively result in lower overall yields, extended processing times, and a larger environmental footprint, making traditional routes less attractive for cost-sensitive supply chains.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent data employs sodium decatungstate as a cost-effective and earth-abundant photocatalyst to drive the transformation under exceptionally mild conditions. This method eliminates the need for expensive noble metals and avoids the use of disodium hydrogen phosphate, thereby simplifying the reaction workup and reducing the generation of inorganic waste streams. The process operates at room temperature under nitrogen protection with 390 nm LED irradiation, ensuring that thermally labile substrates remain intact throughout the synthesis. By optimizing the catalyst loading and reaction time, this strategy significantly enhances the yield of the desired trifluoromethyl thioester while minimizing side reactions that often complicate purification. 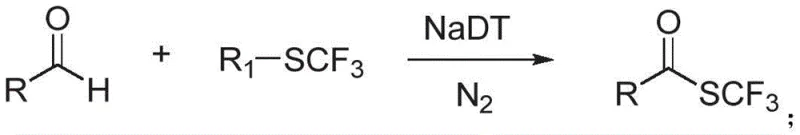
Mechanistic Insights into NaDT-Catalyzed Trifluoromethylthiolation
The mechanistic underpinning of this synthesis relies on a sophisticated photocatalytic cycle initiated by the excitation of the sodium decatungstate catalyst under 390 nm irradiation. Upon absorbing light energy, the catalyst transitions to an excited state which rapidly decays to form a triplet intermediate capable of abstracting a hydrogen atom from the aldehyde substrate. This hydrogen atom transfer (HAT) process generates a reactive acyl radical and a reduced catalyst species, setting the stage for the subsequent bond-forming events that define the efficiency of the transformation. The acyl radical then engages in a radical transfer process with the trifluoromethylthiolating agent, effectively installing the SCF3 group onto the carbonyl carbon to yield the target thioester product. This radical-mediated pathway is distinct from ionic mechanisms and offers unique selectivity profiles that are advantageous for complex molecule synthesis.
Crucially, the catalytic cycle is completed through a single electron transfer (SET) process between the reduced decatungstate species and the radical byproducts, regenerating the active photocatalyst for further turnover. This regeneration step is vital for maintaining catalytic efficiency and ensuring that only minimal amounts of the sodium decatungstate are required to drive the reaction to completion. The mechanism also accounts for the high functional group compatibility observed, as the mild radical conditions do not aggressively attack other sensitive moieties within the substrate molecule. Understanding this catalytic loop is essential for process chemists aiming to troubleshoot potential scale-up issues or adapt the conditions for novel substrate classes. 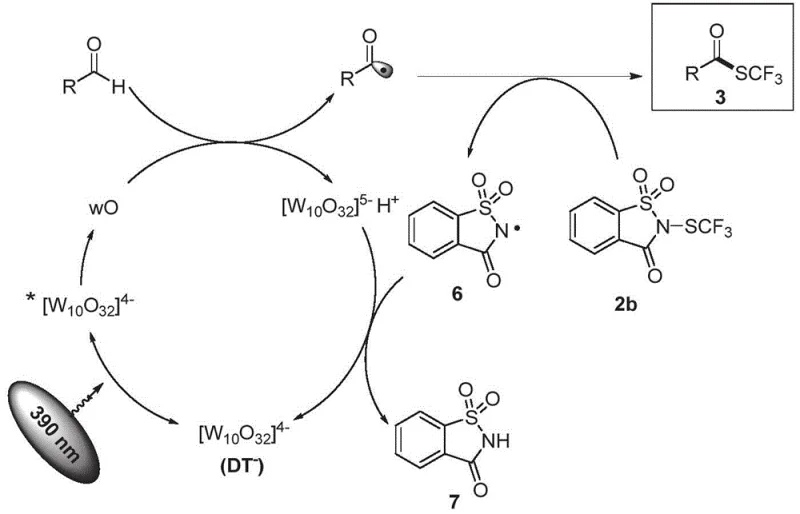
How to Synthesize Trifluoromethyl Thioester Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the specific operational parameters outlined in the patent to ensure optimal reproducibility and yield. The process begins with the preparation of a reaction mixture containing the aldehyde substrate and the chosen trifluoromethylthiolating reagent in dry acetonitrile, strictly under a nitrogen atmosphere to prevent oxidative side reactions. Following the addition of the sodium decatungstate catalyst, the mixture is subjected to irradiation from a 40W LED light source with a wavelength of 390 nm, which provides the necessary energy to drive the photocatalytic cycle without excessive heat generation. The reaction typically proceeds for a duration of 10 to 14 hours, after which the product is isolated through a straightforward workup involving aqueous washing and flash chromatography purification.
- Mix the aldehyde substrate and the trifluoromethylthiolating agent (R1-SCF3) in dry acetonitrile under a nitrogen atmosphere.
- Add sodium decatungstate (NaDT) catalyst (1.5 to 2 mol%) to the reaction mixture.
- Irradiate the mixture with a 390 nm LED light source at room temperature for 10 to 14 hours, then purify via flash chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this sodium decatungstate-catalyzed methodology offers substantial strategic advantages that directly impact the bottom line and operational resilience. The elimination of expensive precious metal catalysts translates into a significant reduction in raw material costs, allowing for more competitive pricing structures in the final supply of trifluoromethyl thioester intermediates. Moreover, the mild reaction conditions and the use of common solvents like acetonitrile simplify the safety protocols required for manufacturing, thereby reducing the regulatory burden and insurance costs associated with hazardous chemical processing. The robustness of the reaction across a wide range of substrates ensures a reliable supply of diverse intermediates, mitigating the risk of production delays caused by substrate-specific failures. These factors collectively contribute to a more agile and cost-efficient supply chain capable of responding rapidly to market demands.
- Cost Reduction in Manufacturing: The substitution of costly noble metal catalysts with inexpensive sodium decatungstate drastically lowers the direct material costs associated with the synthesis process. By removing the need for expensive metal scavenging steps to meet residual metal specifications, the downstream processing costs are also significantly reduced, leading to overall economic efficiency. The simplified reaction setup and the ability to run the reaction at room temperature further decrease energy consumption and equipment wear, contributing to long-term operational savings. These cumulative cost benefits make the process highly attractive for large-scale commercial production where margin optimization is critical.
- Enhanced Supply Chain Reliability: The use of readily available and stable reagents ensures that the supply chain is not vulnerable to the geopolitical or market fluctuations often associated with rare earth or precious metal sourcing. The high functional group tolerance of the method means that a single standardized process can be applied to a broad portfolio of intermediates, streamlining inventory management and reducing the complexity of the manufacturing schedule. This standardization enhances the predictability of lead times and ensures a consistent flow of materials to downstream customers, fostering stronger long-term partnerships. Reliability is further bolstered by the mild conditions which reduce the likelihood of batch failures due to thermal runaways or equipment malfunctions.
- Scalability and Environmental Compliance: The photochemical nature of the reaction is inherently scalable using modern flow chemistry or large-scale LED reactor technologies, facilitating a smooth transition from laboratory grams to commercial tonnage. The reduction in hazardous waste generation, particularly the avoidance of heavy metal residues, aligns with increasingly stringent environmental regulations and corporate sustainability goals. This eco-friendly profile not only minimizes waste disposal costs but also enhances the brand reputation of the manufacturer as a responsible supplier of fine chemicals. The process design supports continuous improvement initiatives aimed at further reducing the environmental footprint of pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis technology for trifluoromethyl thioesters. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details is crucial for R&D teams evaluating the feasibility of this route for their specific pipeline candidates and for procurement officers assessing the long-term viability of the supply source.
Q: What catalyst is used in this trifluoromethyl thioester synthesis?
A: The process utilizes sodium decatungstate (NaDT) as a cheap and efficient photocatalyst, avoiding expensive precious metals.
Q: What are the reaction conditions for this transformation?
A: The reaction proceeds under nitrogen protection at room temperature using 390 nm LED irradiation in acetonitrile solvent.
Q: Does this method tolerate diverse functional groups?
A: Yes, the method shows excellent compatibility with various substrates including heteroaromatic and aliphatic aldehydes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Thioester Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure necessary to translate this innovative patent technology into a robust commercial supply chain for trifluoromethyl thioesters. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of global pharmaceutical and agrochemical clients without compromising on quality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for drug substance manufacturing. Our commitment to process excellence allows us to offer a reliable source of high-purity trifluoromethyl thioesters that support your critical development timelines.
We invite you to engage with our technical procurement team to discuss how this cost-effective synthesis route can be integrated into your supply strategy for complex pharmaceutical intermediates. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your target molecules and volume needs. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate the performance of this technology against your current benchmarks. Let us collaborate to optimize your supply chain and accelerate the delivery of life-saving medicines to the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
