Scalable Metal-Free Synthesis of Trifluoromethyl Thioesters for Pharmaceutical Applications
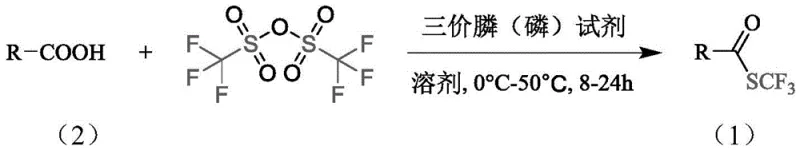
The introduction of fluorine-containing groups, particularly the trifluoromethylthio (SCF3) moiety, has become a cornerstone strategy in modern medicinal chemistry for enhancing the lipophilicity and metabolic stability of drug candidates. However, the practical application of these valuable motifs has long been hindered by the prohibitive cost and toxicity of traditional sulfenylation reagents. A groundbreaking solution to these challenges is detailed in patent CN112358427B, which discloses a novel, metal-free synthetic methodology for constructing carbon-sulfur bonds using trifluoromethanesulfonic anhydride as the sulfur source. This innovative approach represents a paradigm shift for the production of high-purity pharmaceutical intermediates, offering a route that is not only economically superior but also operationally simpler and environmentally friendlier than existing state-of-the-art technologies.
By leveraging widely available commodity chemicals such as triphenylphosphine and trifluoromethanesulfonic anhydride, this process eliminates the dependency on precious metal catalysts and hazardous mercury or silver salts. For R&D directors and process chemists, this translates to a robust platform capable of generating diverse trifluoromethyl thioester libraries with excellent functional group tolerance. The ability to perform these transformations under mild conditions (0°C to 50°C) and even in the presence of air significantly lowers the barrier to entry for scaling these reactions from milligram discovery batches to multi-kilogram pilot runs, addressing a critical bottleneck in the development of next-generation fluorinated therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl thioesters has relied heavily on reagents that pose significant safety, environmental, and economic liabilities. Traditional protocols often utilize bis(trifluoromethyl)mercury or silver trifluoromethanethiolate, both of which are associated with severe toxicity profiles and exorbitant market prices, with some reagents costing upwards of 5000 yuan per gram. Furthermore, many established methods require the pre-functionalization of substrates into highly reactive acyl chlorides, adding extra synthetic steps and generating additional waste streams. The reliance on transition metal catalysts, such as copper or nickel complexes, introduces another layer of complexity, necessitating rigorous downstream purification to meet stringent residual metal specifications required for active pharmaceutical ingredients (APIs). These factors collectively create a fragile supply chain vulnerable to raw material shortages and regulatory scrutiny.
The Novel Approach
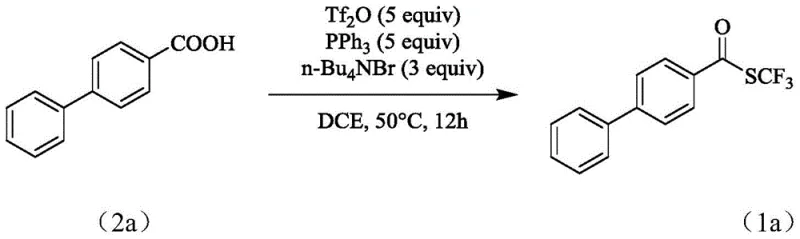
In stark contrast, the methodology described in patent CN112358427B circumvents these obstacles by employing trifluoromethanesulfonic anhydride, a reagent priced at merely 1.98 yuan/g, as the trifluoromethylthio source. This direct deoxydative trifluoromethylthiolation of carboxylic acids proceeds efficiently in the presence of a trivalent phosphine reducing agent, such as triphenylphosphine, without the need for any metal catalysis. The reaction conditions are remarkably benign, operating effectively at temperatures ranging from 0°C to 50°C over a period of 8 to 24 hours. Crucially, the process demonstrates insensitivity to moisture and oxygen, allowing reactions to be conducted under normal pressure air atmospheres rather than requiring expensive inert gas manifolds or gloveboxes. This simplification of operational parameters drastically reduces the technical overhead for manufacturing teams while maintaining high yields, as evidenced by the successful synthesis of complex biphenyl derivatives with yields reaching up to 80.1%.
Mechanistic Insights into Phosphine-Mediated Deoxydative Trifluoromethylthiolation
The core of this transformative chemistry lies in the activation of the carboxylic acid hydroxyl group by trifluoromethanesulfonic anhydride to form a highly reactive mixed anhydride intermediate. This activated species is subsequently attacked by the trivalent phosphine reagent, which serves a dual role as both a nucleophile and a reducing agent. The phosphine facilitates the cleavage of the carbon-oxygen bond and the concurrent formation of the carbon-sulfur bond, effectively replacing the hydroxyl group with the trifluoromethylthio moiety. The inclusion of additives, such as quaternary ammonium salts (e.g., tetrabutylammonium bromide or iodide), further enhances the reaction efficiency by stabilizing ionic intermediates and improving the solubility of reagents in organic solvents like 1,2-dichloroethane. This mechanistic pathway avoids the generation of free radical species that often lead to side reactions, thereby ensuring a clean impurity profile that is essential for pharmaceutical applications.
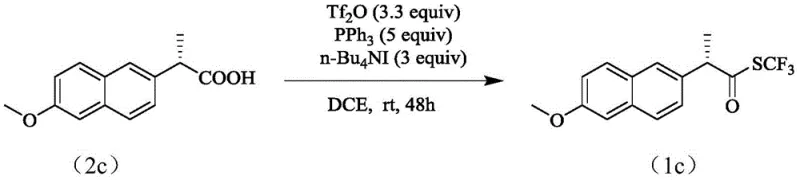
From an impurity control perspective, the absence of transition metals eliminates the risk of metal-catalyzed decomposition pathways or difficult-to-remove metal residues. The reaction byproducts primarily consist of phosphine oxides and trifluoromethanesulfonic acid derivatives, which are generally easier to separate from the target thioester via standard aqueous workup and chromatography. The compatibility of this mechanism with a wide array of functional groups—including halogens, ethers, and even sensitive chiral centers—underscores its versatility. For instance, the successful conversion of naproxen, a chiral non-steroidal anti-inflammatory drug precursor, into its corresponding trifluoromethyl thioester without racemization highlights the mildness and selectivity of this activation strategy, making it an ideal candidate for the late-stage functionalization of complex drug molecules.
How to Synthesize Trifluoromethyl Thioesters Efficiently
The implementation of this synthesis route offers a streamlined workflow for process development teams aiming to access trifluoromethylated building blocks. The protocol begins with the dissolution of the carboxylic acid starting material and the phosphine reducing agent in a suitable anhydrous organic solvent, followed by the controlled addition of the anhydride activator. The reaction progress is easily monitored via standard analytical techniques, and the workup involves simple aqueous quenching and extraction, avoiding complex distillation or crystallization steps often required in metal-catalyzed processes. Detailed standardized synthetic steps for optimizing yield and purity across different substrate classes are outlined in the guide below, providing a clear roadmap for laboratory execution.
- Dissolve the carboxylic acid substrate and trivalent phosphine reducing agent (e.g., triphenylphosphine) in a dry organic solvent such as 1,2-dichloroethane.
- Add trifluoromethanesulfonic anhydride slowly under cooling conditions (0°C to 50°C) optionally with a quaternary ammonium salt additive.
- Stir the reaction mixture for 8 to 24 hours, then quench with water, extract with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this metal-free synthesis protocol offers compelling economic and logistical benefits that directly impact the bottom line. By shifting away from exotic, high-cost reagents like silver salts or unstable tetramethylammonium derivatives, manufacturers can achieve substantial cost savings in raw material acquisition. The use of trifluoromethanesulfonic anhydride, which is produced on a massive industrial scale for other applications, ensures a stable and reliable supply chain that is less susceptible to the volatility often seen in niche fine chemical markets. Furthermore, the elimination of expensive metal catalysts removes the need for costly scavenging resins or specialized filtration equipment, simplifying the capital expenditure requirements for production facilities.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the dramatic disparity in reagent pricing, where the key sulfur source in this new method costs a fraction of traditional alternatives. Additionally, the removal of transition metal catalysts eliminates the entire unit operation dedicated to metal removal and validation, which is a significant cost driver in GMP manufacturing. The simplified downstream processing reduces solvent consumption and waste disposal fees, contributing to a leaner and more cost-effective overall production process that enhances profit margins for high-volume intermediates.
- Enhanced Supply Chain Reliability: Reliance on commodity chemicals such as triphenylphosphine and common organic solvents ensures that production schedules are not held hostage by the lead times of specialized reagents. The robustness of the reaction conditions, which tolerate air and moisture, reduces the risk of batch failures due to environmental excursions, thereby improving overall equipment effectiveness (OEE). This reliability allows for more accurate forecasting and inventory management, ensuring consistent delivery of critical intermediates to downstream API synthesis sites without unexpected interruptions.
- Scalability and Environmental Compliance: The mild thermal profile of the reaction (0°C to 50°C) minimizes energy consumption for heating or cryogenic cooling, aligning with green chemistry principles and reducing the carbon footprint of the manufacturing process. The absence of toxic heavy metals like mercury or cadmium simplifies regulatory compliance and waste treatment, as the effluent streams do not require specialized hazardous waste handling protocols. This environmental advantage facilitates smoother regulatory approvals and supports corporate sustainability goals, making the technology attractive for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethylthiolation technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on the practical aspects of adopting this method for industrial scale-up. Understanding these nuances is critical for project managers evaluating the feasibility of integrating this route into existing production pipelines.
Q: What are the cost advantages of this trifluoromethylthiolation method compared to traditional silver or mercury reagents?
A: This method utilizes trifluoromethanesulfonic anhydride, which costs approximately 1.98 yuan/g, compared to traditional reagents like tetramethylammonium trifluoromethanethiolate (5952 yuan/g) or silver trifluoromethanethiolate (790 yuan/g), resulting in drastic raw material cost reductions.
Q: Does this synthesis require inert atmosphere or specialized equipment?
A: No, the process is robust and can be conducted under normal pressure air atmosphere without the need for gloveboxes or strictly anhydrous conditions, simplifying operational requirements significantly.
Q: Is this method compatible with chiral pharmaceutical intermediates?
A: Yes, the mild reaction conditions (0-50°C) preserve stereochemistry, as demonstrated by the successful conversion of naproxen derivatives without racemization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Thioester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for the rapid development of fluorinated pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of trifluoromethyl thioester delivered meets the highest quality standards required for global drug registration.
We invite you to collaborate with our technical team to explore how this cost-effective technology can optimize your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this novel protocol for your target molecules. We encourage you to contact our technical procurement team today to discuss your project requirements,索取 specific COA data, and receive comprehensive route feasibility assessments tailored to your unique production needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
