Advanced Mitsunobu Cyclization for High-Purity Escitalopram Manufacturing and Commercial Scale-Up
Introduction to Next-Generation Escitalopram Synthesis
The pharmaceutical landscape for antidepressant manufacturing is undergoing a significant transformation driven by the need for higher optical purity and more sustainable processing methods. Patent CN101607951A introduces a groundbreaking approach to the preparation of Escitalopram, the S-enantiomer of Citalopram, which is renowned for its superior therapeutic index compared to the racemic mixture. This technology leverages the Mitsunobu reaction to achieve cyclization of the S-type diol intermediate, addressing critical limitations found in legacy synthetic routes. By shifting away from harsh acidic conditions or unstable ester intermediates, this method ensures exceptional retention of stereochemical integrity while delivering yields that approach theoretical maximums. For R&D leaders and procurement strategists, understanding this shift is vital for securing a reliable escitalopram supplier capable of meeting stringent global pharmacopeia standards.
The core innovation lies in the specific combination of a coordination phosphine compound, an azo reagent, and a proton-donating agent within an aprotic organic solvent system. This triad of reagents facilitates a smooth intramolecular substitution that constructs the phthalan ring system essential for the drug's bioactivity. Unlike previous iterations of this synthesis which struggled with byproduct formation and chiral degradation, this protocol operates under mild thermal conditions, typically between 0°C and 30°C. The result is a robust manufacturing pathway that minimizes waste generation and maximizes the utility of expensive chiral starting materials, positioning it as a preferred choice for cost reduction in pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the cyclization of the diol precursor to form the Escitalopram backbone has been plagued by significant chemical inefficiencies. Traditional acidic cyclization methods, such as those utilizing 70% concentrated sulfuric acid at elevated temperatures, are notorious for inducing racemization. This loss of optical purity is catastrophic for a chiral drug where the S-isomer is over 100 times more potent than the R-isomer, necessitating complex and costly downstream purification to remove the inactive or potentially harmful enantiomer. Furthermore, alkaline approaches involving the formation of unstable esters via mesyl or tosyl chlorides suffer from poor selectivity. These methods often lead to the di-esterification of both hydroxyl groups on the diol substrate, creating difficult-to-separate impurities that drag overall yields down to a mere 55% to 65% range.
Beyond the chemical yield issues, conventional methods impose heavy burdens on supply chain reliability and environmental compliance. The use of strong mineral acids or reactive acid chlorides requires specialized corrosion-resistant equipment and rigorous safety protocols to handle hazardous exotherms. Additionally, the generation of stoichiometric amounts of salt waste and the need for extensive chromatographic purification to achieve acceptable enantiomeric excess (ee) values inflate the cost of goods sold. For a procurement manager, these factors translate into volatile pricing and extended lead times, as any deviation in reaction control can render an entire batch off-specification. The inherent instability of the intermediates in these older processes also limits the ability to scale up production without risking significant batch failures.
The Novel Approach
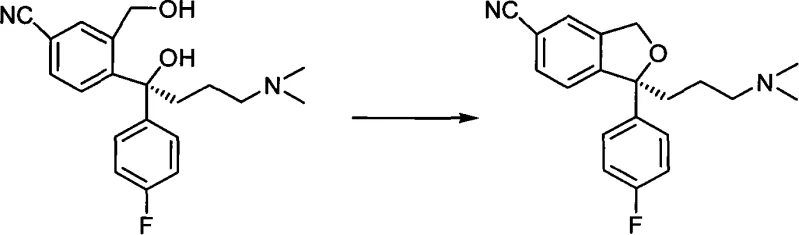
The methodology described in CN101607951A represents a paradigm shift by employing the Mitsunobu reaction for this specific cyclization step. This approach elegantly bypasses the pitfalls of acid-catalyzed racemization and base-mediated side reactions. By utilizing reagents such as triphenylphosphine or trimethylphosphine in conjunction with azodicarboxylates like DIAD or DEAD, the reaction activates the primary hydroxyl group selectively while leaving the tertiary alcohol untouched due to steric constraints. This selectivity is the key to achieving the reported yields of over 94% in optimized embodiments, effectively doubling the material efficiency compared to older alkaline methods. The process maintains the chiral center's configuration with remarkable fidelity, preserving ee values above 98.5% throughout the transformation.
Furthermore, the operational simplicity of this novel approach cannot be overstated. The reaction proceeds smoothly in common aprotic solvents like tetrahydrofuran or ethyl acetate under a nitrogen atmosphere, eliminating the need for extreme temperatures or pressures. Post-reaction workup is straightforward, often involving simple filtration to remove phosphine oxide byproducts followed by standard aqueous extraction. This streamlined workflow significantly reduces the cycle time per batch and lowers the energy consumption associated with heating and cooling. For commercial scale-up of complex pharmaceutical intermediates, this translates to a more predictable production schedule and a drastic simplification of the plant's utility requirements, making it an ideal candidate for high-volume manufacturing facilities.
Mechanistic Insights into Mitsunobu-Mediated Cyclization
To fully appreciate the technical superiority of this route, one must examine the mechanistic underpinnings of the Mitsunobu reaction in this context. The process initiates with the formation of a betaine intermediate between the phosphine and the azo reagent, which subsequently activates the proton-donating acid, typically formic acid in this specific application. This activated species then reacts with the primary hydroxyl group of the S-type diol to form an alkoxyphosphonium ion. This intermediate is highly susceptible to nucleophilic attack. In the intramolecular setting of Escitalopram synthesis, the proximal phenolic oxygen or the specific carbon center acts as the nucleophile, displacing the phosphine oxide leaving group in a classic SN2 fashion. The beauty of this mechanism lies in its stereospecificity; while SN2 reactions typically invert configuration, the specific geometry of this cyclization ensures the final product retains the desired S-configuration relative to the starting material's chiral axis.
Crucially, the steric bulk of the phosphine oxide transition state plays a protective role. In the diol substrate, there are two hydroxyl groups: a primary one and a tertiary one. Conventional esterification methods struggle to distinguish between them, leading to the aforementioned di-ester byproducts. However, the Mitsunobu reagents are sterically demanding. The bulky triphenylphosphine or even the smaller trimethylphosphine, when complexed, creates a transition state that is energetically unfavorable for attack at the hindered tertiary position. Consequently, the reaction is highly regioselective for the primary hydroxyl group. This intrinsic selectivity eliminates the formation of the di-ester impurity at the source, rather than relying on difficult purification steps to remove it later. This mechanistic feature is the primary driver for the observed high yields and the clean impurity profile, offering R&D directors a clear path to robust process validation.
How to Synthesize Escitalopram Efficiently
Implementing this synthesis route requires precise control over reagent stoichiometry and environmental conditions to maximize the benefits outlined above. The patent data suggests that maintaining anhydrous conditions is paramount, as moisture can quench the reactive intermediates and reduce the efficiency of the azo reagent. The molar ratios are also critical; while a 1:1 ratio is theoretically sufficient, the patent recommends using a slight excess of the phosphine and azo components, typically in the range of 1.2 to 2.5 equivalents relative to the diol substrate, to drive the reaction to completion. The following guide outlines the standardized operational procedure derived from the patent examples, serving as a baseline for process engineers looking to adopt this technology.
- Prepare the reaction vessel under inert gas protection with anhydrous aprotic solvent such as tetrahydrofuran.
- Add the S-type diol substrate followed by the coordination phosphine compound and proton donating agent at room temperature.
- Dropwise add the azo reagent while maintaining temperature between 0°C and 30°C, then stir until completion and purify via extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the adoption of this Mitsunobu-based cyclization offers compelling economic arguments beyond simple yield improvements. The elimination of harsh reagents like concentrated sulfuric acid or corrosive acid chlorides reduces the wear and tear on reactor vessels and piping, extending the lifespan of capital equipment and lowering maintenance costs. Moreover, the high selectivity of the reaction means that raw material consumption is optimized; less starting diol is wasted on side products, directly reducing the variable cost per kilogram of the final API. This efficiency gain is particularly significant given that the chiral diol precursor is itself a high-value intermediate. By maximizing the conversion of this expensive input, manufacturers can achieve substantial cost savings without negotiating lower prices from upstream suppliers.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dramatic increase in yield and the simplification of purification. Traditional methods yielding 55-65% require nearly double the amount of raw materials to produce the same output, inherently doubling the material cost burden. By pushing yields to the 95% range, the effective cost of goods is nearly halved regarding material input. Additionally, the removal of complex chromatographic purification steps, which are often required to fix the optical purity issues of older methods, reduces solvent consumption and labor hours. The ability to use simpler workup procedures like filtration and extraction further lowers the operational expenditure, making the final product more price-competitive in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commercially available and stable reagents. Triphenylphosphine, DIAD, and formic acid are commodity chemicals with robust global supply networks, reducing the risk of bottlenecks associated with specialized or hazardous reagents. The mild reaction conditions (0-30°C) also mean that the process is less sensitive to minor fluctuations in utility performance, such as cooling water temperature variations, which can often cause batch deviations in more sensitive exothermic reactions. This robustness ensures consistent batch-to-batch quality, reducing the rate of rejected batches and ensuring a steady flow of inventory to meet market demand. It effectively de-risks the manufacturing timeline, providing procurement managers with greater confidence in delivery schedules.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is significantly cleaner. The avoidance of heavy metal catalysts or large volumes of inorganic salts simplifies wastewater treatment protocols. The byproduct, triphenylphosphine oxide, is a solid that can often be filtered off and potentially recycled or disposed of more easily than acidic aqueous waste streams. This aligns well with increasingly strict environmental regulations in major pharmaceutical manufacturing hubs. Furthermore, the scalability is proven by the linear nature of the Mitsunobu reaction; moving from laboratory glassware to industrial reactors does not introduce new kinetic complexities. The simple addition protocols and ambient pressure operation make it easy to transfer technology from pilot plants to full-scale commercial production facilities without extensive re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and operational parameters detailed in the patent literature, aiming to clarify the feasibility of adopting this route for large-scale production. Understanding these nuances is essential for technical teams evaluating the transition from legacy processes to this modernized approach.
Q: How does the Mitsunobu method prevent racemization compared to acidic cyclization?
A: Unlike acidic methods using concentrated sulfuric acid which promote racemization, the Mitsunobu reaction proceeds via an SN2 mechanism with inversion that is controlled to retain the specific S-configuration without degrading optical purity.
Q: What yields can be expected from this cyclization process?
A: The patent data demonstrates yields ranging from 89.2% to 96.9%, significantly outperforming traditional alkaline esterification methods which typically achieve only 55% to 65%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes commercially available reagents, operates at mild temperatures (0-30°C), and involves simple post-treatment steps like filtration and extraction, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Escitalopram Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the Mitsunobu cyclization requires a partner with deep technical expertise and proven manufacturing capabilities. Our team has extensively evaluated the pathway described in CN101607951A and integrated its principles into our own process development frameworks. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and optical purity observed in the lab are faithfully reproduced on an industrial scale. Our facilities are equipped with state-of-the-art reactors capable of handling the specific solvent and temperature requirements of this chemistry, backed by rigorous QC labs that enforce stringent purity specifications to meet international pharmacopeia standards.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to optimize their Escitalopram supply chain. By leveraging our technical proficiency in chiral synthesis and cyclization reactions, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data from our recent batches and to discuss route feasibility assessments for your projects. Let us help you secure a stable, high-quality supply of this critical antidepressant intermediate while driving down your overall manufacturing costs through superior process chemistry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
