Advanced Chemoenzymatic Route for High-Purity Escitalopram Intermediates and Commercial Scale-Up
Advanced Chemoenzymatic Route for High-Purity Escitalopram Intermediates and Commercial Scale-Up
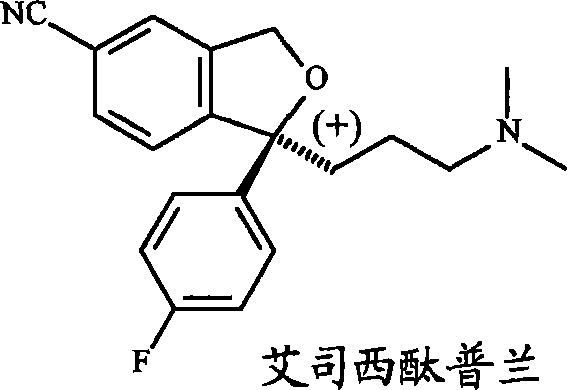
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective methodologies for the production of chiral active pharmaceutical ingredients (APIs), particularly for high-volume antidepressants like S-escitalopram. A pivotal advancement in this domain is detailed in Chinese Patent CN101203614A, which discloses a novel chemoenzymatic method for the synthesis of escitalopram and its pharmaceutically acceptable salts. This technology represents a significant departure from traditional chemical resolution methods by leveraging the specificity of biological catalysts enhanced by chemical complexation. The core innovation lies in the enantioselective enzymatic deacylation of a specific EDTA-complexed substrate using esterase derived from Aspergillus niger. This approach addresses critical pain points in chiral synthesis, including low yields, expensive chiral auxiliaries, and difficult purification steps, positioning it as a highly attractive route for reliable escitalopram intermediate suppliers aiming to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of S-escitalopram, the S(+) enantiomer of citalopram, has relied heavily on classical chemical resolution techniques that are often fraught with inefficiencies. Traditional methods, such as those described in earlier patents like EP347066, typically involve the resolution of racemic mixtures using chiral acids, such as tartaric acid derivatives, to form diastereomeric salts. While effective on a small scale, these processes suffer from a theoretical maximum yield of only 50% for the desired enantiomer, necessitating the disposal or complex recycling of the unwanted isomer. Furthermore, the use of stoichiometric amounts of expensive chiral resolving agents significantly inflates the cost of goods sold (COGS). Alternative approaches involving simulated moving bed (SMB) chromatography offer continuous processing but require substantial capital investment in specialized equipment and high-purity solvents, creating barriers to entry for many manufacturers. Additionally, chemical cyclization steps often require harsh conditions or toxic reagents, complicating waste management and environmental compliance.
The Novel Approach
In stark contrast, the methodology outlined in CN101203614A introduces a sophisticated chemoenzymatic strategy that dramatically enhances process efficiency. Instead of relying on stoichiometric chiral acids, this method utilizes a catalytic amount of immobilized esterase to selectively deacylate a racemic ester substrate. The breakthrough lies in the pre-complexation of the substrate with ethylenediaminetetraacetic acid (EDTA). This novel substrate modification stabilizes the enzyme-substrate interaction, allowing for high enantioselectivity under mild aqueous conditions. By shifting from a stoichiometric chemical resolution to a catalytic enzymatic process, the technology eliminates the need for large quantities of chiral acids and reduces solvent consumption. Moreover, the process is designed to accommodate the recycling of the unwanted enantiomer through acid-catalyzed racemization, theoretically pushing the overall yield well beyond the 50% limit inherent to classical resolutions, thus offering a compelling value proposition for cost reduction in API manufacturing.
Mechanistic Insights into EDTA-Stabilized Enzymatic Resolution
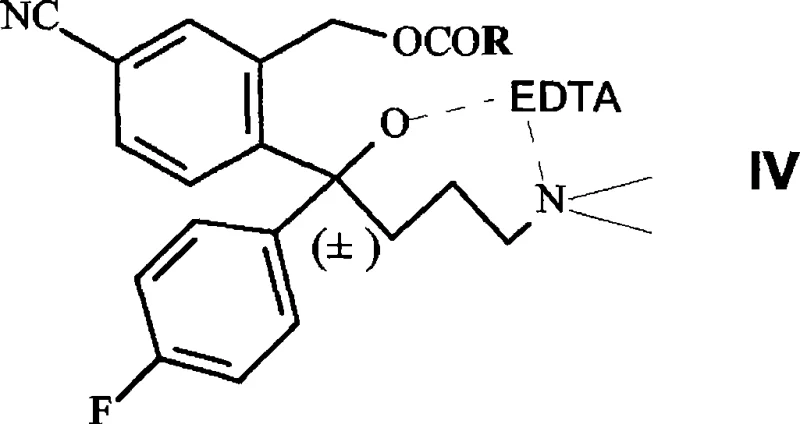
The mechanistic elegance of this process centers on the interaction between the substrate, the enzyme, and the chelating agent. The synthesis begins with the preparation of a racemic ester, specifically 4-(4-dimethylamino)-1-(4'-fluorophenyl)-1-(hydroxybutyl)-3-(acetoxy-methyl)-benzonitrile. In conventional enzymatic resolutions, the presence of trace metal ions or the inherent instability of the enzyme in organic-aqueous mixtures can lead to rapid deactivation. However, in this patented process, the substrate is first converted into a complex with EDTA, forming the structure designated as Formula IV. This complexation serves a dual purpose: it solubilizes the substrate in the aqueous-organic reaction medium and, more critically, creates a microenvironment that preserves the structural integrity of the Aspergillus niger esterase. Patent data indicates that the presence of EDTA improves enzyme stability and increases catalytic activity by approximately 20-30% compared to reactions performed without the chelating agent.
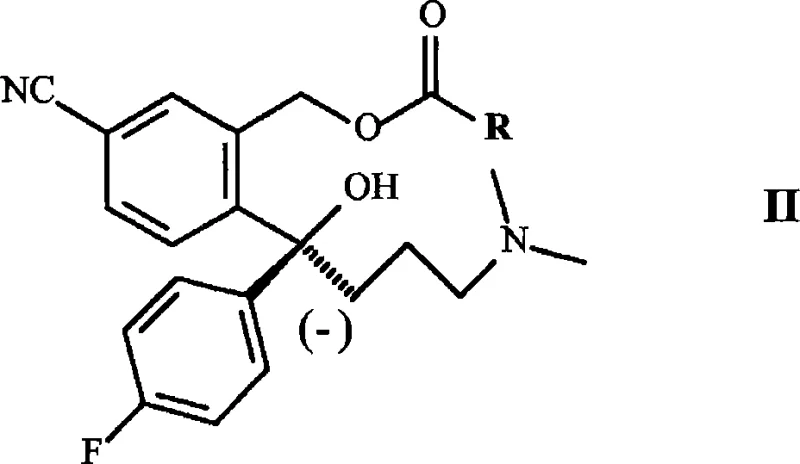
Following complexation, the enzymatic deacylation proceeds with high stereospecificity. The esterase selectively hydrolyzes the ester bond of one enantiomer (the (-) enantiomer in the intermediate stage), leaving the other enantiomer untouched. This kinetic resolution is conducted at a controlled pH of 6 to 8, typically using a phosphate buffer system to maintain optimal enzyme activity. The use of immobilized enzymes, such as esterase fixed on epoxy resin (Eupergit C), further enhances the process by allowing for easy filtration and potential reuse of the biocatalyst. The resulting (-) enantiomer intermediate is then isolated and subjected to hydrolysis to remove the remaining acetyl group, yielding the chiral diol. Finally, a cyclization step, often mediated by activating agents like mesyl chloride or via Mitsunobu-type conditions, closes the phthalan ring to form the final S-escitalopram structure. This multi-step cascade ensures that impurities are minimized at each stage, resulting in a product with high enantiomeric excess (ee) suitable for pharmaceutical applications.
How to Synthesize S-Escitalopram Efficiently
The synthesis of S-escitalopram via this chemoenzymatic route involves a sequence of precise chemical and biological transformations that must be carefully controlled to ensure reproducibility and high purity. The process initiates with a Grignard reaction involving 5-cyanobenzofuranone, followed by acetylation to generate the racemic substrate. The critical step involves the formation of the EDTA complex in an alcohol-water solvent system, followed by the addition of the immobilized esterase. Maintaining the pH within the narrow window of 6.0 to 8.0 is essential for maximizing enzyme turnover. Detailed standardized synthetic steps, including specific reagent ratios, temperature profiles, and workup procedures, are provided in the technical guide below.
- Preparation of the Racemic Precursor: React 5-cyanobenzofuranone with 4-fluorophenyl magnesium bromide and dimethylaminopropyl magnesium chloride to form the diol intermediate, followed by acetylation.
- EDTA Complex Formation: Dissolve the acetylated racemic compound in an alcohol-water mixture with EDTA disodium salt (1.2-1.3 equivalents) to form the stable Formula IV complex.
- Enzymatic Resolution and Cyclization: Treat the EDTA complex with immobilized Aspergillus niger esterase at pH 6-8 to isolate the (-) enantiomer, followed by hydrolysis and cyclization to yield S-escitalopram.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this chemoenzymatic technology translates into tangible strategic advantages beyond mere technical feasibility. The shift from stoichiometric chemical resolution to catalytic enzymatic resolution fundamentally alters the cost structure of production. By eliminating the need for expensive chiral resolving agents and reducing the volume of organic solvents required for crystallization and purification, the process drives down variable costs significantly. Furthermore, the ability to recycle the unwanted enantiomer back into the process stream enhances raw material utilization, effectively lowering the cost per kilogram of the final API. This efficiency is crucial in a competitive generic market where margin compression is a constant challenge.
- Cost Reduction in Manufacturing: The implementation of this EDTA-stabilized enzymatic route removes the dependency on high-cost chiral acids and reduces the number of purification cycles required. The catalytic nature of the enzyme means that a small amount of biocatalyst can process large volumes of substrate, drastically reducing reagent costs. Additionally, the mild reaction conditions (ambient temperature and pressure) lower energy consumption compared to high-temperature chemical cyclizations. The qualitative reduction in hazardous waste generation also minimizes disposal costs, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Sourcing high-purity chiral intermediates is often a bottleneck in API supply chains. This process utilizes readily available starting materials like 5-cyanobenzofuranone and commodity chemicals for the Grignard reagents, reducing reliance on specialized single-source suppliers. The robustness of the immobilized enzyme system ensures consistent batch-to-batch quality, mitigating the risk of production delays caused by out-of-specification batches. Moreover, the scalability of the enzymatic step allows for flexible production scheduling, enabling manufacturers to respond rapidly to fluctuations in market demand without compromising product quality.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental profile of a synthesis route becomes a key differentiator. This chemoenzymatic method operates primarily in aqueous-alcoholic media, significantly reducing the load of volatile organic compounds (VOCs) compared to traditional solvent-heavy processes. The absence of heavy metal catalysts simplifies the purification process and ensures that the final product meets stringent limits for residual metals. The process is inherently safer and more environmentally benign, facilitating easier regulatory approval and long-term sustainability compliance for large-scale commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the chemoenzymatic synthesis of escitalopram intermediates. These insights are derived directly from the experimental data and claims presented in patent CN101203614A, providing a factual basis for evaluating the technology's applicability to your specific manufacturing needs. Understanding these nuances is critical for R&D teams assessing process transfer and procurement teams evaluating supplier capabilities.
Q: How does the EDTA complex improve the enzymatic resolution of escitalopram intermediates?
A: According to patent CN101203614A, forming a complex with EDTA significantly stabilizes the esterase enzyme from Aspergillus niger. This complexation prevents enzyme deactivation and increases catalytic activity by approximately 20-30% compared to non-complexed substrates, leading to higher enantiomeric excess and yield.
Q: Can the unwanted (+) enantiomer be recycled in this process?
A: Yes, the process allows for the recycling of the unwanted (+) enantiomer. By subjecting the residual (+) enantiomer to acid-catalyzed racemization (pH 0.5-4), it can be converted back into the racemic mixture and re-entered into the enzymatic resolution step, thereby maximizing atom economy and reducing raw material costs.
Q: What are the advantages of using immobilized esterase over free enzymes in this synthesis?
A: Using esterase immobilized on epoxy resin (such as Eupergit C) offers superior operational stability and facilitates easy separation of the biocatalyst from the reaction mixture. This enables the reuse of the enzyme derivative, reduces downstream processing complexity, and ensures consistent batch-to-batch quality essential for GMP manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Escitalopram Supplier
The chemoenzymatic synthesis route described in CN101203614A exemplifies the type of innovative process chemistry that NINGBO INNO PHARMCHEM specializes in scaling and optimizing. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs are successfully translated into industrial reality. Our facilities are equipped with state-of-the-art bioreactors and rigorous QC labs capable of handling sensitive enzymatic processes while maintaining stringent purity specifications required for global regulatory markets. We understand the complexities of chiral resolution and are committed to delivering high-quality intermediates that meet the exacting standards of the pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Our technical team is ready to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with NINGBO INNO PHARMCHEM, you gain access to deep process expertise and a reliable supply of critical intermediates. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of confidence and efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
