Advanced Continuous Manufacturing of High-Purity Cyclic Carbonates for Global Supply Chains
The global demand for high-performance energy storage solutions has placed unprecedented pressure on the supply chains of critical battery components, specifically cyclic carbonates like ethylene carbonate and propylene carbonate. Patent CN102250052A introduces a transformative continuous preparation process that addresses the longstanding inefficiencies of traditional batch synthesis. By leveraging a synergistic catalytic system composed of ionic liquids and halogenated zinc salts, this technology enables the atom-economic fixation of carbon dioxide with epoxides under controlled continuous flow conditions. The significance of this innovation extends beyond mere chemical synthesis; it represents a paradigm shift towards sustainable, high-throughput manufacturing capable of meeting the rigorous purity standards required by the lithium-ion battery industry. As a reliable cyclic carbonate supplier, understanding the nuances of this continuous methodology is essential for securing long-term supply stability and cost competitiveness in the volatile electronic chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclic carbonates has been plagued by significant technical and economic hurdles that hinder large-scale commercialization. Traditional methods often rely on the phosgene route, which involves highly toxic reagents and generates substantial hazardous waste, posing severe environmental and safety risks that are increasingly unacceptable in modern regulatory landscapes. Alternatively, transesterification processes require expensive dialkyl carbonates as starting materials and suffer from equilibrium limitations that necessitate complex separation steps to drive the reaction forward. Even direct cycloaddition methods using conventional Lewis acid catalysts frequently encounter issues with catalyst deactivation, difficult product-catalyst separation, and the inability to operate continuously without significant loss of catalytic activity. These batch-oriented processes result in inconsistent product quality, higher energy consumption due to repeated heating and cooling cycles, and ultimately, inflated production costs that erode profit margins for downstream manufacturers.
The Novel Approach
The novel approach detailed in the patent data circumvents these legacy issues through a sophisticated integration of continuous flow chemistry and advanced separation technologies. By employing a dual-reactor system consisting of a stirred tank followed by a plug-flow tubular reactor, the process ensures optimal contact time and mixing efficiency, driving the conversion of epoxides to near completion levels exceeding 99%. Crucially, the implementation of a thin-film evaporator for downstream processing allows for the gentle separation of the high-boiling cyclic carbonate product from the non-volatile ionic liquid catalyst mixture. This design not only preserves the thermal integrity of the heat-sensitive product but also facilitates the immediate recycling of the catalyst solution back into the reactor loop. Consequently, this continuous methodology drastically simplifies the operational workflow, reduces solvent usage, and establishes a robust framework for the cost reduction in battery electrolyte manufacturing by minimizing raw material waste and maximizing asset utilization.
Mechanistic Insights into Ionic Liquid-Zinc Salt Catalyzed Cycloaddition
The core of this technological breakthrough lies in the unique cooperative catalysis mechanism provided by the ionic liquid and zinc halide binary system. The ionic liquid, typically an imidazolium or pyridinium halide, acts as a nucleophilic activator that attacks the less hindered carbon of the epoxide ring, facilitating ring opening. Simultaneously, the zinc halide co-catalyst functions as a Lewis acid, coordinating with the oxygen atom of the epoxide to further polarize the carbon-oxygen bond, thereby lowering the activation energy for the subsequent insertion of carbon dioxide. This dual activation strategy creates a highly reactive intermediate that rapidly cyclizes to form the stable five-membered cyclic carbonate ring. The structural versatility of the ionic liquid cation, where substituents R1 and R2 can be varied to tune solubility and steric properties, allows for precise optimization of the catalyst for specific epoxide substrates, ensuring high selectivity and minimizing the formation of polycarbonate byproducts.
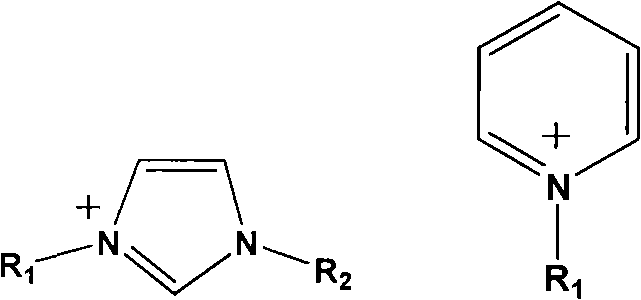
Furthermore, the stability of this catalytic system under continuous operation is a critical factor for industrial viability. Unlike molecular catalysts that may degrade or precipitate out of solution over time, the ionic liquid-zinc salt complex remains homogeneously dissolved in the reaction medium, maintaining consistent activity over extended run times. The mechanism also inherently suppresses side reactions; the mild nucleophilicity of the halide anion prevents excessive polymerization of the epoxide, which is a common impurity in lower-quality grades. By controlling the molar ratio of the zinc salt to the ionic liquid within the range of 3:1 to 10:1, the process achieves a balance between catalytic turnover frequency and selectivity. This precise mechanistic control is what enables the production of high-purity cyclic carbonate suitable for sensitive applications such as pharmaceutical intermediates and electronic grade solvents, where trace impurities can lead to catastrophic failure in end-use devices.
How to Synthesize Cyclic Carbonate Efficiently
The synthesis of high-purity cyclic carbonates via this continuous process requires careful attention to reactor configuration and separation parameters to maximize yield and catalyst longevity. The procedure begins with the preparation of a homogeneous catalyst solution, followed by the continuous feeding of gaseous carbon dioxide and liquid epoxide into the reactor train. Maintaining strict control over temperature and pressure profiles across the stirred tank and tubular reactor sections is vital to ensure complete conversion while preventing thermal degradation. Following the reaction, the effluent stream undergoes a multi-stage separation process involving flash evaporation and thin-film distillation to isolate the product. For a comprehensive understanding of the specific operational parameters and equipment setup required to replicate this efficiency, please refer to the detailed technical guidelines below.
- Prepare the catalyst solution by dissolving imidazolium or pyridinium halide ionic liquids and zinc halide co-catalysts in cyclic carbonate solvent within a mixing unit at 40-80°C.
- Feed the epoxy compound and carbon dioxide gas continuously into a series reactor system comprising a stirred tank followed by a tubular reactor at 120-170°C and 2.0-5.5 MPa.
- Separate the crude product from the catalyst solution using a decompression gas-liquid separator followed by a thin-film evaporator to recover high-purity cyclic carbonate and recycle the catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this continuous ionic liquid catalysis technology offers profound strategic advantages that extend well beyond simple chemical yield improvements. The transition from batch to continuous processing fundamentally alters the cost structure of cyclic carbonate production by eliminating the downtime associated with reactor charging, discharging, and cleaning. This operational continuity translates directly into enhanced supply chain reliability, as production capacity becomes predictable and scalable without the need for exponential increases in physical footprint or capital expenditure. Moreover, the ability to recycle the expensive ionic liquid catalyst system multiple times without significant loss of activity represents a substantial cost saving opportunity, reducing the dependency on fresh catalyst purchases and minimizing the generation of hazardous chemical waste that requires costly disposal.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the elimination of expensive transition metal catalysts and the reduction of solvent consumption through efficient recycling loops. By utilizing a thin-film evaporator for product isolation, the energy demand for separation is significantly lowered compared to traditional vacuum distillation, which often requires high temperatures that can degrade the product. The high single-pass conversion rate of the epoxide means that unreacted raw materials are minimized, reducing the load on recovery systems and lowering the overall cost of goods sold. These efficiencies collectively contribute to a more competitive pricing structure for high-purity cyclic carbonates in the global market.
- Enhanced Supply Chain Reliability: The continuous nature of the reactor system ensures a steady output of product, mitigating the risks of supply interruptions common in batch manufacturing where equipment failures can halt production for days. The robustness of the ionic liquid catalyst system against poisoning and deactivation further secures the production schedule, allowing for long campaign runs that stabilize inventory levels. This reliability is crucial for downstream customers in the battery and pharmaceutical sectors who operate on just-in-time manufacturing schedules and cannot afford fluctuations in the quality or availability of their key solvent inputs.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is streamlined due to the modular nature of the tubular reactor design, which allows for capacity increases by numbering up rather than scaling up vessel size. From an environmental perspective, the process aligns with green chemistry principles by utilizing carbon dioxide as a feedstock, effectively sequestering a greenhouse gas into a valuable chemical product. The closed-loop catalyst recycling system drastically reduces the volume of liquid waste generated, simplifying compliance with increasingly stringent environmental regulations and enhancing the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous synthesis of cyclic carbonates, derived from the specific operational data and benefits outlined in the patent literature. These insights are intended to clarify the feasibility of implementing this technology within existing industrial frameworks and to highlight the specific value propositions for potential partners. Understanding these details is critical for evaluating the technical readiness and commercial viability of adopting this advanced manufacturing route for high-value fine chemicals.
Q: What are the primary advantages of using ionic liquid catalysts for cyclic carbonate synthesis?
A: Ionic liquid catalysts, particularly when paired with zinc halide co-catalysts, offer superior thermal stability and solubility characteristics that facilitate continuous processing. Unlike traditional homogeneous catalysts that are difficult to separate, this system allows for efficient recycling via thin-film evaporation, significantly reducing waste and operational costs while maintaining high catalytic activity over extended periods.
Q: How does the continuous process improve product purity compared to batch methods?
A: The continuous process described utilizes a combination of a stirred tank and a plug-flow tubular reactor, which ensures uniform mixing and complete conversion of reactants. Coupled with downstream separation using a thin-film evaporator operating under reduced pressure, this method effectively isolates the cyclic carbonate from the catalyst matrix, achieving purities greater than 99.5% essential for sensitive applications like lithium-ion battery electrolytes.
Q: Is this process scalable for industrial production of battery-grade electrolytes?
A: Yes, the process is specifically designed for industrial scalability. The use of continuous flow reactors eliminates the bottlenecks associated with batch processing, such as heating and cooling cycles, allowing for consistent throughput. Furthermore, the closed-loop catalyst recycling system minimizes raw material consumption, making it economically viable for large-scale manufacturing of high-purity electronic chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclic Carbonate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced continuous manufacturing processes is key to securing a competitive edge in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102250052A are fully realized in practical, industrial settings. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of cyclic carbonate meets the exacting standards required for battery electrolytes and pharmaceutical applications. Our commitment to technical excellence ensures that our clients receive not just a commodity chemical, but a high-performance material backed by robust process validation.
We invite you to engage with our technical procurement team to discuss how our continuous synthesis capabilities can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how our process efficiencies translate into tangible financial benefits for your organization. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique project requirements, ensuring a seamless integration of our high-purity cyclic carbonates into your production workflow.
