Advancing Pharmaceutical Intermediate Synthesis via Metal-Free Halogen Bond Organocatalysis
Advancing Pharmaceutical Intermediate Synthesis via Metal-Free Halogen Bond Organocatalysis
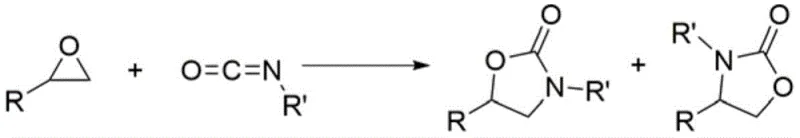
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards sustainable, metal-free methodologies, driven by stringent regulatory requirements and the need for cost-effective scalability. Patent CN113072517B introduces a groundbreaking synthetic method for five-membered oxygen-containing heterocyclic compounds, specifically leveraging carbon-halogen bond catalysis to facilitate the [3+2] cyclization of epoxides with heteroaccumulated dienes. This technology represents a significant departure from traditional transition metal catalysis, offering a robust pathway to synthesize valuable scaffolds such as oxazolidinones and thiocyclic carbonates without the burden of heavy metal contamination. For R&D directors and procurement specialists alike, this innovation addresses the critical pain points of impurity control and supply chain continuity, positioning it as a vital tool for the reliable production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted five-membered heterocycles has relied heavily on metal ion catalysts or metal complex systems, which, despite their efficacy, introduce unavoidable toxic metal residues into the final product. In the context of pharmaceutical intermediate manufacturing, the presence of trace metals necessitates rigorous and expensive purification protocols, often involving specialized scavengers that drive up operational costs and extend lead times. Furthermore, previous non-covalent organocatalytic attempts, such as hydrogen bond catalysis, have struggled with limited scope and often require harsh thermal conditions exceeding 100°C to achieve acceptable conversion rates. A particularly persistent challenge in this domain is regioselectivity; for instance, the reaction of epoxides with isocyanates can yield multiple isomeric oxazolidinones depending on the nucleophilic attack site, complicating isolation and reducing overall yield.
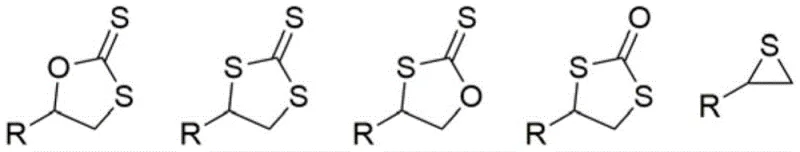
The complexity escalates when utilizing carbon disulfide as a substrate, where the intermediate dithiocarbonate species are highly reactive and prone to radical-mediated sulfur-oxygen exchange. This phenomenon leads to the generation of a myriad of byproducts, including various thiocyclic carbonate derivatives, making the targeted synthesis of a single desired compound exceptionally difficult. Such lack of selectivity not only diminishes the atom economy of the process but also creates significant bottlenecks in downstream processing, as separating these structurally similar impurities often requires resource-intensive chromatographic techniques that are ill-suited for large-scale commercial operations.
The Novel Approach
In response to these enduring challenges, the patented methodology employs aryl-substituted 2-haloimidazolium salts as potent halogen bond donors to catalyze the [3+2] cycloaddition with unprecedented precision. By exploiting the anisotropic electrostatic distribution of the halogen atom, specifically the positive sigma-hole region along the R-X bond extension, this system activates the epoxide substrate through strong, directional non-covalent interactions. This mechanism effectively lowers the activation energy barrier, allowing the reaction to proceed efficiently at mild temperatures ranging from 70°C to 80°C, thereby preserving the integrity of sensitive functional groups and reducing energy consumption. Crucially, this organocatalytic approach completely eliminates the need for transition metals, ensuring that the final product is free from metal residues and compliant with the strictest global pharmacopoeia standards without additional purification steps.
Mechanistic Insights into Halogen Bond Catalyzed Cyclization
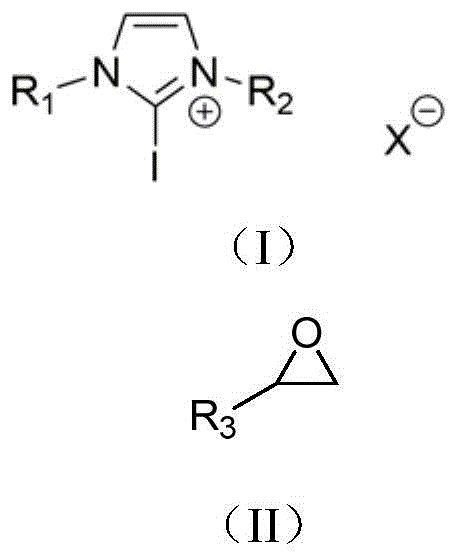
The core of this technological breakthrough lies in the unique electronic properties of the 2-haloimidazolium catalyst, which functions as a Lewis acid through its halogen bond donor capability. Unlike traditional hydrogen bonds, the halogen bond offers superior directionality and hydrophobicity, enabling the catalyst to precisely orient the epoxide and the heteroaccumulated diene within the transition state. The catalyst structure, characterized by bulky aryl substituents at the nitrogen positions and a halogen atom at the C2 position of the imidazolium ring, creates a sterically defined environment that guides the nucleophilic attack. This steric and electronic control is paramount in suppressing the formation of unwanted regioisomers during the reaction with isocyanates, ensuring that the ring-opening of the epoxide occurs at the specific carbon atom required to form the thermodynamically favored five-membered ring.
Furthermore, the mechanism effectively mitigates the radical scrambling issues associated with carbon disulfide reactions. By stabilizing the developing negative charge on the oxygen atom of the epoxide through halogen bonding, the catalyst facilitates a concerted or tightly ion-paired mechanism that bypasses the formation of free radical intermediates responsible for sulfur-oxygen exchange. This results in a clean reaction profile where the primary product is the targeted thiocyclic carbonate or oxazolidinone, drastically simplifying the impurity profile. For process chemists, this level of mechanistic control translates directly into higher crude purity, reducing the load on purification units and enhancing the overall throughput of the manufacturing line for these complex heterocyclic building blocks.
How to Synthesize Five-Membered Oxygen-Containing Heterocycles Efficiently
The practical implementation of this catalytic system is designed for ease of adoption in both laboratory and pilot-scale settings, utilizing readily available starting materials and straightforward operational procedures. The synthesis begins with the preparation of the 2-haloimidazolium catalyst, which can be achieved in near-quantitative yields by reacting the corresponding imidazolium chloride with N-iodosuccinimide in dichloromethane, requiring no complex purification. Once the catalyst is prepared, the cyclization reaction is performed by simply mixing the epoxide, the diene substrate, and the catalyst in a common organic solvent such as chlorobenzene or toluene. The detailed standardized synthesis steps for optimizing yield and selectivity are outlined below.
- Prepare the 2-haloimidazolium catalyst by reacting the corresponding imidazolium chloride with N-iodosuccinimide in dichloromethane at 40°C.
- Combine the epoxide substrate, heteroaccumulated diene (isocyanate or carbon disulfide), and the catalyst in an organic solvent such as chlorobenzene or toluene.
- Heat the reaction mixture to 70-80°C for approximately 24 hours under inert atmosphere, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, the adoption of this halogen bond catalyzed process offers transformative benefits that extend far beyond simple yield improvements. The elimination of transition metals from the catalytic cycle removes the necessity for expensive metal scavenging resins and the associated validation testing required to prove residual metal levels are within safe limits. This streamlining of the downstream process significantly reduces the cost of goods sold (COGS) and shortens the manufacturing cycle time, allowing for faster response to market demands. Additionally, the use of stable, easily synthesized organocatalysts enhances supply chain reliability, as these materials are not subject to the geopolitical volatility and price fluctuations often seen with precious metal catalysts like palladium or rhodium.
- Cost Reduction in Manufacturing: The metal-free nature of this process fundamentally alters the cost structure of producing high-value heterocyclic intermediates. By obviating the need for costly metal removal technologies and reducing the number of purification steps, manufacturers can achieve substantial savings in both material and operational expenditures. The mild reaction conditions further contribute to cost efficiency by lowering energy requirements compared to high-temperature alternatives, while the high selectivity minimizes waste generation and maximizes the utilization of raw materials, driving down the effective cost per kilogram of the final active pharmaceutical ingredient precursor.
- Enhanced Supply Chain Reliability: Dependence on specialized metal catalysts often introduces single points of failure in the supply chain, where shortages can halt production entirely. This organocatalytic method utilizes robust, shelf-stable salts that can be sourced or synthesized from commodity chemicals, ensuring a continuous and reliable supply of critical reagents. The simplified logistics of handling non-hazardous organic salts, as opposed to air-sensitive or toxic metal complexes, also reduces storage and transportation costs, making the overall supply chain more resilient and adaptable to changing production volumes.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies environmental and safety challenges, particularly those involving heavy metals and harsh reaction conditions. This technology aligns perfectly with green chemistry principles by operating at moderate temperatures and generating minimal hazardous waste, facilitating easier regulatory approval for commercial scale-up. The absence of metal contaminants simplifies waste stream treatment and disposal, reducing the environmental footprint of the manufacturing facility and ensuring long-term compliance with increasingly stringent global environmental regulations regarding industrial effluent.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel catalytic technology in industrial settings. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the operational advantages. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production workflows for complex organic synthesis.
Q: Why is halogen bond catalysis superior to metal catalysis for pharmaceutical intermediates?
A: Halogen bond catalysis eliminates the risk of toxic metal residue contamination, which is a critical regulatory hurdle in API manufacturing. Unlike traditional metal complexes that require expensive and time-consuming scavenging steps to meet ppm-level limits, this organocatalytic approach ensures a inherently cleaner product profile, simplifying downstream purification and reducing overall production costs.
Q: How does this method address the selectivity challenges in synthesizing thiocyclic carbonates?
A: Conventional methods often suffer from radical-mediated sulfur-oxygen exchange, leading to complex mixtures of byproducts. The patented 2-haloimidazolium catalyst utilizes specific non-covalent halogen bonding interactions to activate the epoxide selectively, suppressing side reactions and enabling the targeted synthesis of single isomer products with high fidelity.
Q: What are the typical reaction conditions for this organocatalytic cyclization?
A: The process operates under mild thermal conditions, typically between 70°C and 80°C, which is significantly lower than many non-covalent catalytic systems that require temperatures exceeding 100°C. This moderate temperature range enhances energy efficiency and improves the stability of sensitive functional groups often present in complex pharmaceutical building blocks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxazolidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalytic methods like the one described in CN113072517B for the production of next-generation pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial realities. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of high-purity oxazolidinone or thiocyclic carbonate meets the exacting standards required by global regulatory bodies.
We invite forward-thinking partners to collaborate with us to leverage this metal-free technology for their specific pipeline needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis to quantify the economic benefits of switching to this organocatalytic route for your specific molecule. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for efficiency, compliance, and long-term success in the competitive pharmaceutical marketplace.
