Advanced Copper-Catalyzed Synthesis of Quinoline Derivatives for Commercial Scale-Up
Introduction to Next-Generation Quinoline Synthesis
The landscape of heterocyclic chemistry is constantly evolving, driven by the urgent need for greener, more efficient synthetic pathways that can meet the rigorous demands of modern pharmaceutical manufacturing. A significant breakthrough in this domain is documented in patent CN112538045A, which details a novel method for synthesizing quinoline derivatives under the catalysis of copper. This technology represents a paradigm shift from classical approaches, utilizing readily available 2-aminobenzyl alcohol and acetophenone derivatives as starting materials to directly construct the quinoline skeleton. By leveraging a stable copper catalyst system combined with nitrogen-containing ligands, this process achieves high yields under remarkably mild conditions, specifically at room temperature. For R&D directors and process chemists, this patent offers a compelling alternative to traditional routes that often suffer from harsh reaction environments and poor atom economy. The ability to operate without inert atmosphere protection further simplifies the operational complexity, making it an attractive candidate for both laboratory optimization and industrial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinoline scaffold has relied heavily on classical condensation reactions such as the Friedländer synthesis or Skraup reaction. While these methods are well-established in academic literature, they present significant hurdles for modern commercial application. The Friedländer synthesis, for instance, typically requires strong acidic or basic conditions and elevated temperatures, which can lead to the degradation of sensitive functional groups often present in complex drug candidates. Furthermore, alternative modern approaches involving azido-compounds, while effective in building nitrogen heterocycles, introduce severe safety hazards due to the explosive nature of azides and their high toxicity. These factors not only increase the cost of safety compliance and waste disposal but also limit the substrate scope, preventing the synthesis of diverse libraries required for drug discovery. Consequently, there is a critical industry demand for a methodology that balances efficiency with safety and environmental sustainability.
The Novel Approach
The method disclosed in patent CN112538045A addresses these challenges through an elegant copper-catalyzed oxidative cyclization strategy. By employing inexpensive cuprous iodide (CuI) or cuprous bromide (CuBr) as the catalyst, the reaction proceeds smoothly at room temperature, eliminating the need for energy-intensive heating or cryogenic cooling. The use of 2-aminobenzyl alcohol as a key building block allows for a direct and atom-economical assembly of the quinoline core, avoiding the generation of excessive byproducts. As illustrated in the reaction scheme below, the transformation is robust and tolerates a wide variety of substituents on the acetophenone ring.
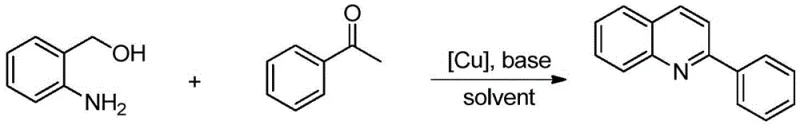
This novel approach not only simplifies the synthetic workflow but also aligns perfectly with the principles of green chemistry by minimizing hazardous reagent usage. The reaction system utilizes common organic solvents like toluene or tetrahydrofuran and simple inorganic bases such as potassium carbonate, ensuring that the process remains cost-effective and environmentally benign. For procurement teams, this translates to a supply chain less dependent on exotic or regulated chemicals, thereby enhancing overall supply security.
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
From a mechanistic perspective, this transformation likely proceeds through a copper-mediated dehydrogenative coupling pathway. The copper catalyst, coordinated by bidentate nitrogen ligands like 2,2'-bipyridine or phenanthroline, activates the benzylic C-H bond of the 2-aminobenzyl alcohol. This activation facilitates an initial condensation with the carbonyl group of the acetophenone derivative, followed by an intramolecular cyclization. The presence of the base is crucial for neutralizing protons generated during the process and driving the equilibrium forward. Unlike palladium-catalyzed cross-couplings which often require expensive ligands and strict oxygen-free conditions, this copper system appears robust enough to tolerate ambient conditions, suggesting a unique catalytic cycle that efficiently manages the oxidative steps required to aromatize the dihydroquinoline intermediate. This mechanistic simplicity is a key driver for the high reproducibility observed across different substrate classes.
Furthermore, the impurity profile of this reaction is exceptionally clean, which is a major advantage for pharmaceutical intermediate manufacturing. The high regioselectivity ensures that the quinoline nitrogen is incorporated at the correct position without forming isomeric byproducts that are difficult to separate. The patent data indicates that even with sterically hindered substrates, such as 2-methylacetophenone, or electronically diverse substrates like 4-chloroacetophenone, the reaction maintains high fidelity. For example, the synthesis of chloro-substituted quinolines proceeds with excellent yields, demonstrating the method's compatibility with halogenated intermediates often used in downstream cross-coupling reactions.
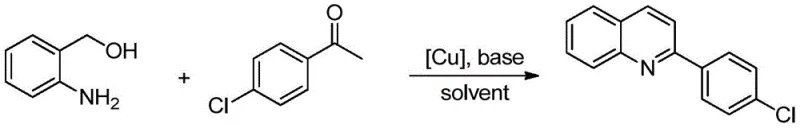
This level of control over the reaction outcome significantly reduces the burden on downstream purification processes. In a commercial setting, minimizing the formation of closely related impurities means less solvent consumption for chromatography and higher overall throughput. For quality control departments, the predictable nature of this catalytic system ensures consistent batch-to-batch quality, a critical parameter for regulatory compliance in API production.
How to Synthesize 2-Phenylquinoline Efficiently
Implementing this synthesis in a practical setting involves a straightforward protocol that minimizes operational risks. The general procedure entails mixing the amine alcohol and ketone substrates with the catalyst system in a standard reactor. The reaction is allowed to stir at ambient temperature, monitoring progress via TLC or HPLC until completion. Following the reaction, the workup is simplified to concentration and standard purification techniques. This ease of execution makes it accessible for both small-scale medicinal chemistry campaigns and larger pilot plant operations. For detailed standard operating procedures and specific molar ratios optimized for different substrates, please refer to the technical guide below.
- Mix 2-aminobenzyl alcohol, acetophenone derivatives, alkaline reagent (K2CO3/Na2CO3), copper catalyst (CuI/CuBr), and nitrogen-containing ligand in an organic solvent.
- React the mixture at room temperature for 6-12 hours under non-inert atmosphere conditions.
- Concentrate the reaction solution and perform column chromatography separation to isolate the pure quinoline derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed technology offers tangible strategic benefits beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the raw material portfolio. By relying on commodity chemicals like acetophenone derivatives and 2-aminobenzyl alcohol, manufacturers can mitigate the risks associated with sourcing specialized or monopolized reagents. This diversification of the supply base enhances resilience against market volatility and ensures continuous production capability. Moreover, the elimination of toxic azide reagents removes a significant regulatory and safety burden, reducing the costs associated with hazardous waste disposal and specialized containment infrastructure.
- Cost Reduction in Manufacturing: The economic impact of switching to this method is substantial, primarily driven by the replacement of precious metal catalysts with abundant copper salts. Copper iodide and bromide are orders of magnitude cheaper than palladium or rhodium alternatives, leading to direct material cost savings. Additionally, the ability to run reactions at room temperature eliminates the energy costs associated with heating large reactors or maintaining cryogenic conditions. The simplified workup procedure, which avoids complex extraction or quenching steps, further reduces labor and utility expenses, contributing to a lower overall cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of this chemical process translates directly into supply chain reliability. Since the reaction does not require an inert atmosphere (nitrogen or argon protection), the operational complexity is significantly lowered, reducing the likelihood of batch failures due to equipment leaks or operator error. The use of stable catalysts and ligands means that raw materials have long shelf lives and do not require special cold-chain logistics. This stability ensures that production schedules can be maintained consistently, reducing lead times for high-purity pharmaceutical intermediates and allowing for more responsive inventory management.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this methodology is inherently designed for scalability. The exothermic profile is manageable at room temperature, reducing the risk of thermal runaway in large vessels. From an environmental standpoint, the avoidance of heavy metals and toxic reagents aligns with increasingly stringent global environmental regulations. This "green" credential not only simplifies permitting for new manufacturing lines but also enhances the corporate sustainability profile, which is becoming a key criterion for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable basis for feasibility assessments. Understanding these nuances is essential for project managers evaluating the integration of this technology into existing production workflows.
Q: What are the advantages of this copper-catalyzed method over traditional Friedländer synthesis?
A: Unlike traditional methods requiring strong acids and harsh conditions, this copper-catalyzed protocol operates at room temperature with excellent functional group tolerance and avoids toxic azide reagents.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses cheap catalysts (CuI/CuBr), common solvents (toluene/THF), and does not require inert gas protection, making it highly scalable and cost-effective for commercial manufacturing.
Q: What is the substrate scope for the acetophenone component?
A: The method demonstrates broad universality, successfully accommodating acetophenone derivatives with electron-donating groups (methyl, amino) and electron-withdrawing groups (chloro, fluoro) at various positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed synthesis described in patent CN112538045A for the production of high-value quinoline scaffolds. As a leading CDMO partner, we possess the technical expertise to rapidly adapt and optimize this pathway for your specific project needs. Our facilities are equipped to handle complex organic transformations with precision, boasting extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of quinoline intermediate meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next drug development program. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team today to request specific COA data for our quinoline library and to discuss detailed route feasibility assessments for your target molecules.
