Scalable Visible Light Promoted Synthesis of Homoallylamine Intermediates for Commercial Production
Introduction to Advanced Visible Light Mediated Synthesis
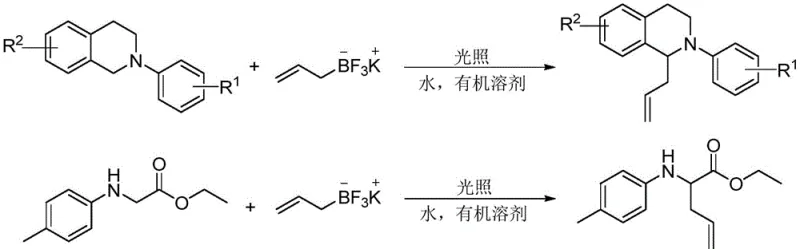
The landscape of organic synthesis is undergoing a paradigm shift towards sustainable and energy-efficient methodologies, particularly in the production of complex nitrogen-containing scaffolds. A groundbreaking development in this field is detailed in patent CN114874105B, which discloses a novel preparation method for homoallylamine compounds promoted by visible light and water. This technology represents a significant departure from traditional thermal or transition-metal catalyzed processes, offering a cleaner, more direct route to accessing high-value chemical intermediates. Homoallylamine structures are pivotal motifs found in numerous natural products and pharmaceutical agents, including protease inhibitors like Eponemycin and antitumor antibiotics. The ability to construct these C-C bonds at the alpha-position of amines under mild, photocatalyst-free conditions addresses critical pain points in modern process chemistry, specifically regarding environmental impact and operational simplicity. For R&D teams and procurement strategists alike, this innovation signals a move towards greener manufacturing protocols that do not compromise on yield or substrate scope.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of homoallylamine derivatives has relied heavily on Cross-Dehydrogenative Coupling (CDC) reactions that necessitate the use of stoichiometric oxidants and expensive transition metal catalysts. Traditional approaches often employ copper, palladium, or iron salts to mediate the activation of the C-H bond adjacent to the nitrogen atom. While these methods can be effective, they introduce significant downstream processing challenges, including the removal of trace heavy metals to meet stringent pharmaceutical purity standards. Furthermore, many conventional protocols require harsh reaction conditions, such as elevated temperatures or strong acidic/basic environments, which can lead to the degradation of sensitive functional groups and reduced overall yields. The reliance on toxic oxidants also generates substantial chemical waste, increasing the environmental footprint and disposal costs associated with large-scale manufacturing. These factors collectively hinder the economic viability and scalability of producing complex amine intermediates for the global supply chain.
The Novel Approach
In stark contrast, the methodology outlined in CN114874105B utilizes visible light irradiation and water as a promoter to drive the allylation reaction without any external photocatalyst. This approach leverages the inherent photochemical properties of the reaction system to generate reactive intermediates under ambient conditions. By employing allyl potassium trifluoroborate as the coupling partner, the reaction achieves high atom economy and excellent functional group tolerance. The absence of transition metal catalysts eliminates the need for costly metal scavengers and simplifies the purification workflow to basic silica gel chromatography. Moreover, the use of water as an additive not only enhances reaction efficiency but also aligns with green chemistry principles by reducing the reliance on purely organic solvent systems. This novel strategy effectively bypasses the limitations of traditional CDC reactions, offering a robust platform for the synthesis of diverse homoallylamine structures.
Mechanistic Insights into Visible Light Promoted Allylation
The mechanistic pathway of this transformation involves the generation of radical species through the absorption of visible light energy, initiating a sequence of single electron transfer (SET) events. Upon irradiation with blue LEDs, the amine substrate undergoes excitation, facilitating the abstraction of an alpha-hydrogen atom to form a stabilized alpha-amino radical intermediate. This radical species then engages in a nucleophilic attack on the allyl trifluoroborate salt, leading to the formation of the new carbon-carbon bond. The presence of water plays a synergistic role in this mechanism, potentially by stabilizing charged transition states or facilitating proton shuttling processes that are essential for the turnover of the catalytic cycle. Unlike traditional photoredox catalysis which requires exogenous dyes or metal complexes, this system appears to operate via a direct substrate-catalyzed or self-sensitized pathway, drastically reducing the complexity of the reaction mixture. Understanding these mechanistic nuances is crucial for R&D directors aiming to optimize reaction parameters for specific substrate classes and ensure consistent batch-to-batch reproducibility.
Impurity control is inherently superior in this photocatalyst-free system due to the mildness of the reaction conditions. The avoidance of strong oxidants prevents the over-oxidation of the amine substrate to iminium ions or N-oxides, which are common side products in metal-catalyzed CDC reactions. Additionally, the high chemoselectivity of the radical addition step ensures that other sensitive functional groups on the aromatic rings, such as cyano, bromo, or methoxy groups, remain intact throughout the process. This high level of selectivity translates directly into a cleaner crude reaction profile, reducing the burden on downstream purification units. For quality assurance teams, this means a lower risk of genotoxic impurities or metal residues in the final API intermediate, thereby streamlining regulatory filings and ensuring compliance with international pharmacopoeia standards for fine chemical intermediates.
How to Synthesize Homoallylamine Compounds Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it highly suitable for both laboratory scale optimization and pilot plant production. The standard protocol involves charging a reaction vessel with the N-aryl tetrahydroisoquinoline substrate and allyl trifluoroborate potassium salt in a precise 1:2 molar ratio to ensure complete conversion of the limiting reagent. The reaction is conducted in a mixed solvent system of acetonitrile and tetrahydrofuran (2:1 v/v) with the critical addition of water, which acts as the reaction promoter. Detailed standardized synthetic steps, including specific workup procedures and purification guidelines, are provided in the technical guide below to ensure successful replication of the high yields reported in the patent literature.
- Charge the reactor with N-aryl tetrahydroisoquinoline or ethyl 2-[(4-methylphenyl)amino]acetate and allyl trifluoroborate potassium salt in a 1: 2 molar ratio.
- Add a mixture of acetonitrile and tetrahydrofuran (2: 1 v/v) to achieve a substrate concentration of 0.08 mmol/mL, followed by the addition of water as a promoter.
- Irradiate the reaction mixture with blue LEDs (3W x 6) at room temperature for 8 to 12 hours, then purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this visible light promoted technology offers transformative advantages for procurement managers and supply chain heads focused on cost reduction and operational resilience. The elimination of precious metal photocatalysts represents a direct saving on raw material costs, as Iridium and Ruthenium complexes are among the most expensive reagents in fine chemical synthesis. Furthermore, the simplified workup procedure, which avoids complex metal scavenging resins or extensive aqueous washes, reduces solvent consumption and waste treatment expenses. The mild reaction conditions also imply lower energy consumption for heating or cooling, contributing to a reduced carbon footprint and lower utility costs per kilogram of product. These factors combine to create a highly competitive cost structure for the manufacturing of high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts and external photocatalysts significantly lowers the Bill of Materials (BOM) cost for each production batch. Without the need for expensive metal scavengers to meet residual metal specifications, the downstream processing becomes drastically simpler and cheaper. This qualitative shift in process design allows for substantial cost savings in homoallylamine manufacturing, making it economically feasible to produce these complex intermediates at a commercial scale without compromising on margin.
- Enhanced Supply Chain Reliability: The starting materials, including N-aryl tetrahydroisoquinolines and allyl trifluoroborate salts, are readily available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction against moisture and oxygen, facilitated by the aqueous promoter, minimizes the risk of batch failures due to minor environmental fluctuations. This reliability ensures consistent delivery schedules and reduces the lead time for high-purity pharmaceutical intermediates, allowing supply chain planners to maintain leaner inventory levels while meeting production demands.
- Scalability and Environmental Compliance: The use of visible light LEDs and room temperature conditions makes this process inherently safer and easier to scale compared to exothermic thermal reactions. The reduced generation of hazardous waste and the absence of heavy metals simplify environmental compliance and wastewater treatment protocols. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports corporate sustainability goals, positioning the supply chain for long-term viability in an increasingly regulated global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light promoted synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational capabilities and limitations of the method. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific product pipelines.
Q: Is an external photocatalyst required for this homoallylamine synthesis?
A: No, the patented process described in CN114874105B operates without the need for expensive transition metal photocatalysts such as Iridium or Ruthenium complexes. The reaction is driven directly by visible light interaction with the substrate system, significantly reducing raw material costs and eliminating the need for complex metal scavenging steps during purification.
Q: What is the specific role of water in this reaction mechanism?
A: Water acts as a crucial promoter in this transformation rather than just a solvent. It facilitates the reaction progress under mild conditions, likely by stabilizing radical intermediates or assisting in proton transfer steps during the cross-dehydrogenative coupling. This allows the reaction to proceed efficiently at room temperature with high functional group tolerance.
Q: Can this method be applied to substrates other than tetrahydroisoquinolines?
A: Yes, the methodology demonstrates broad substrate compatibility. Beyond N-aryl tetrahydroisoquinolines, the protocol is effective for glycine derivatives such as ethyl 2-[(4-methylphenyl)amino]acetate. This versatility makes it a valuable tool for generating diverse homoallylamine scaffolds found in various bioactive molecules and pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Homoallylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic technologies like the visible light promoted allylation described in CN114874105B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to manufacturing plant is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications for complex amine intermediates. We are committed to delivering high-quality homoallylamine compounds that adhere to the highest industry standards, supporting your drug development timelines with reliable and scalable supply solutions.
We invite you to collaborate with our technical team to explore how this cost-effective synthesis route can be integrated into your supply chain. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market for fine chemical intermediates.
