Revolutionizing Sumatriptan Production: A Deep Dive into Palladium-Catalyzed Indole Formation
Revolutionizing Sumatriptan Production: A Deep Dive into Palladium-Catalyzed Indole Formation
The pharmaceutical landscape for migraine treatment has long relied on the efficacy of Sumatriptan, a potent serotonin receptor agonist. However, the manufacturing of this critical active pharmaceutical ingredient (API) has historically faced significant challenges regarding yield and purity. A pivotal advancement in this domain is detailed in patent CN102212027B, which discloses a novel preparation method that fundamentally shifts the synthetic paradigm from traditional acid-catalyzed cyclization to a robust palladium-catalyzed intramolecular coupling strategy. This technological breakthrough addresses the longstanding issues of reaction instability and by-product formation that have plagued conventional routes. By leveraging a zero-valent palladium system under alkaline conditions, this method not only stabilizes the sensitive indole ring formation but also integrates a deprotection step, streamlining the entire production workflow. For global procurement and R&D teams, understanding this shift is crucial for securing a reliable supply chain of high-quality anti-migraine intermediates.
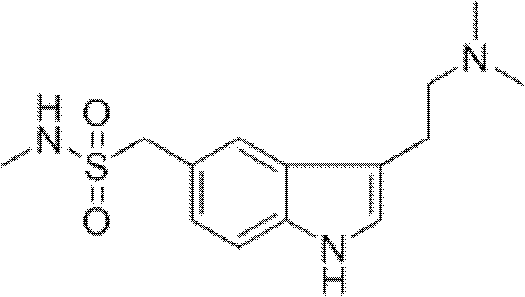
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sumatriptan and its key intermediates has predominantly relied on the Fischer-Indole synthesis method. This classical approach involves the acid-catalyzed rearrangement and cyclization of hydrazones to construct the indole nucleus. While theoretically sound, the practical application of this method in an industrial setting reveals severe deficiencies. The primary issue lies in the inherent instability of indole compounds under the strongly acidic conditions required for the Fischer-Indole reaction. This instability triggers a cascade of unwanted side reactions, most notably the formation of polymeric by-products which are notoriously difficult to separate from the desired product. Consequently, the effective conversion rate of raw materials is drastically compromised, with literature reporting cyclization yields hovering around a mere 15%. Such low efficiency not only inflates the cost of goods sold (COGS) due to wasted starting materials but also creates a bottleneck in purification, requiring extensive chromatographic or recrystallization steps that further erode profit margins and extend lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to the acidic fragility of the Fischer-Indole route, the methodology outlined in patent CN102212027B introduces a sophisticated transition metal-catalyzed pathway. This novel approach utilizes an intramolecular coupling reaction, specifically a Heck-type cyclization, mediated by a zero-valent palladium catalyst. By shifting the reaction environment from acidic to alkaline, the process effectively circumvents the degradation pathways that plague traditional methods. The alkaline conditions provide a much gentler milieu for the sensitive indole scaffold to form, thereby significantly enhancing reaction stability. Furthermore, this route is designed with operational efficiency in mind; it facilitates a one-pot procedure where the cyclization and the removal of the amino protecting group occur concurrently. This integration eliminates the need for isolated deprotection steps, reducing unit operations and solvent usage. For a reliable sumatriptan intermediate supplier, adopting this chemistry translates directly into a more robust, scalable, and economically viable manufacturing process that can consistently meet the stringent quality demands of the global pharmaceutical market.
Mechanistic Insights into Pd-Catalyzed Intramolecular Coupling
The core of this technological advancement lies in the mechanistic elegance of the palladium-catalyzed cyclization. The reaction initiates with the oxidative addition of the zero-valent palladium species into the carbon-bromine bond of the brominated aniline precursor. This step generates a reactive organopalladium intermediate that is poised for the subsequent migratory insertion. Unlike the chaotic acid-catalyzed rearrangements of the past, this organometallic pathway offers precise control over the bond-forming events. The palladium complex coordinates with the pendant alkene moiety introduced during the earlier condensation step, facilitating a regioselective insertion that constructs the five-membered pyrrole ring of the indole system. Following the insertion, a beta-hydride elimination restores the palladium catalyst to its active state while establishing the necessary unsaturation within the heterocyclic framework. Crucially, the presence of a base in the reaction mixture, such as potassium carbonate or triethylamine, serves a dual purpose: it neutralizes the hydrogen halide by-product to drive the equilibrium forward and simultaneously facilitates the hydrolysis of the trifluoroacetyl protecting group. This synergistic action ensures that the final deprotected Sumatriptan is obtained directly, minimizing the accumulation of protected intermediates that would otherwise require additional processing.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed alternatives. The specificity of the palladium catalyst minimizes non-selective electrophilic aromatic substitutions that often lead to regioisomers and polymeric tars in acidic media. The mild alkaline conditions prevent the protonation of the indole nitrogen, which is a common trigger for polymerization in traditional syntheses. As a result, the crude reaction profile is significantly cleaner, with the major impurities being easily removable through standard aqueous workups rather than complex chromatographic separations. This high level of chemoselectivity is paramount for achieving the stringent purity specifications required for API manufacturing. The ability to suppress polymeric by-products not only improves the isolated yield but also enhances the safety profile of the process by reducing the potential for energetic or toxic impurities to carry through to the final drug substance. For R&D directors evaluating process scalability, this mechanistic robustness provides the confidence needed to transition from laboratory benchtop to multi-ton commercial production without the fear of yield collapse.
How to Synthesize Sumatriptan Efficiently
The synthesis of Sumatriptan via this patented route involves a logical sequence of five distinct chemical transformations, each optimized for high conversion and ease of handling. The process begins with the reduction of a nitro precursor to an aniline, followed by regioselective bromination to install the handle for cyclization. Subsequent protection of the amine and coupling with an allylic alcohol side chain set the stage for the critical ring-closing step. The brilliance of this route lies in the convergence of these linear steps into a highly efficient final transformation. Detailed below is the strategic overview of the synthesis, highlighting the critical parameters that ensure success. For process chemists looking to implement this technology, adhering to the specific molar ratios and temperature controls described in the patent is essential to replicate the high yields and purity profiles observed in the examples. The following guide outlines the standardized operational framework derived from the patent data.
- Perform nitro reduction of N-methyl-4-nitrobenzenemethanesulfonamide using Pd/C and ammonium formate in methanol to obtain the amino intermediate.
- Execute bromination using N-bromosuccinimide (NBS) in DMF to introduce the bromine atom ortho to the amino group.
- Conduct acylation with trifluoroacetic anhydride to protect the amine, followed by Mitsunobu condensation with 4-dimethylamino-2-buten-1-ol.
- Finalize the synthesis via intramolecular Heck coupling using a zero-valent palladium catalyst in alkaline conditions to form the indole ring and remove the protecting group simultaneously.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this palladium-catalyzed technology represents a strategic opportunity to optimize the cost structure and reliability of the Sumatriptan supply chain. Traditional manufacturing methods, burdened by low yields and complex purifications, inherently carry higher risk premiums and volatile pricing. By transitioning to this novel route, manufacturers can eliminate the inefficiencies associated with acid-mediated degradation. The qualitative improvements in process stability mean that production batches are more consistent, reducing the likelihood of failed runs that disrupt supply continuity. Furthermore, the simplification of the post-reaction workup—specifically the avoidance of difficult polymer removal—translates into reduced consumption of solvents and chromatography media. These operational efficiencies compound to offer substantial cost savings in migraine drug manufacturing, allowing for more competitive pricing without compromising on quality. The robustness of the alkaline conditions also implies less corrosion on reactor equipment, extending asset life and lowering maintenance overheads.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous acid catalysts, combined with the significant improvement in cyclization yield, drives down the raw material cost per kilogram of the final product. By avoiding the formation of intractable polymeric by-products, the process reduces the need for costly purification resins and excessive solvent volumes. This streamlined approach ensures that the cost reduction in pharmaceutical intermediates manufacturing is realized through fundamental process intensification rather than mere supplier negotiation. The one-pot nature of the final step further consolidates processing time and utility consumption, delivering a leaner manufacturing footprint that is highly attractive for margin-sensitive generic drug production.
- Enhanced Supply Chain Reliability: The enhanced stability of the reaction intermediates under alkaline conditions mitigates the risk of batch-to-batch variability, a common pain point in the supply of complex heterocyclic intermediates. With a more predictable reaction profile, manufacturers can commit to tighter delivery schedules and larger volume contracts with greater confidence. The use of readily available reagents such as palladium acetate and common organic bases ensures that the supply chain is not dependent on exotic or single-source catalysts that could become bottlenecks. This resilience is critical for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API producers receive their materials on time to meet their own regulatory filing and commercial launch timelines.
- Scalability and Environmental Compliance: The transition from a corrosive acid process to a milder alkaline system significantly improves the environmental, health, and safety (EHS) profile of the manufacturing site. Reduced acid waste lowers the burden on wastewater treatment facilities and minimizes the generation of hazardous salt by-products. The high atom economy of the Heck coupling, coupled with the high conversion rates reported in the patent examples, means less chemical waste is generated per unit of product. This aligns perfectly with modern green chemistry initiatives and facilitates easier regulatory approval for new manufacturing sites. The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, allowing for seamless expansion from pilot plant quantities to full-scale commercial production without the need for drastic process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the comparative data and experimental examples provided in the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer. The answers reflect the specific advantages of the palladium-catalyzed method over legacy technologies, focusing on yield, purity, and operational simplicity. This section serves as a quick reference for decision-makers assessing the viability of this advanced manufacturing pathway.
Q: Why is the traditional Fischer-Indole method problematic for Sumatriptan synthesis?
A: The traditional Fischer-Indole method relies on strong acid catalysis, which causes instability in the indole intermediates, leading to significant polymerization by-products and low cyclization yields of approximately 15%, making purification difficult and costly.
Q: How does the palladium-catalyzed route improve reaction stability?
A: By utilizing an intramolecular Heck coupling reaction under alkaline conditions, this novel method avoids the harsh acidic environment that degrades indole compounds, thereby enhancing reaction stability, minimizing by-product formation, and significantly improving the overall yield of the cyclization step.
Q: What are the commercial benefits of the one-pot deprotection strategy?
A: The process allows for the simultaneous formation of the indole ring and removal of the trifluoroacetyl protecting group in a single pot, which drastically simplifies the post-processing workflow, reduces solvent consumption, and shortens the overall production cycle time for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sumatriptan Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN102212027B are fully realized in a commercial setting. We operate under stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Sumatriptan intermediate meets the highest international standards. Our commitment to quality is backed by a robust infrastructure capable of handling sensitive palladium-catalyzed reactions with precision, ensuring minimal residual metal content and superior impurity profiles. We understand that consistency is key in the pharmaceutical supply chain, and our processes are validated to deliver that reliability batch after batch.
We invite global partners to collaborate with us to leverage this cutting-edge technology for their Sumatriptan requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how this route can optimize your overall budget. We encourage you to contact us to request specific COA data and route feasibility assessments for your upcoming projects. By partnering with NINGBO INNO PHARMCHEM, you secure not just a supplier, but a strategic ally dedicated to advancing the efficiency and quality of your pharmaceutical manufacturing operations.
