Scalable Manufacturing of Sumatriptan Base via Optimized Fischer Indole Cyclization
Scalable Manufacturing of Sumatriptan Base via Optimized Fischer Indole Cyclization
The pharmaceutical landscape for migraine treatment has long been dominated by selective 5-HT1B/1D receptor agonists, with Sumatriptan standing as a cornerstone therapy since its initial launch in the early 1990s. As demand for this critical active pharmaceutical ingredient continues to grow globally, the efficiency of its synthetic manufacturing processes becomes a paramount concern for production strategists. Patent CN100497308C introduces a significant technological advancement in the large-scale preparation of 3-[2-(dimethylamino)ethyl]-N-methyl-1-hydro-indole-5-methanesulfonamide, commonly known as Sumatriptan base. This intellectual property outlines a robust methodology that leverages N-methyl-4-hydrazinobenzenemethanesulfonamide hydrochloride and dimethylaminobutyraldehyde acetals as key starting materials. By integrating a dual-catalyst system comprising polyphosphoric acid and sodium phosphate salts, the invention successfully addresses historical bottlenecks related to side reactions and purification complexity. The following analysis dissects the technical merits of this approach, offering valuable insights for R&D directors seeking process optimization and procurement managers focused on cost-effective supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sumatriptan has relied heavily on the Fischer indole cyclization strategy, yet early iterations of this chemistry faced substantial hurdles regarding industrial viability. The original route proposed by GlaxoSmithKline, while scientifically valid, necessitated the use of column chromatography for product purification, a technique that is notoriously difficult to translate from laboratory benchtop to multi-ton manufacturing scales. Furthermore, subsequent attempts to optimize this pathway, such as those documented in WO 2004/099141A1, struggled with inefficient conversion rates, reporting final yields as low as 10 percent despite avoiding chromatographic separation. These conventional methods often suffered from uncontrolled exothermic profiles and the formation of complex polymeric tars due to the harsh acidic environments required for cyclization. The reliance on expensive reagents like 5-bromoindole in alternative pathways further exacerbated cost structures, rendering them economically unfeasible for generic competition. Consequently, the industry has been in urgent need of a synthesis route that balances high purity with operational simplicity and economic efficiency.
The Novel Approach
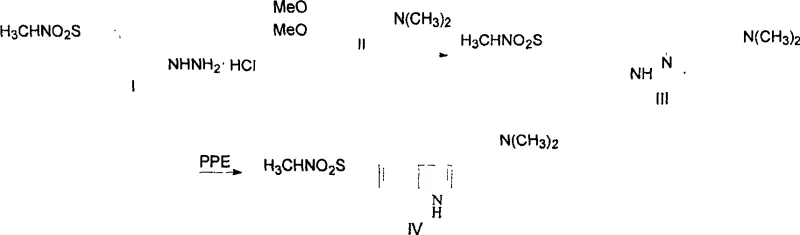
The methodology disclosed in CN100497308C represents a paradigm shift by introducing a buffered catalytic system that mitigates the aggressive nature of traditional polyphosphoric acid cyclizations. Instead of relying solely on strong acids that degrade sensitive functional groups, this novel approach incorporates sodium phosphate salts, specifically sodium dihydrogen phosphate, to modulate the acidity of the reaction medium. This buffering action is critical in suppressing side reactions that lead to impurity profiles difficult to remove. The process flow is streamlined into two distinct stages: the formation of a substituted phenylhydrazone intermediate under mild aqueous conditions, followed by the cyclization step in an organic solvent system. Crucially, the final purification is achieved through a combination of alkalization extraction and recrystallization, completely eliminating the need for silica gel column chromatography. This transition from chromatographic purification to crystallization-based isolation is a major breakthrough for cost reduction in pharmaceutical intermediate manufacturing, enabling true large-scale production capabilities.
Mechanistic Insights into PPA-Catalyzed Fischer Indole Cyclization
The core chemical transformation in this synthesis is the Fischer indole cyclization, a classic organic reaction that constructs the indole ring from a phenylhydrazone and an aldehyde or ketone derivative. In the context of Sumatriptan production, the mechanism involves the acid-catalyzed rearrangement of the phenylhydrazone intermediate derived from N-methyl-4-hydrazinobenzenemethanesulfonamide. The presence of polyphosphoric acid (PPA) serves as both the solvent and the proton source, facilitating the protonation of the hydrazone nitrogen and the subsequent [3,3]-sigmatropic rearrangement. However, the innovation lies in the co-presence of the phosphate salt. Mechanistically, the phosphate anion acts as a weak base within the highly acidic matrix, creating a micro-environment that prevents the over-protonation of the amine side chain. This protection is vital because excessive protonation of the dimethylamino group can deactivate the nucleophilic centers required for ring closure or lead to decomposition pathways. By fine-tuning the proton activity, the catalyst system ensures that the cyclization proceeds selectively towards the desired 5-position sulfonamide indole structure rather than forming regio-isomers or polymeric by-products.
Impurity control in this process is achieved through a sophisticated interplay of solubility and pH management during the workup phase. Following the cyclization reaction, which typically runs at mild temperatures between 20°C and 40°C to prevent thermal degradation, the reaction mixture is quenched into ice water. The addition of concentrated sodium hydroxide solution raises the pH to a highly alkaline range of 12 to 13. At this pH, the sulfonamide moiety and the indole nitrogen remain neutral or deprotonated, while many acidic by-products and residual phosphoric acid species become water-soluble salts. This allows for a clean separation where the desired Sumatriptan base partitions efficiently into the organic extraction solvent, such as ethyl acetate or dichloromethane. The subsequent recrystallization from isopropyl ether and acetone further refines the crystal lattice, excluding structurally similar impurities that might have co-extracted. This multi-stage purification logic ensures that the final API intermediate meets the rigorous purity specifications required for downstream salt formation and tabletting.
How to Synthesize Sumatriptan Base Efficiently
The execution of this synthesis protocol requires precise control over stoichiometry and temperature to maximize the benefits of the patented catalyst system. The process begins with the condensation of the hydrazine salt and the acetal in a water-alcohol mixture, where pH control is maintained to drive the equilibrium toward hydrazone formation without hydrolyzing the acetal prematurely. Once the intermediate is isolated or used in situ, the critical cyclization step demands the slow addition of the substrate into the pre-mixed PPA and phosphate catalyst solution to manage heat evolution. Detailed standard operating procedures regarding agitation speeds, addition rates, and crystallization cooling ramps are essential for reproducibility. For a comprehensive breakdown of the specific molar ratios, solvent volumes, and temperature profiles validated in the patent examples, please refer to the standardized synthesis guide provided below.
- Condense N-methyl-4-hydrazinobenzenemethanesulfonamide hydrochloride with 4-N,N-dimethylaminobutyraldehyde acetal in aqueous alcohol solvent to form the phenylhydrazone intermediate.
- Cyclize the phenylhydrazone in dichloromethane using a catalytic system of polyphosphoric acid and sodium dihydrogen phosphate at controlled temperatures below 40°C.
- Purify the crude product through alkaline extraction followed by recrystallization using a mixed solvent system of isopropyl ether and acetone to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound advantages for procurement managers and supply chain directors tasked with securing reliable sources of high-purity pharmaceutical intermediates. The most significant value driver is the elimination of column chromatography, which traditionally represents a massive bottleneck in terms of both consumable costs and processing time. By replacing silica gel columns with liquid-liquid extraction and crystallization, manufacturers can drastically reduce the consumption of expensive stationary phases and the vast quantities of solvents required for elution. This simplification translates directly into substantial cost savings in manufacturing overheads and waste disposal fees. Furthermore, the ability to recover and recycle organic solvents like dichloromethane and ethyl acetate, as highlighted in the patent description, contributes to a more sustainable and economically resilient production model. These efficiencies allow suppliers to offer more competitive pricing structures without compromising on the quality of the final product.
- Cost Reduction in Manufacturing: The removal of chromatographic purification steps fundamentally alters the cost basis of Sumatriptan production. Silica gel is a single-use consumable that generates significant solid waste, whereas the extraction and crystallization workflow utilized in this patent relies on reusable equipment and recoverable solvents. Additionally, the use of readily available starting materials such as hydrazine salts and acetals, rather than expensive brominated precursors, lowers the raw material entry cost. The mild reaction conditions also reduce energy consumption associated with heating and cooling, further enhancing the overall economic viability of the process for large-volume contracts.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex synthesis routes that are prone to failure or low yields. This optimized method demonstrates robust performance with crude purities around 84 percent achievable consistently, which provides a stable foundation for final purification. The reliance on commodity chemicals for the catalyst system (polyphosphoric acid and sodium phosphate) ensures that production is not held hostage by the supply constraints of exotic reagents. This stability allows for better forecasting and inventory management, reducing lead time for high-purity pharmaceutical intermediates and ensuring that downstream API manufacturers can maintain their own production schedules without interruption.
- Scalability and Environmental Compliance: Scaling chemical processes from the gram scale to the metric ton scale often introduces unforeseen engineering challenges, particularly regarding heat transfer and mixing. The described process operates at near-ambient temperatures (20-40°C), which significantly reduces the risk of thermal runaway incidents and simplifies reactor design requirements. Moreover, the simplified workup procedure minimizes the generation of hazardous waste streams, aligning with increasingly stringent environmental regulations. The ability to implement solvent recovery loops not only lowers costs but also reduces the facility's environmental footprint, making this a preferred route for companies committed to green chemistry principles and regulatory compliance in their supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Sumatriptan synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding the nuances of the catalyst system and purification steps is essential for evaluating the feasibility of technology transfer. We encourage technical teams to review these points carefully when assessing potential manufacturing partners or licensing opportunities.
Q: How does the PPA and sodium phosphate catalyst system improve the Fischer indole synthesis?
A: The combination of polyphosphoric acid (PPA) and sodium phosphate salts creates a buffered acidic environment that effectively regulates the pH of the reaction system. This modulation significantly reduces the generation of tarry by-products and promotes the stable existence of the indole product, eliminating the need for complex column chromatography purification.
Q: What are the purity specifications achievable with this large-scale preparation method?
A: According to patent data, the crude product obtained after extraction typically exhibits a purity of approximately 84% by HPLC. Following a straightforward recrystallization step using isopropyl ether and acetone, the final off-white solid product achieves a purity exceeding 98%, meeting stringent pharmaceutical intermediate standards.
Q: Is this synthesis route suitable for industrial scale-up compared to traditional methods?
A: Yes, this route is specifically designed for industrial application. Unlike earlier methods that required column chromatography or suffered from yields as low as 10%, this process utilizes mild reaction conditions (20-40°C), readily available raw materials, and solvent recovery systems, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sumatriptan Base Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the CN100497308C process are fully realized in practice. We maintain stringent purity specifications across all batches through our rigorous QC labs, utilizing advanced HPLC and spectroscopic methods to verify that every kilogram of Sumatriptan base meets the exacting standards required for global pharmaceutical markets. Our commitment to quality assurance means that clients can rely on us not just as a vendor, but as a strategic partner capable of navigating the complexities of API intermediate manufacturing.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized production capabilities, we can help you achieve significant reductions in total cost of ownership while securing a stable supply of critical materials. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced synthesis technologies can drive efficiency and reliability in your supply chain.
